Scalable Synthesis of High-Purity Eribulin Intermediates for Commercial API Production
Scalable Synthesis of High-Purity Eribulin Intermediates for Commercial API Production
The pharmaceutical industry continuously seeks robust and scalable pathways for complex oncology agents, and Eribulin mesylate stands as a paramount example of structural complexity meeting therapeutic efficacy. As detailed in patent CN108341828B, a novel process has been developed for the preparation of Eribulin and its critical intermediates, specifically targeting the efficient synthesis of compounds of Formula 5. This technology addresses the historical challenges associated with the total synthesis of Halichondrin B analogues, which are characterized by intricate macrocyclic ketone structures and multiple chiral centers. The innovation lies in a streamlined approach that leverages readily available natural sugar derivatives as chiral pool starting materials, ensuring high stereochemical fidelity from the outset. By optimizing the conversion of protected glucose derivatives into advanced intermediates, this method offers a viable solution for cost reduction in API manufacturing while maintaining the rigorous purity standards required for metastatic breast cancer treatments.
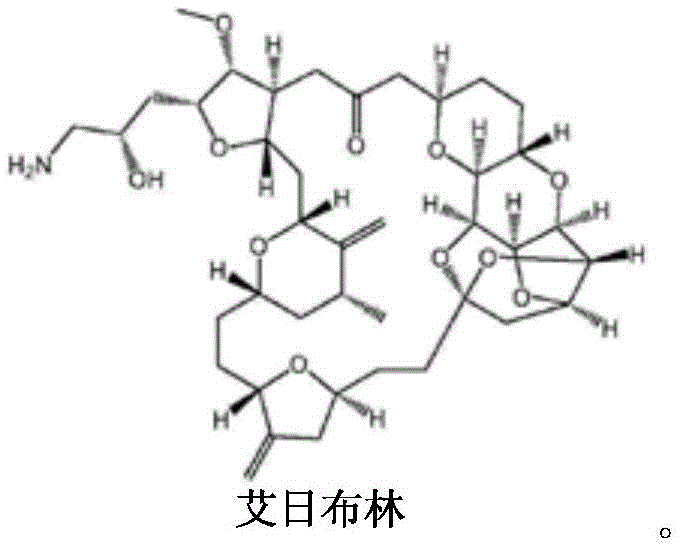
Eribulin is a first-in-class macrocyclic ketone analogue derived from the marine sponge product Halichondrin B. While highly effective, its commercial production has been hindered by the sheer difficulty of constructing its carbon backbone with the necessary stereochemical precision. Traditional routes often suffer from low overall yields due to the accumulation of losses over dozens of synthetic steps. The disclosed invention provides a strategic breakthrough by focusing on the preparation of key building blocks, such as the compound of Formula 5, which serves as a pivotal precursor for the C1-C13 fragment of the Eribulin molecule. This targeted intermediate strategy allows manufacturers to decouple the most challenging stereochemical setups from the later macrocyclization steps, thereby de-risking the overall supply chain. For reliable eribulin intermediate suppliers, mastering this specific fragment synthesis is the key to unlocking consistent, high-volume production capabilities that can support global clinical and commercial demands without the bottlenecks of legacy synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Halichondrin B and its analogues, as reported in earlier patents like US6214865 and US5436238, relied heavily on convergent strategies that joined multiple complex fragments. While scientifically elegant, these conventional methods often involve extensive protection and deprotection sequences that add significant mass intensity and waste to the process. The reliance on fragile intermediates that require cryogenic conditions or highly specialized reagents can severely limit commercial scale-up of complex pharmaceutical intermediates. Furthermore, controlling the stereochemistry at each junction point often necessitates costly chiral chromatography or resolution steps, which are economically unsustainable at the multi-kilogram or tonne scale. The accumulation of impurities from these lengthy sequences not only drives up the cost of goods sold (COGS) but also complicates the regulatory filing process due to the need for extensive impurity profiling and control strategies.
The Novel Approach
The novel approach disclosed in the present invention fundamentally simplifies the construction of the tetrahydrofuran core found in Eribulin intermediates. Instead of building the ring system from scratch, the process utilizes 1,2,5,6-diisopropylidene glucose, a cheap and abundant chiral pool material. The core innovation involves a direct oxidative cleavage of the 5,6-isopropylidene group to generate an aldehyde, which is then immediately subjected to a stereoselective olefination reaction. This sequence, transforming a compound of Formula 2 into a compound of Formula 4, effectively installs the necessary carbon chain length and unsaturation in a single operational logic. By employing specific phosphonium salt reagents or Horner-Wadsworth-Emmons reagents, the process ensures the formation of the desired alkene geometry with high selectivity. This reduction in step count and the use of robust, scalable reactions represent a significant leap forward in high-purity pharmaceutical intermediate manufacturing, offering a pathway that is both chemically efficient and economically viable for industrial application.
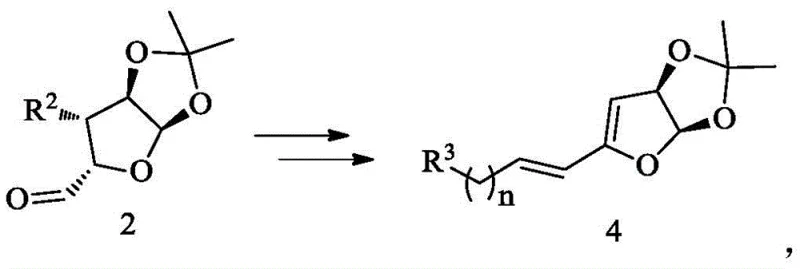
Mechanistic Insights into Stereoselective Olefination and Elimination
The heart of this synthetic advancement lies in the precise manipulation of the sugar scaffold to install the side chain required for the Eribulin backbone. The mechanism begins with the hydrolysis of the acetonide protecting group under acidic conditions, followed by oxidative cleavage using sodium periodate (NaIO4). This generates a reactive aldehyde species (Formula 2) which retains the stereochemical information of the original sugar. The subsequent reaction with a phosphonium salt (Formula 9) proceeds via a Wittig or Horner-Wadsworth-Emmons mechanism. In this critical step, the ylide attacks the carbonyl carbon to form an oxaphosphetane intermediate, which collapses to release the phosphine oxide and form the new carbon-carbon double bond. The choice of base and reaction conditions is crucial here; the patent specifies the use of bases like n-BuLi or tetrabutylammonium fluoride to facilitate the formation of the ylide and drive the elimination. This careful control ensures that the resulting alkene (Formula 3 or 4) possesses the correct E/Z geometry, which is essential for the biological activity of the final drug substance.
Furthermore, the process incorporates a sophisticated elimination strategy to refine the olefin structure. In embodiments where a leaving group (LG) such as a mesylate or tosylate is present on the sugar ring, a base-mediated elimination is employed to generate an enol ether or conjugated system (Formula 2d). This transformation is particularly valuable because it allows for the introduction of unsaturation directly into the ring system or the side chain without affecting other sensitive functional groups. The ability to perform these transformations in a 'one-pot' or 'one-pot multi-step' manner significantly enhances the process efficiency. By avoiding the isolation of unstable intermediates, the risk of decomposition or racemization is minimized. This mechanistic elegance translates directly into higher yields and superior purity profiles, addressing the primary concerns of R&D Directors who must ensure that every batch of intermediate meets stringent specifications before proceeding to the next stage of synthesis.
How to Synthesize Eribulin Intermediate Formula 5 Efficiently
The practical execution of this synthesis involves a sequence of well-defined unit operations that are amenable to standard chemical manufacturing equipment. The process initiates with the treatment of the protected glucose derivative with periodic acid in a solvent system such as ethyl acetate, effecting both deprotection and oxidative cleavage simultaneously. The resulting crude aldehyde is then reacted with the appropriate phosphorus ylide in an anhydrous environment, typically using tetrahydrofuran (THF) as the solvent. Following the olefination, the reaction mixture undergoes a workup to remove phosphine oxides, often via filtration or extraction. The subsequent steps involve catalytic hydrogenation to reduce the newly formed double bond and any ester functionalities to alcohols, followed by selective protection of the hydroxyl groups using silyl chlorides like TBDPSCl. Detailed standardized operating procedures for these transformations are critical for reproducibility.
- Hydrolyze the 5,6-isopropylidene protecting group of the starting glucose derivative and perform oxidative cleavage using periodic acid to generate the aldehyde intermediate.
- React the resulting aldehyde with a phosphonium salt reagent (Wittig or Horner-Wadsworth-Emmons) to form the olefinic intermediate with high stereocontrol.
- Perform catalytic hydrogenation to reduce the double bond and ester functionalities, followed by hydroxyl protection to yield the final Formula 5 intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage is the significant simplification of the raw material portfolio. By relying on glucose derivatives, the process taps into a commodity chemical market that is stable, high-volume, and cost-effective, unlike specialized chiral building blocks that may have limited suppliers. This shift drastically reduces the risk of supply disruption and price volatility, ensuring a more resilient supply chain for critical oncology ingredients. Moreover, the reduction in synthetic steps directly correlates with a reduction in manufacturing lead time. Fewer isolation and purification stages mean faster throughput in the production facility, allowing for quicker response to market demand fluctuations and more agile inventory management.
- Cost Reduction in Manufacturing: The streamlined nature of this process eliminates the need for multiple chromatographic purifications and cryogenic reactions that are energy-intensive and solvent-heavy. By consolidating steps, such as the simultaneous hydrolysis and oxidation, the consumption of utilities and consumables is substantially lowered. Additionally, the high stereochemical purity achieved early in the synthesis reduces the burden on downstream purification, minimizing yield losses associated with recrystallization or chiral separation. These efficiencies collectively drive down the cost of goods, making the production of Eribulin intermediates more economically sustainable and competitive in the global marketplace.
- Enhanced Supply Chain Reliability: The robustness of the chemistry described allows for greater flexibility in sourcing. Since the reagents used, such as triphenylphosphine and common bases, are widely available industrial chemicals, there is no dependency on single-source specialty vendors. This diversification of the supply base mitigates the risk of bottlenecks. Furthermore, the stability of the intermediates generated, such as the protected tetrahydrofuran derivatives, facilitates easier storage and transportation. This stability is crucial for maintaining a continuous supply flow to downstream API manufacturers, ensuring that production schedules are met without unexpected delays caused by material degradation.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The avoidance of hazardous reagents and the potential for solvent recycling align with modern green chemistry principles. By reducing the overall waste generation and improving the atom economy of the synthesis, manufacturers can meet increasingly stringent environmental regulations more easily. This compliance not only avoids potential fines but also enhances the corporate sustainability profile, which is becoming a key criterion for selection by major pharmaceutical partners seeking responsible supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages and operational details outlined in the patent documentation, providing clarity for technical teams evaluating the feasibility of this route for their own production needs. Understanding these nuances is essential for making informed decisions about technology transfer and process validation.
Q: How does this process improve stereochemical purity compared to conventional methods?
A: The process utilizes naturally derived sugar precursors which possess inherent chirality. By maintaining the stereochemical integrity through mild oxidative cleavage and controlled Wittig olefination conditions, the method achieves diastereomeric excess (d.e.) values exceeding 99.5%, significantly reducing the need for difficult chiral separations later in the synthesis.
Q: What are the key advantages for large-scale manufacturing of Eribulin intermediates?
A: The methodology allows for 'one-pot' or 'one-pot multi-step' transformations, such as the simultaneous hydrolysis and oxidative cleavage of protecting groups. This consolidation of steps minimizes solvent usage, reduces processing time, and eliminates the isolation of unstable intermediates, thereby enhancing overall process robustness and throughput for commercial production.
Q: Can this synthetic route be adapted for other halichondrin analogues?
A: Yes, the versatility of the phosphonium salt reagents and the modular nature of the protecting group strategies (such as TBDMS or Benzyl ethers) allow this core pathway to be adapted for synthesizing various halichondrin B analogues. The flexibility in the R-groups enables the construction of diverse macrocyclic ketone structures required for different therapeutic candidates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eribulin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the successful commercialization of complex oncology drugs like Eribulin. Our technical team has extensively analyzed the pathways described in CN108341828B and possesses the expertise to execute this chemistry with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including the analysis of diastereomeric excess and trace impurities, guaranteeing that every batch meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can improve your bottom line. We encourage you to contact our technical procurement team to request specific COA data from our pilot runs and to discuss route feasibility assessments for your project. By partnering with us, you secure a reliable source of high-purity intermediates that will support the uninterrupted development and commercial success of your therapeutic programs.
