Advanced Manufacturing of Z-Isomer Cephalosporin Intermediates via Low-Temperature Wittig Reaction
The pharmaceutical industry continuously seeks robust synthetic routes for advanced beta-lactam antibiotics, particularly third-generation cephalosporins like Cefditoren. Patent CN1265662A introduces a groundbreaking methodology for the selective preparation of Z-isomers of 3-(2-substituted vinyl) cephalosporins, addressing a long-standing challenge in stereochemical control. This technology enables the high-yield production of critical intermediates where the spatial arrangement of the vinyl side chain dictates biological efficacy. The Z-configuration is paramount for Gram-negative coverage, making this process a vital asset for manufacturers aiming to produce high-quality active pharmaceutical ingredients (APIs) and their precursors.
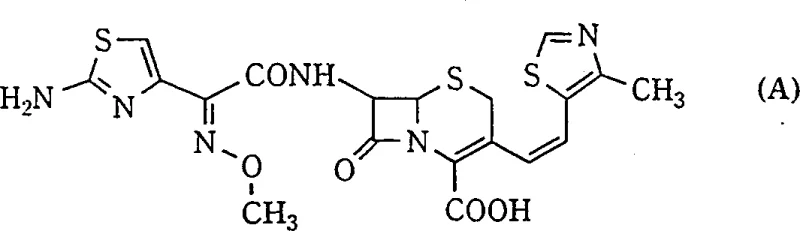
Conventionally, the synthesis of these compounds relies on the Wittig reaction, which notoriously produces a mixture of Z and E isomers. Traditional protocols often operate at room temperature using dichloromethane or aqueous mixtures, resulting in poor selectivity ratios, typically around 4.7:1 in favor of the Z-isomer. This lack of selectivity necessitates cumbersome downstream processing, such as repeated recrystallization or column chromatography, to isolate the biologically active Z-form. These purification steps not only degrade overall yield but also introduce significant operational costs and solvent waste, creating a bottleneck in the supply chain for reliable cephalosporin intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Existing manufacturing techniques for 3-vinyl cephalosporins suffer from inherent thermodynamic limitations that favor the formation of unwanted E-isomers alongside the desired Z-product. When standard phosphorane ylides react with thiazole aldehydes at ambient temperatures, the energy barrier difference between the transition states leading to Z and E isomers is insufficient to drive high stereoselectivity. Consequently, manufacturers are forced to deal with complex reaction mixtures where the E-isomer acts as a persistent impurity. Separating these geometric isomers is chemically difficult due to their similar physical properties, often requiring silica gel chromatography which is impractical for large-scale industrial production. This results in substantial material loss, extended production cycles, and increased environmental burden due to excessive solvent consumption.
The Novel Approach
The innovative process described in the patent data revolutionizes this landscape by manipulating reaction kinetics through precise solvent engineering and temperature control. By employing a mixed solvent system comprising a chlorinated hydrocarbon and a lower alkanol, specifically optimized at volume ratios between 1:3 and 1:0.25, the reaction environment is tuned to favor the Z-transition state. Crucially, conducting the reaction at low temperatures, ranging from +5°C down to -50°C, drastically suppresses the formation of the E-isomer. This approach allows for the direct generation of the target compound with exceptional purity, often exceeding 94% Z-isomer content directly from the reaction pot.
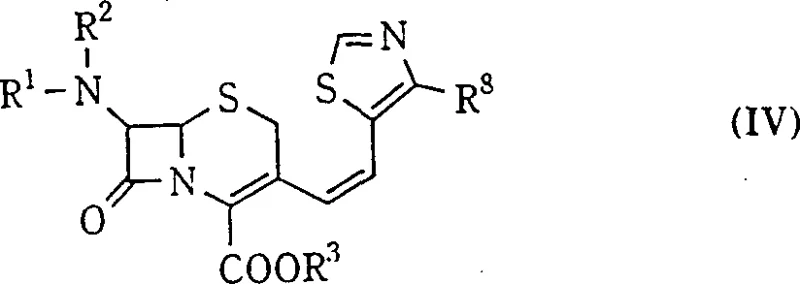
This methodological shift eliminates the need for complex purification trains. Instead of chromatography, the desired Z-isomer can be isolated simply by concentrating the reaction mixture and inducing crystallization with anti-solvents like methanol or butyl acetate. The E-isomer remains in the mother liquor in trace amounts, ensuring that the final crystalline product meets stringent purity specifications required for pharmaceutical applications. This streamlined workflow represents a significant leap forward in cost reduction in antibiotic manufacturing, offering a scalable solution that aligns with modern green chemistry principles by reducing solvent usage and waste generation.
Mechanistic Insights into Low-Temperature Stereoselective Wittig Reaction
The success of this process lies in the subtle interplay between solvent polarity, hydrogen bonding capability, and thermal energy. The phosphorane ylide intermediate, generated in situ from the corresponding phosphonium salt, exists in equilibrium between different conformers. In the presence of the mixed solvent system, specifically chloroform and n-propanol, the solvation shell around the ylide and the incoming aldehyde is modified. The lower alkanol likely participates in hydrogen bonding networks that stabilize the specific transition state leading to the Z-alkene. At reduced temperatures, the kinetic control of the reaction is enhanced, preventing the equilibration to the more thermodynamically stable but biologically inactive E-isomer.
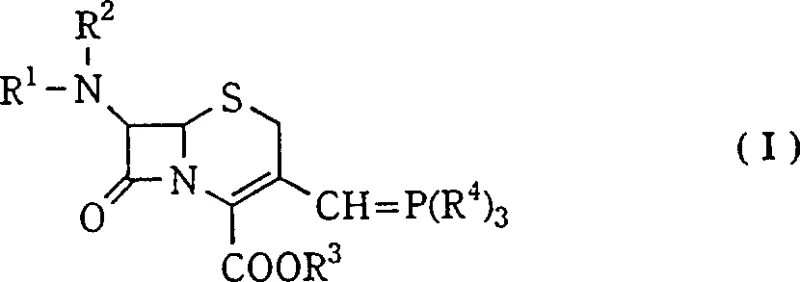
Furthermore, the choice of reactants plays a pivotal role in impurity control. The use of 4-substituted or unsubstituted thiazole-5-carbaldehydes in this specific solvent matrix ensures that the steric bulk of the thiazole ring interacts favorably with the cephalosporin nucleus during the bond-forming step. The low temperature freezes out competing reaction pathways that might lead to side products or isomerization post-reaction. This mechanistic understanding allows process chemists to fine-tune parameters such as the specific ratio of chloroform to n-propanol, with data suggesting that a 1:0.4 ratio at -20°C yields optimal results. Such precision ensures that the impurity profile is managed at the source, rather than relying on end-of-pipe purification.
How to Synthesize Z-Isomer Cephalosporin Intermediates Efficiently
Implementing this synthesis requires strict adherence to the patented conditions to replicate the high selectivity reported. The process begins with the preparation of the phosphonium salt from the 3-halomethyl cephalosporin, followed by ylide generation under basic conditions. The critical step involves cooling the reaction mixture to the specified sub-zero range before introducing the aldehyde component. Maintaining this temperature throughout the reaction period is essential to prevent thermal erosion of selectivity. Following the reaction, a simplified workup involving aqueous washes and solvent exchange facilitates the crystallization of the pure Z-isomer.
- Preparation of the phosphorane ylide intermediate from the corresponding 3-halomethyl cephalosporin and triphenylphosphine in a biphasic system.
- Cooling the reaction mixture to between -10°C and -30°C and adding a mixed solvent of chloroform and n-propanol.
- Reacting with 4-substituted thiazole-5-carbaldehyde, followed by washing and direct crystallization to isolate the high-purity Z-isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this low-temperature Wittig protocol offers transformative benefits beyond mere chemical elegance. The primary advantage is the drastic simplification of the downstream processing unit operations. By removing the requirement for column chromatography, manufacturers can significantly reduce the consumption of silica gel and elution solvents, which are major cost drivers in fine chemical production. This reduction in material usage translates directly into lower variable costs per kilogram of produced intermediate, enhancing the overall margin profile for the final API.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps removes a significant bottleneck in production throughput. Chromatography is not only expensive in terms of materials but also labor-intensive and time-consuming. By switching to a crystallization-based isolation method, facilities can increase batch frequency and reduce utility consumption associated with solvent recovery. Additionally, the higher yield of the desired isomer means that less starting material is wasted on producing inactive byproducts, optimizing the atom economy of the entire synthetic route.
- Enhanced Supply Chain Reliability: A simpler process with fewer unit operations inherently carries lower operational risk. Complex purification steps are prone to variability and equipment failures, which can disrupt supply schedules. This robust synthetic method ensures consistent batch-to-batch quality and predictable cycle times. For buyers seeking a reliable cephalosporin intermediate supplier, this reliability is crucial for maintaining uninterrupted production of finished dosage forms, especially for high-demand antibiotics where market shortages can have severe clinical consequences.
- Scalability and Environmental Compliance: Scaling up chromatographic processes from the lab to multi-ton production is notoriously difficult and often requires specialized, expensive equipment. In contrast, crystallization is a standard unit operation that scales linearly and predictably in existing reactor infrastructure. Furthermore, the reduction in solvent volume and the avoidance of silica waste align with increasingly stringent environmental regulations. This makes the process not only economically viable but also sustainable, reducing the carbon footprint associated with the commercial scale-up of complex beta-lactams.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this stereoselective synthesis. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains. The answers are derived directly from the experimental data and claims presented in the patent literature, ensuring accuracy and relevance for technical decision-makers.
Q: Why is the Z-isomer critical for Cefditoren activity?
A: The Z-configuration (cis-isomer) at the 3-vinyl position is essential for optimal binding to penicillin-binding proteins in Gram-negative bacteria, providing significantly higher antimicrobial potency compared to the E-isomer.
Q: How does the low-temperature solvent system improve selectivity?
A: Using a mixed solvent of chlorinated hydrocarbons and lower alkanols at temperatures below +5°C kinetically favors the formation of the Z-isomer, achieving ratios as high as 45:1 compared to conventional room temperature methods.
Q: Does this process require chromatographic purification?
A: No, the high selectivity of this novel method allows for the direct crystallization of the Z-isomer from the reaction mixture, eliminating the need for expensive and time-consuming column chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefditoren Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of stereochemical purity in the development of next-generation antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations like this low-temperature Wittig process are successfully translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical tools to verify Z/E ratios and impurity profiles, guaranteeing that every batch meets the highest international standards.
We invite global partners to collaborate with us on optimizing their cephalosporin supply chains. By leveraging our expertise in process intensification and cost-effective manufacturing, we can help you secure a stable source of high-purity intermediates. Please contact our technical procurement team to request a Customized Cost-Saving Analysis for your specific project. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals.
