Advanced 4-Step Synthesis of Venlafaxine Hydrochloride for Commercial Scale-Up
Advanced 4-Step Synthesis of Venlafaxine Hydrochloride for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust, scalable, and economically viable pathways for the production of critical antidepressant medications. A significant breakthrough in this domain is detailed in patent CN115650863A, which discloses a highly efficient method for preparing venlafaxine hydrochloride. This intellectual property outlines a strategic departure from traditional, hazardous synthesis routes, offering a streamlined four-step process that begins with readily available raw materials such as anisole and pyruvate. By leveraging a sequence involving Friedel-Crafts alkylation, acid-catalyzed dehydration, amine addition, and a sophisticated Grignard cyclization, this technology addresses long-standing challenges regarding yield, safety, and impurity profiles. For global procurement leaders and R&D directors, understanding the nuances of this patent is essential for securing a reliable venlafaxine hydrochloride supplier capable of delivering high-purity active pharmaceutical ingredients (APIs) with enhanced supply chain continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of venlafaxine hydrochloride has been plagued by significant technical and economic inefficiencies. Traditional strategies often rely on the condensation of p-methoxyphenylacetonitrile with cyclohexanone, a route that typically suffers from a low total yield of approximately 38%. Furthermore, these legacy processes frequently necessitate the use of n-butyl lithium at cryogenic temperatures (-78°C), creating severe operational hazards and demanding expensive specialized equipment. Another common approach involves the hydrogenation reduction of cyano groups, which requires high-pressure hydrogenation kettles. This not only escalates capital expenditure (CAPEX) due to the need for pressure-rated reactors but also introduces substantial safety risks associated with handling high-pressure hydrogen gas. Additionally, alternative multi-step routes starting from anisole and chloroacetyl chloride have been documented to have extremely low yields around 11%, generating excessive impurities and making them unsuitable for cost reduction in API manufacturing on a commercial scale.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the technology described in patent CN115650863A introduces a concise and high-yielding synthetic strategy. This novel approach utilizes anisole and pyruvate esters as foundational building blocks, executing a Friedel-Crafts reaction to establish the core carbon skeleton efficiently. The process flows seamlessly through dehydration and dimethylamine addition before culminating in a key Grignard reaction that constructs the cyclohexanol ring system. This optimized pathway boasts a total molar yield reaching up to 67%, nearly doubling the efficiency of standard nitrile-based routes. By eliminating high-pressure hydrogenation and cryogenic steps, the process significantly simplifies the engineering requirements for commercial scale-up of complex pharmaceutical intermediates. The result is a greener, safer, and more economically attractive manufacturing protocol that aligns perfectly with modern sustainable chemistry goals.
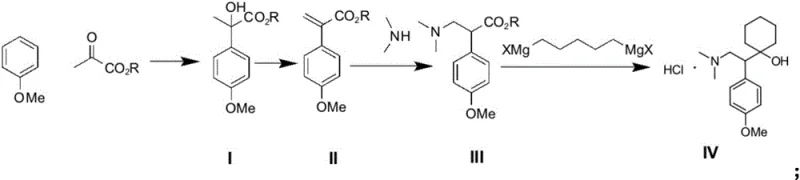
Mechanistic Insights into Friedel-Crafts Alkylation and Grignard Cyclization
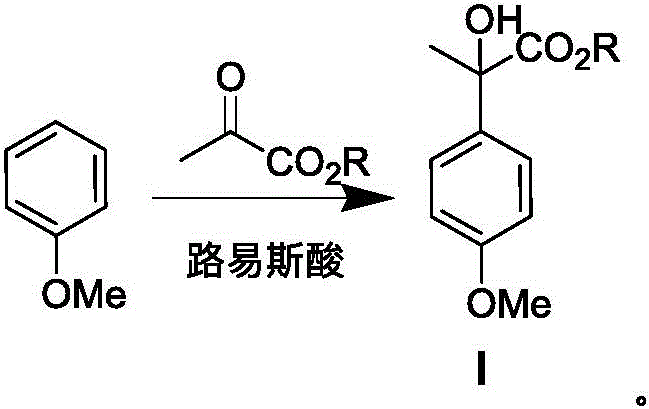
The cornerstone of this synthesis lies in the initial Friedel-Crafts reaction, where anisole reacts with a pyruvate ester in the presence of a Lewis acid catalyst such as aluminum trichloride. This electrophilic aromatic substitution is meticulously controlled at temperatures between 0°C and 5°C to ensure regioselectivity and minimize side reactions. The choice of solvent, specifically 1,2-dichloroethane or dichloromethane, plays a critical role in solubilizing the Lewis acid complex and stabilizing the intermediate carbocation, thereby driving the reaction toward the formation of the aryl lactate (Compound I) with high fidelity. Following this, an acid-catalyzed dehydration step converts the lactate into an aryl acrylate (Compound II), setting the stage for a Michael-type addition with dimethylamine. This sequence effectively installs the necessary amine functionality while preserving the ester group for the subsequent cyclization event.
The final and most chemically intricate transformation involves the reaction of the dimethylamine ester (Compound III) with a bis-Grignard reagent derived from 1,5-dihalopentane. This step is pivotal for constructing the cyclohexane ring fused to the ethanolamine side chain. The use of a double Grignard reagent allows for a tandem addition-cyclization sequence that efficiently closes the ring to form the tertiary alcohol center characteristic of venlafaxine. Conducted in solvents like methyltetrahydrofuran at mild temperatures (0-10°C), this reaction avoids the harsh conditions associated with earlier methods. The mechanistic precision here ensures that impurity generation is minimized, leading to a crude product that is easily purified to exceed 99% purity specifications. This level of control over the reaction trajectory is vital for maintaining a clean impurity profile, a key concern for regulatory compliance in high-purity API production.
How to Synthesize Venlafaxine Hydrochloride Efficiently
Implementing this synthesis requires strict adherence to the optimized conditions outlined in the patent to maximize yield and safety. The process begins with the careful preparation of the Lewis acid complex followed by the controlled addition of anisole. Subsequent steps involve precise temperature management during dehydration and amine addition to prevent polymerization or degradation. The final Grignard step demands anhydrous conditions and careful quenching protocols to isolate the hydrochloride salt effectively. While the chemical transformations are robust, the operational details regarding solvent recovery and recrystallization are equally important for achieving the reported commercial viability. For a comprehensive breakdown of the specific operational parameters, reagents, and workup procedures required to execute this synthesis successfully, please refer to the standardized guide below.
- Perform a Friedel-Crafts reaction between anisole and pyruvate ester using a Lewis acid catalyst to obtain aryl lactate (Compound I).
- Dehydrate Compound I under acid catalysis to form the corresponding aryl acrylate (Compound II).
- Conduct an addition reaction between Compound II and dimethylamine to synthesize the dimethylamine ester intermediate (Compound III).
- Execute a Grignard reaction using Compound III and a bis-Grignard reagent derived from 1,5-dihalopentane to cyclize and form venlafaxine hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic benefits beyond mere chemical elegance. The shift away from high-pressure hydrogenation and cryogenic reagents translates directly into reduced operational complexity and lower infrastructure costs. Facilities do not need to invest in specialized high-pressure vessels or maintain extensive safety protocols for handling bulk hydrogen, which drastically simplifies the manufacturing footprint. Furthermore, the use of commodity chemicals like anisole and pyruvate esters ensures a stable and diversified raw material supply base, mitigating the risk of shortages that often plague specialty precursor markets. This stability is crucial for maintaining consistent lead times for high-purity pharmaceutical intermediates in a volatile global market.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as n-butyl lithium and high-pressure hydrogen significantly lowers the variable cost of production. By increasing the overall molar yield from roughly 38% to 67%, the amount of raw material required per kilogram of final API is nearly halved, leading to substantial cost savings. Additionally, the reduced number of purification steps and the ability to use common solvents like toluene and dichloroethane further decrease waste disposal costs and solvent procurement expenses, enhancing the overall economic efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: Relying on widely available starting materials like anisole reduces dependency on single-source suppliers for exotic intermediates. The robustness of the reaction conditions, which operate at near-ambient temperatures for most steps, minimizes the risk of batch failures due to equipment malfunction or temperature excursions. This reliability ensures a steady flow of product, allowing supply chain heads to plan inventory levels with greater confidence and reduce the need for excessive safety stock, thereby optimizing working capital.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, avoiding unit operations that are difficult to scale, such as reactions at -78°C. The milder conditions and higher atom economy contribute to a reduced environmental footprint, aligning with increasingly stringent global regulations on pharmaceutical manufacturing emissions. The simplified waste stream, devoid of heavy metal catalysts or high-pressure gas residues, facilitates easier treatment and disposal, ensuring long-term environmental compliance and sustainability for large-scale production facilities.
Frequently Asked Questions (FAQ)
Understanding the technical specifics of this synthesis is critical for stakeholders evaluating its potential for integration into their supply chains. The following questions address common inquiries regarding yield optimization, safety protocols, and purity standards associated with this patented method. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making.
Q: How does the yield of this new synthesis method compare to conventional routes?
A: According to patent CN115650863A, this novel 4-step route achieves a total molar yield of approximately 67%, which is significantly higher than the 38% yield reported for traditional methods involving p-methoxyphenylacetonitrile and cyclohexanone.
Q: What are the primary safety advantages of this manufacturing process?
A: This method eliminates the need for high-pressure hydrogenation of cyano groups, a hazardous step common in older synthesis routes. Instead, it utilizes milder Friedel-Crafts and Grignard reactions, substantially reducing operational risks and equipment requirements.
Q: What purity levels can be achieved with this synthetic pathway?
A: The patent data indicates that the final venlafaxine hydrochloride product obtained through this process consistently exceeds 99% purity by weight, meeting stringent pharmaceutical standards without requiring complex purification sequences.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Venlafaxine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in patent CN115650863A for the global antidepressant market. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory methodology into robust commercial reality. Our facilities are equipped to handle diverse synthetic pathways, ranging from early-stage process development to full-scale production. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest international pharmacopeial standards.
We invite you to collaborate with us to leverage this advanced synthesis for your venlafaxine hydrochloride requirements. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how this efficient route can optimize your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to secure a sustainable, high-quality supply of this critical API, driving value and reliability throughout your pharmaceutical supply chain.
