Advanced Synthesis of Venlafaxine Hydrochloride Related Substance I for Pharmaceutical QC
Advanced Synthesis of Venlafaxine Hydrochloride Related Substance I for Pharmaceutical QC
The pharmaceutical industry faces increasing regulatory pressure to identify and quantify trace impurities in active pharmaceutical ingredients (APIs), particularly for widely prescribed antidepressants like Venlafaxine Hydrochloride. Patent CN112094230A, published in December 2020, addresses a critical gap in the supply chain for quality control materials by disclosing a robust synthesis method for Venlafaxine Related Substance I. This specific impurity, which arises from dehydration and subsequent cyclization during the manufacturing process, has historically been difficult to source as a certified reference standard. The inability to accurately quantify such related substances poses significant risks to patient safety and regulatory compliance. By establishing a reliable pathway to generate this complex molecule with high purity, this technology empowers analytical laboratories to enhance their detection capabilities and ensures stricter adherence to ICH guidelines regarding impurity profiling.
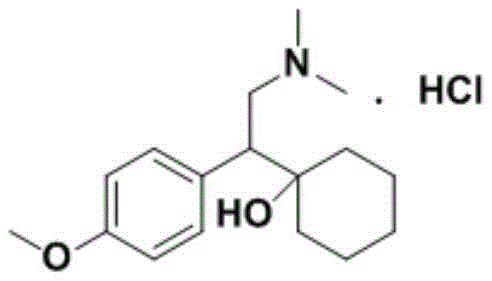
Venlafaxine Hydrochloride, chemically known as (±)-1-[2-(dimethylamino)-1-(4-methoxyphenyl)ethyl]cyclohexanol hydrochloride, is a potent serotonin-norepinephrine reuptake inhibitor (SNRI). Its complex chemical structure makes it susceptible to forming various degradation products and process-related impurities during synthesis. The presence of these impurities, even in minute quantities, can alter the pharmacological profile or introduce toxicity. Consequently, the development of precise analytical methods relies heavily on the availability of authentic impurity standards. The traditional reliance on self-contrast methods without specific reference compounds leads to poor accuracy in qualitative and quantitative determination. This patent introduces a targeted solution that transforms an unstable intermediate into a stable, isolable impurity standard, thereby mitigating clinical medication risks associated with unidentified contaminants.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in CN112094230A, the synthesis of Venlafaxine Hydrochloride typically involved the condensation of p-methoxyphenylacetonitrile with cyclohexanone, followed by reduction and methylation steps. While effective for producing the API, this conventional route inadvertently generates Related Substance I as an uncontrolled by-product. In standard manufacturing environments, the acidic conditions employed during the reduction step can catalyze the dehydration of the hydroxyl group, leading to the formation of a double bond and subsequent cyclization. However, because this transformation was historically viewed as an undesirable side reaction rather than a target synthesis, no dedicated protocols existed to isolate or reproduce this substance intentionally. Literature searches confirmed that neither the European Pharmacopoeia nor the United States Pharmacopoeia included specific monographs for Related Substance I, leaving manufacturers without a validated reference material. This absence forced quality control teams to rely on imprecise estimation techniques, compromising the integrity of batch release testing and stability studies.
The Novel Approach
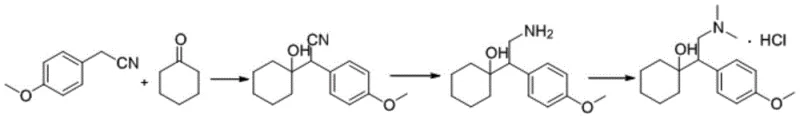
The novel approach detailed in the patent fundamentally shifts the paradigm by treating the formation of Related Substance I as a deliberate synthetic objective rather than an accidental occurrence. Instead of attempting to suppress the dehydration and cyclization pathways, the inventors have optimized these specific reaction mechanisms to maximize yield and purity. The method utilizes 1-[2-amino-1-(4-methoxyphenyl)ethyl]cyclohexanol as a strategic starting material, subjecting it to controlled acid-catalyzed dehydration to form a distinct alkene intermediate. This intermediate is then subjected to a cyclization reaction involving formaldehyde and formic acid. By decoupling the impurity formation from the main API synthesis line, manufacturers can produce large quantities of the reference standard independently. This independence ensures a steady supply of high-purity material specifically designed for calibration and validation purposes, eliminating the variability associated with extracting trace impurities from crude API batches.
Mechanistic Insights into Acid-Catalyzed Dehydration and Cyclization
The core chemical innovation lies in the precise manipulation of reaction conditions to drive the equilibrium towards the desired cyclized product. The first stage involves the dehydration of the beta-amino alcohol starting material. Under the catalysis of strong acids such as concentrated sulfuric acid or p-toluenesulfonic acid, the hydroxyl group is protonated and eliminated as water. This elimination reaction generates a reactive alkene species, identified in the patent as Intermediate II. The choice of solvent plays a pivotal role in this step; while dichloromethane is preferred for low-temperature reactions to control exothermicity, solvents like toluene allow for higher temperature reflux conditions that facilitate water removal via azeotropic distillation. The molar ratio of acid to substrate is carefully maintained between 1:1 and 1:3 to ensure complete conversion without promoting excessive polymerization or charring of the sensitive amine functionality.
Following the formation of the alkene intermediate, the second stage employs an Eschweiler-Clarke type mechanism adapted for ring closure. The introduction of formaldehyde and formic acid creates an electrophilic environment that facilitates the attack of the amine nitrogen on the electron-deficient carbons generated during the dehydration phase. This intramolecular cyclization results in the formation of a fused ring system characteristic of Related Substance I. The reaction is typically conducted at temperatures ranging from 80°C to 110°C, providing sufficient thermal energy to overcome the activation barrier for ring closure. The use of formic acid serves a dual purpose: it acts as both a solvent and a reducing agent, stabilizing the intermediate iminium ions formed during the reaction with formaldehyde. This mechanistic understanding allows for fine-tuning of the process parameters to minimize side products and achieve the reported HPLC purity of over 99%.
How to Synthesize Venlafaxine Related Substance I Efficiently
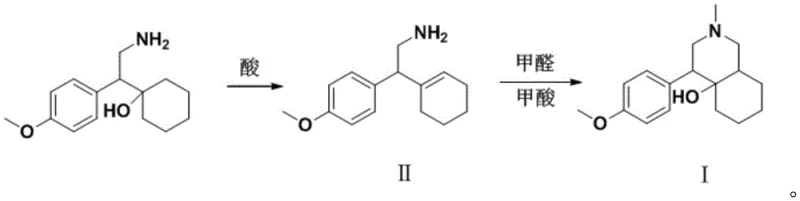
The synthesis protocol outlined in the patent offers a streamlined workflow that balances reaction efficiency with ease of purification. The process begins with the dissolution of the amino-alcohol precursor in a suitable organic solvent, followed by the controlled addition of the acid catalyst. Temperature management is critical during this exothermic dehydration phase to prevent the formation of tarry by-products. Once the intermediate is formed and isolated, it undergoes the cyclization step in an aqueous or alcoholic medium containing formaldehyde and formic acid. The reaction mixture is refluxed for an extended period, typically between 10 to 20 hours, to ensure complete conversion. Post-reaction workup involves pH adjustment to basic conditions to free the amine, followed by extraction into organic solvents like ethyl acetate. The final purification is achieved through column chromatography, yielding a white solid with exceptional purity suitable for analytical applications. For detailed operational parameters and safety precautions, please refer to the standardized guide below.
- Perform acid-catalyzed dehydration of 1-[2-amino-1-(4-methoxyphenyl)ethyl]cyclohexanol using sulfuric acid or p-toluenesulfonic acid in solvents like dichloromethane or toluene to form Intermediate II.
- Subject Intermediate II to a cyclization reaction using 37% formaldehyde and 88% formic acid under reflux conditions (80-110°C) to close the ring structure.
- Purify the final crude product via column chromatography using ethyl acetate and n-hexane to achieve purity levels exceeding 99%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the implementation of this synthesis method represents a significant opportunity to stabilize the supply of critical quality control materials. The reliance on external vendors for obscure impurity standards often leads to long lead times and exorbitant costs, creating bottlenecks in the drug approval and release processes. By adopting this in-house or contract-manufacturing friendly route, organizations can drastically reduce their dependency on single-source suppliers. The raw materials required, such as formaldehyde, formic acid, and the amino-alcohol precursor, are commodity chemicals available from multiple global suppliers. This abundance ensures that production schedules are not held hostage by the scarcity of exotic reagents, thereby enhancing the overall resilience of the supply chain against market fluctuations and geopolitical disruptions.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the simplicity of the reaction conditions and the high yield of the final product. Unlike complex multi-step syntheses that require expensive transition metal catalysts or cryogenic conditions, this method operates at atmospheric pressure and moderate temperatures. The elimination of precious metal catalysts removes the need for costly scavenging steps to meet residual metal specifications, which is a common expense in pharmaceutical manufacturing. Furthermore, the high selectivity of the reaction minimizes the generation of waste streams, reducing the burden on wastewater treatment facilities and lowering the overall cost of goods sold. The ability to produce the impurity standard in high yield means that less starting material is wasted, directly contributing to substantial cost savings in the long term.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount for pharmaceutical companies facing strict regulatory deadlines. The robustness of this synthetic route ensures consistent batch-to-batch reproducibility, a key factor in maintaining a reliable inventory of reference standards. Because the process does not rely on proprietary enzymes or biologically derived catalysts that may suffer from supply variability, the risk of production stoppages is significantly mitigated. The use of standard chemical engineering unit operations, such as reflux and liquid-liquid extraction, means that the process can be easily transferred between different manufacturing sites or contract development and manufacturing organizations (CDMOs) without extensive re-validation. This flexibility allows procurement teams to diversify their manufacturing base, ensuring that the supply of critical QC materials remains uninterrupted even if one facility faces operational challenges.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process aligns well with modern green chemistry principles. The solvents used, such as ethyl acetate and ethanol, are relatively benign and can be recovered and recycled efficiently, minimizing the environmental footprint of the operation. The absence of heavy metals and hazardous reagents simplifies the disposal of chemical waste, ensuring compliance with increasingly stringent environmental regulations. Scalability is further supported by the fact that the reaction kinetics are well-understood and do not exhibit dangerous exotherms that are difficult to manage on a large scale. This makes the transition from laboratory gram-scale synthesis to kilogram or ton-scale commercial production straightforward, allowing companies to build strategic stockpiles of reference materials to buffer against future demand spikes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of Venlafaxine Related Substance I. These insights are derived directly from the experimental data and claims presented in patent CN112094230A, providing a factual basis for decision-making. Understanding the nuances of this synthesis helps stakeholders evaluate its fit within their existing quality control frameworks and manufacturing capabilities. The answers below reflect the specific conditions and outcomes reported in the patent documentation.
Q: What is the primary advantage of the synthesis method in CN112094230A?
A: The primary advantage is the ability to synthesize Venlafaxine Related Substance I, a specific impurity not covered by standard pharmacopoeias, with high purity (>95%) and yield, filling a critical gap for reference standard manufacturing.
Q: What are the key reaction conditions for the cyclization step?
A: The cyclization step utilizes formaldehyde (37%) and formic acid (88%) as reagents, typically conducted under reflux conditions between 80-110°C, often in aqueous or alcoholic solvents.
Q: Is this process suitable for large-scale production of reference standards?
A: Yes, the patent highlights mild reaction conditions, easily obtainable raw materials, and simple post-treatment separation, making it highly scalable for the commercial production of impurity reference materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Venlafaxine Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality impurity standards play in ensuring the safety and efficacy of pharmaceutical products. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and purity of every batch. Whether you require custom synthesis of Venlafaxine Related Substance I or other complex pharmaceutical intermediates, our facility is designed to deliver results that meet the highest international regulatory standards.
We invite you to collaborate with us to optimize your supply chain and reduce your operational costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your R&D and quality control objectives. Let us be your partner in navigating the complexities of pharmaceutical manufacturing with confidence and reliability.
