Advanced Diamine Salt Technology for Scalable Cephalosporin Antibiotic Production
Introduction to Advanced Cephalosporin Purification Technology
The pharmaceutical industry continuously seeks robust methodologies to enhance the purity and yield of critical antibiotic classes, particularly cephalosporins. Patent CN101031574A introduces a groundbreaking approach involving the preparation of cephalosporin antibiotics of formula (I) through the utilization of novel diamine salts. This technology addresses long-standing challenges in the isolation and purification of beta-lactam antibiotics, offering a pathway to significantly reduce impurity profiles while maintaining high structural integrity. The core innovation lies in the formation of stable salts using specific diamine compounds, which serve as superior intermediates compared to traditional alkali metal salts or bulky amine salts previously disclosed in prior art. By leveraging this chemistry, manufacturers can achieve a more streamlined production workflow that is inherently safer and more cost-effective for large-scale operations.
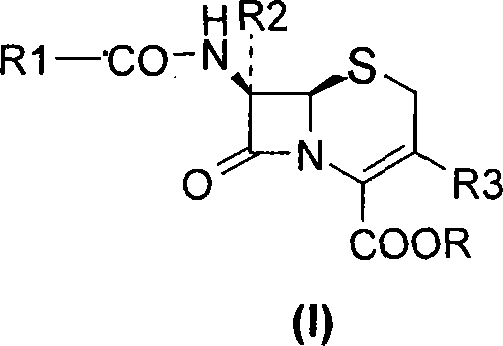
These novel salts, represented by general formula (II), exhibit exceptional stability throughout the entire preparation process, minimizing degradation risks that often plague sensitive beta-lactam rings. The versatility of this method allows for the synthesis of a wide range of commercially vital antibiotics, including cefdinir, cefixime, cefepime, and ceftriaxone derivatives. For R&D directors and process chemists, this represents a pivotal shift towards more predictable crystallization behaviors and simplified downstream processing. The ability to control the stoichiometry of the diamine counter-ion (where n equals 0.5 to 2) provides an additional lever for optimizing physical properties such as solubility and filterability, which are critical parameters for industrial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of cephalosporin antibiotics has relied on methods that often suffer from suboptimal yields and complex operational requirements. For instance, U.S. Patent No. 6,350,869 describes a process for purifying cefdinir via the preparation of an N,N-dicyclohexylamine salt, followed by hydrolysis. While effective to a degree, this multi-step sequence involving crude preparation, salt conversion, and subsequent hydrolysis results in an overall yield that is frequently unattractive for commercial manufacturing. Similarly, other prior art references, such as U.S. Patent No. 5,705,496 and U.S. Patent No. 5,026,843, utilize various amine salts like benzathine or dibenzylethylenediamine to isolate products from aqueous media. However, none of these established methods consistently deliver the desirable combination of high purity, high quantity, and operational simplicity required for modern cost reduction in API manufacturing. The reliance on specific, sometimes expensive, amine counter-ions without optimized recovery protocols can lead to significant material loss and increased waste generation.
The Novel Approach
In contrast, the methodology disclosed in CN101031574A utilizes a new class of diamine salts that overcome these historical inefficiencies. By employing diamines such as N,N'-diisobutylethylenediamine or N,N'-dicyclohexylethylenediamine, the process achieves a level of impurity removal that was previously difficult to attain without extensive chromatographic purification. The novel salts are stable, easily isolable via simple filtration, and can be converted back to the active drug substance with minimal degradation. This approach eliminates the need for harsh hydrolysis steps found in older patents, thereby preserving the integrity of the sensitive beta-lactam core. Furthermore, the flexibility in choosing the diamine component allows process engineers to tailor the physical characteristics of the intermediate to suit specific plant capabilities, ensuring a more reliable cephalosporin intermediate supplier experience for downstream partners.
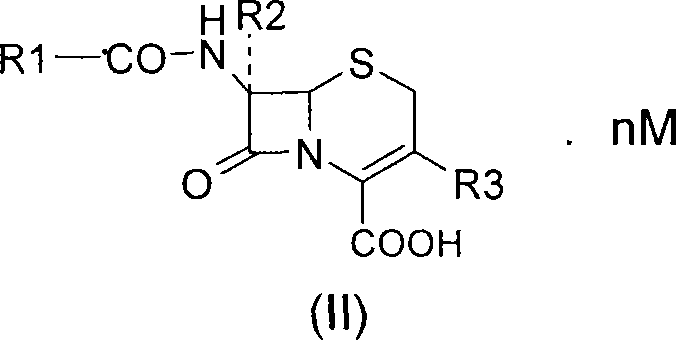
Mechanistic Insights into Diamine Salt Formation and Purification
The chemical mechanism underpinning this technology involves a precise interaction between the carboxylic acid moiety of the cephalosporin nucleus and the basic nitrogen centers of the diamine. When the cephalosporin acid (Formula VI) is treated with the diamine (Formula III) in a suitable solvent system, a proton transfer occurs, generating a zwitterionic or ionic lattice structure. This lattice formation is highly selective; the specific geometry and hydrogen bonding capabilities of the diamine cation favor the incorporation of the target cephalosporin anion while excluding structurally similar impurities and side-products. This phenomenon, known as differential solubility driven by salt formation, is the key to the high purity observed in the final product. The process typically operates under mild conditions, often between 0°C and 40°C, which prevents thermal degradation of the antibiotic scaffold.
Furthermore, the conversion of the diamine salt back to the free acid or sodium salt (Formula I) is achieved through controlled acidification or ion exchange. This step is critical for ensuring the final product meets pharmacopeial standards for residual amines. The use of cation exchange resins or specific sodium exchange reagents like sodium 2-ethylhexanoate allows for a clean swap of the diamine counter-ion without introducing new contaminants. This mechanistic clarity provides R&D teams with the confidence to scale the process, knowing that the critical quality attributes are controlled by fundamental thermodynamic principles rather than kinetic anomalies. The result is a high-purity pharmaceutical intermediate that consistently meets stringent regulatory specifications.
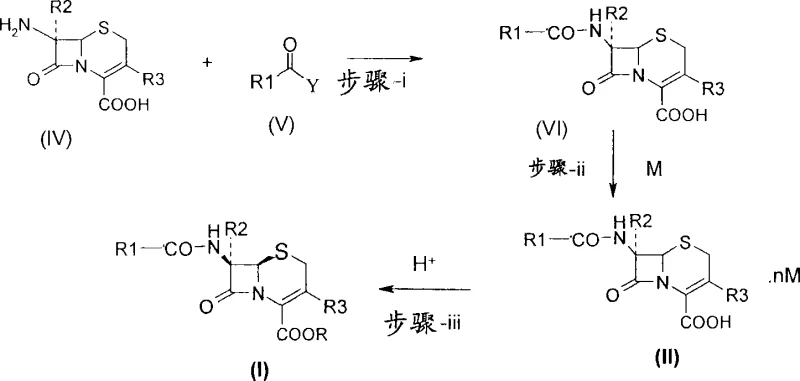
How to Synthesize Cephalosporin Diamine Salts Efficiently
The synthesis of these valuable intermediates follows a logical three-step sequence that is amenable to standard reactor setups found in most fine chemical facilities. The process begins with the condensation of the cephalosporin nucleus with the appropriate side-chain acid derivative to form the acylated intermediate. This is followed by the crucial salt formation step where the diamine is introduced to precipitate the product. Finally, the salt is converted to the desired final form. Detailed operational parameters, including solvent choices like isopropanol, acetone, or water mixtures, and temperature controls, are essential for maximizing yield. For a comprehensive guide on executing this synthesis with precision, please refer to the standardized protocol below.
- Condense a compound of general formula (IV) or its salt with a compound of general formula (V) to produce the intermediate compound of general formula (VI).
- Treat the compound of general formula (VI) with a diamine compound of general formula (III) to precipitate and isolate the novel salt of general formula (II).
- Convert the isolated salt of general formula (II) into the final cephalosporin antibiotic of general formula (I) using acid treatment or sodium exchange reagents.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this diamine salt technology offers substantial benefits for procurement managers and supply chain heads looking to optimize their API sourcing strategies. The primary advantage lies in the simplification of the purification train. By achieving high purity through crystallization rather than complex chromatography or multiple recrystallizations, the overall processing time is drastically reduced. This efficiency translates directly into lower manufacturing costs, as fewer unit operations mean reduced energy consumption, lower solvent usage, and decreased labor requirements. Additionally, the high yield associated with this method ensures that raw material utilization is maximized, providing significant cost reduction in API manufacturing without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive and inefficient purification steps, such as the hydrolysis required in older dicyclohexylamine salt methods, leads to a leaner production process. The ability to isolate the product simply by filtration and drying reduces the dependency on specialized equipment and minimizes solvent recovery costs. Furthermore, the diamines used are commercially available and cost-effective, ensuring that the input material costs remain stable and predictable for long-term budgeting.
- Enhanced Supply Chain Reliability: The stability of the novel diamine salts allows for flexible inventory management. Unlike unstable free acids that may degrade upon storage, these salts can be stockpiled as robust intermediates, decoupling the synthesis of the nucleus from the final formulation steps. This buffer capacity enhances supply chain resilience against demand fluctuations. Moreover, the use of common solvents and reagents reduces the risk of supply disruptions associated with specialty chemicals, ensuring a continuous flow of materials for commercial scale-up of complex pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily replicated from laboratory to pilot to production scale. The mild temperatures and ambient pressure requirements reduce safety risks and capital expenditure on high-specification reactors. From an environmental standpoint, the higher yields and simplified workup result in less chemical waste generation. The potential for solvent recycling, particularly of alcohols and ketones used in the precipitation steps, aligns with green chemistry principles and helps facilities meet increasingly strict environmental compliance regulations.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common inquiries regarding the chemical and operational aspects of the diamine salt process. These insights are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for process development discussions. Understanding these nuances is key to successfully integrating this method into existing production lines.
Q: What are the advantages of using diamine salts for cephalosporin purification?
A: According to patent CN101031574A, novel diamine salts provide superior stability throughout the preparation process compared to traditional alkali metal salts. They facilitate the removal of impurities from aqueous media more effectively, resulting in higher purity and attractive yields for antibiotics like cefdinir and cefixime.
Q: Which specific diamines are recommended for this process?
A: The patent specifies several effective diamines including N,N'-diisobutylethylenediamine, N,N'-dicyclohexylethylenediamine, N,N'-di-(p-anisyl)ethylenediamine, and N,N'-dicyclopentylethylenediamine. These form stable crystalline salts that are easy to filter and dry.
Q: How is the final active pharmaceutical ingredient recovered from the salt?
A: The final compound of formula (I) is recovered by treating the diamine salt (formula II) with acids such as HCl or sulfuric acid, or by using sodium exchange reagents like sodium 2-ethylhexanoate. This step regenerates the free acid or sodium salt form of the antibiotic suitable for formulation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cephalosporin Antibiotics Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced purification technologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial reality is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of cephalosporin intermediate meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of beta-lactam chemistry with precision and reliability.
We invite you to collaborate with us to leverage this innovative diamine salt technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to specific COA data and route feasibility assessments that will empower your decision-making process. Contact us today to discuss how we can support your supply chain with high-quality, cost-effective cephalosporin solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
