Optimizing 2-Chloro-5-Nitropyridine Production for High-Purity Agrochemical and Pharmaceutical Applications
The chemical landscape for heterocyclic intermediates is constantly evolving, driven by the need for higher purity and more sustainable manufacturing processes. A pivotal advancement in this domain is documented in patent CN102040554A, which outlines a robust method for preparing 2-chloro-5-nitropyridine, a critical building block in the synthesis of Tri-Biocins and various antiviral agents. This technology addresses long-standing challenges in regioselective nitration and subsequent functional group transformation, offering a pathway that minimizes byproduct formation while maximizing total yield. For R&D Directors and Procurement Managers seeking a reliable agrochemical intermediate supplier, understanding the nuances of this three-step synthesis—comprising the formation of 2-amino-5-nitropyridine, its conversion to 2-hydroxy-5-nitropyridine, and final chlorination—is essential for securing a stable supply chain. The patent details specific molar ratios and temperature gradients that are not merely procedural but are fundamental to achieving the commercial viability required for modern fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chloronitropyridines has been plagued by issues related to isomer contamination and harsh reaction conditions that compromise safety and yield. Traditional methods often rely on direct nitration of chloropyridines or unoptimized chlorination of nitrophenols, which frequently result in significant quantities of unwanted isomers such as 2-amino-3-nitropyridine. These impurities are notoriously difficult to separate, leading to increased downstream purification costs and reduced overall process efficiency. Furthermore, conventional chlorination processes often require excessive amounts of phosphorus oxychloride and elevated temperatures that can degrade sensitive nitro groups, resulting in tarry byproducts that complicate waste treatment. For a procurement manager focused on cost reduction in fine chemical manufacturing, these inefficiencies translate directly into higher raw material consumption and unpredictable batch-to-batch consistency, creating significant supply chain risks.
The Novel Approach
The methodology presented in CN102040554A introduces a strategic sequence that circumvents these traditional pitfalls by prioritizing regioselectivity at the earliest stage of synthesis. By initiating the process with the controlled nitration of 2-aminopyridine using a specific mixture of nitrosonitric acid and sulfuric acid, the reaction is directed preferentially towards the 5-position, drastically reducing the formation of the 3-nitro isomer. This is followed by a mild diazotization step that converts the amino group to a hydroxyl group with high fidelity, setting the stage for an efficient chlorination. This novel approach ensures that the reaction conditions remain relatively mild throughout the sequence, with precise temperature controls preventing thermal runaway and decomposition. The result is a cleaner reaction profile that simplifies work-up procedures and enhances the purity of the final 2-chloro-5-nitropyridine product, aligning perfectly with the stringent quality requirements of pharmaceutical and agrochemical clients.
Mechanistic Insights into Regioselective Nitration and Diazotization
The core of this technological breakthrough lies in the meticulous control of electrophilic aromatic substitution during the initial nitration phase. The use of nitrosonitric acid in conjunction with sulfuric acid generates a highly reactive nitrating species that interacts with the protonated 2-aminopyridine substrate. The protocol mandates a gradual temperature increase, starting below 10°C and carefully ramping to 55-65°C, which is critical for managing the exothermic nature of the reaction while maintaining the orientation of the incoming nitro group. This thermal staging prevents the kinetic formation of undesired isomers and ensures that the thermodynamic product, 2-amino-5-nitropyridine, is favored. Following this, the diazotization step utilizes sodium nitrite in an acidic medium at sub-zero temperatures (-5 to 0°C) to form the diazonium salt intermediate. This low-temperature environment is vital for stabilizing the diazonium ion, preventing its premature decomposition into phenols or other side products before the intended hydrolysis occurs.
Furthermore, the final chlorination mechanism leverages the activation provided by the adjacent nitro group to facilitate nucleophilic aromatic substitution. The patent specifies the use of N,N-Diethylaniline and etamon chloride as additives in the phosphorus oxychloride medium, which act to enhance the reactivity of the hydroxyl leaving group. This catalytic system allows the chlorination to proceed effectively at 120-125°C, a temperature range that is high enough to drive the reaction to completion but low enough to preserve the integrity of the nitro functionality. The mechanistic precision of this route ensures that impurities are minimized at the source rather than being removed through costly purification later. 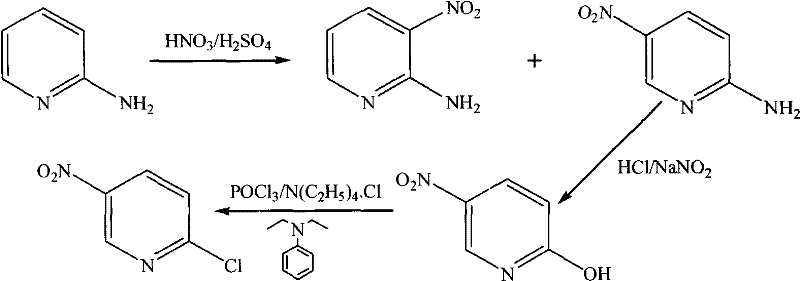
How to Synthesize 2-Chloro-5-Nitropyridine Efficiently
Implementing this synthesis route requires strict adherence to the operational parameters defined in the patent to ensure reproducibility and safety on a commercial scale. The process begins with the dissolution of 2-aminopyridine in sulfuric acid, followed by the controlled addition of the nitrating mixture, where maintaining the temperature below 30°C during addition is crucial for safety and selectivity. Subsequent steps involve precise pH adjustments and recrystallization protocols to isolate the intermediates with high purity before proceeding to the final chlorination. The detailed standardized synthesis steps, including specific molar ratios and work-up procedures, are outlined below to guide process engineers in replicating this high-yield pathway.
- Nitration of 2-aminopyridine using nitrosonitric acid and sulfuric acid at controlled temperatures below 65°C to form 2-amino-5-nitropyridine.
- Diazotization of the amino intermediate with sodium nitrite and hydrochloric acid at sub-zero temperatures (-5 to 0°C) followed by hydrolysis to yield 2-hydroxy-5-nitropyridine.
- Chlorination of the hydroxy derivative using phosphorus oxychloride with N,N-Diethylaniline catalyst at 120-125°C to finalize the 2-chloro-5-nitropyridine structure.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the adoption of this patented synthesis route offers substantial strategic advantages beyond mere technical elegance. The primary benefit is the significant enhancement in overall yield, which directly correlates to a reduction in the cost of goods sold (COGS). By minimizing the formation of isomeric byproducts, the process reduces the load on purification units and decreases the volume of organic waste generated, leading to lower environmental compliance costs. Additionally, the raw materials utilized, such as 2-aminopyridine and common mineral acids, are readily available in the global chemical market, ensuring that supply continuity is not jeopardized by niche reagent shortages. This reliability makes the manufacturer a preferred partner for long-term contracts where consistent volume delivery is paramount.
- Cost Reduction in Manufacturing: The elimination of complex purification steps required to remove isomeric impurities results in a streamlined production workflow that lowers operational expenditures. By achieving higher conversion rates in the chlorination step through the use of optimized catalytic additives, the consumption of expensive reagents like phosphorus oxychloride is minimized per kilogram of final product. This efficiency gain allows for a more competitive pricing structure without compromising margin, providing a tangible economic benefit to downstream buyers seeking cost reduction in agrochemical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, particularly the tolerance for standard industrial equipment and the avoidance of exotic catalysts, ensures that production can be scaled rapidly to meet demand spikes. The process design inherently reduces the risk of batch failures due to thermal instability, which is a common disruption point in nitration chemistry. Consequently, suppliers utilizing this method can offer more reliable lead times and maintain higher inventory levels of high-purity intermediates, securing the production schedules of their clients against market volatility.
- Scalability and Environmental Compliance: The moderate temperature profiles and the ability to recover and recycle solvents and acid streams make this process highly scalable from pilot plant to multi-ton commercial production. The reduction in tarry byproducts simplifies wastewater treatment protocols, aligning with increasingly stringent global environmental regulations. This sustainability aspect not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the supply chain, appealing to multinational corporations with strict vendor sustainability codes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-chloro-5-nitropyridine based on the patented methodology. These insights are derived directly from the experimental data and beneficial effects reported in the intellectual property documentation, providing clarity on process capabilities and quality standards. Understanding these details helps stakeholders make informed decisions regarding specification alignment and process integration.
Q: How does this patented method improve yield over conventional nitration processes?
A: The patented method utilizes a specific nitrosonitric acid mixture and strict temperature staging (<10°C to 65°C) which significantly reduces the formation of 2-amino-3-nitro isomers, thereby increasing the overall yield of the desired 5-nitro isomer.
Q: What are the critical safety controls during the diazotization step?
A: The process requires maintaining the reaction temperature between -5°C and 0°C during the addition of sodium nitrite to prevent premature decomposition of the diazonium salt and ensure safe gas evolution management.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process avoids extremely hazardous reagents and utilizes standard industrial unit operations like filtration and distillation, making it highly adaptable for commercial scale-up from 100 kgs to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-5-Nitropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent laboratory data to commercial reality requires deep technical expertise and rigorous quality control. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity demonstrated in CN102040554A are maintained at an industrial level. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-chloro-5-nitropyridine meets the exacting standards required for the synthesis of complex agrochemicals and pharmaceuticals. We are committed to being a reliable agrochemical intermediate supplier that bridges the gap between innovative chemistry and commercial success.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can evaluate the specific economic benefits of switching to this high-efficiency process for your manufacturing needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to validate the quality and scalability of our production capabilities firsthand.
