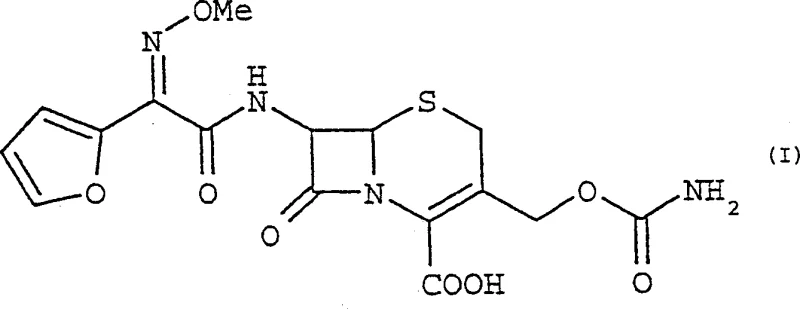
Advanced Carbamylation Process for Cefuroxime Acid: Enhancing Safety and Scalability in Beta-Lactam Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high yield with operational safety, particularly for critical antibiotic intermediates like Cefuroxime acid. Patent CN1364169A introduces a transformative approach to the synthesis of beta-lactam derivatives, specifically focusing on the carbamylation of 3-hydroxymethylceph-3-ene precursors. This innovation replaces hazardous traditional solvents with safer C1-C4 alkyl carbonates, such as dimethyl carbonate and diethyl carbonate, fundamentally altering the risk profile of the manufacturing process. By utilizing these green solvents in conjunction with activated isocyanates, the process achieves exceptional conversion rates while mitigating the flammability and toxicity issues associated with legacy methods involving acetone or tetrahydrofuran. This technical breakthrough offers a compelling value proposition for manufacturers aiming to modernize their production lines for second-generation cephalosporins.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefuroxime acid and its derivatives has relied heavily on solvents that pose significant safety and environmental challenges during large-scale production. Prior art methods, such as those disclosed in US 3,966,717 and US 4,284,767, typically employ solvents like acetone, tetrahydrofuran (THF), or alkyl acetates for the critical carbamylation step. These solvents are characterized by extremely low flash points; for instance, acetone has a flash point of -18.0°C and THF is even lower at -17.2°C, creating a substantial fire hazard in industrial settings where static electricity or heat sources are present. Furthermore, the toxicity profile of these conventional solvents is concerning, with THF exhibiting an oral LD50 in mice of only 2816 mg/kg, necessitating rigorous containment and ventilation systems that drive up capital and operational expenditures. The reliance on such volatile organic compounds also complicates waste management and regulatory compliance, adding layers of complexity to the supply chain.
The Novel Approach
In stark contrast, the methodology outlined in patent CN1364169A leverages the unique physicochemical properties of alkyl carbonates to create a inherently safer reaction environment. By switching to solvents like dimethyl carbonate (flash point 21.7°C) or diethyl carbonate (flash point 33.0°C), the process moves the operation well above ambient temperature thresholds, effectively eliminating the risk of flash fires under normal processing conditions. Beyond safety, the toxicity data presents a dramatic improvement, with dimethyl carbonate showing an oral LD50 of 13000 mg/kg, which is nearly five times less toxic than THF. This shift not only protects personnel and facilities but also simplifies the regulatory burden associated with handling hazardous materials. The patent demonstrates that these greener solvents do not compromise reaction efficiency; rather, they support high-yield transformations comparable to, or exceeding, those of traditional methods, proving that safety and performance are not mutually exclusive in fine chemical synthesis.
Mechanistic Insights into Carbonate-Mediated Carbamylation
The core of this innovative process lies in the nucleophilic substitution reaction where the hydroxyl group of the 3-hydroxymethylceph-3-ene precursor attacks the electrophilic carbon of an activated isocyanate. In the presence of a carbonate solvent, which acts as a polar aprotic medium, the reactivity of the isocyanate species—preferably chlorosulfonyl isocyanate—is finely tuned to favor the formation of the carbamate linkage without degrading the sensitive beta-lactam ring. The carbonate solvent stabilizes the transition state through dipole interactions, facilitating the attack of the alcohol on the isocyanate while preventing side reactions that often plague cephalosporin chemistry, such as beta-lactam ring opening or epimerization at the chiral centers. This mechanistic stability is crucial for maintaining the stereochemical integrity of the (6R, 7R) configuration, which is essential for the biological activity of the final antibiotic product.
Impurity control is another critical aspect managed by this solvent system. Traditional solvents like acetone can sometimes participate in unwanted condensation reactions or fail to solubilize specific intermediates effectively, leading to heterogeneous mixtures that promote degradation. The use of dimethyl or diethyl carbonate ensures a homogeneous reaction phase or a manageable suspension that allows for precise temperature control, typically maintained between 0°C and 10°C. This thermal regulation is vital for suppressing the formation of regioisomers or polymeric byproducts. The patent data confirms that monitoring the reaction via HPLC shows a rapid decline in substrate concentration to less than 2% within 15 to 60 minutes, indicating a clean and fast kinetic profile. The subsequent quenching with aqueous acid allows for the selective hydrolysis of any excess activated species while precipitating the pure acid product, resulting in a crystalline powder with HPLC purity exceeding 95%.
How to Synthesize Cefuroxime Acid Efficiently
To implement this advanced synthetic route, manufacturers must adhere to specific operational parameters regarding reagent addition and thermal management to maximize yield and safety. The process begins with the preparation of a precursor solution in the chosen carbonate solvent, followed by the controlled addition of the activating agent under inert conditions. Detailed standard operating procedures regarding stoichiometry, addition rates, and work-up protocols are essential for reproducibility on a commercial scale. For the complete standardized synthesis steps and technical specifications, please refer to the guide below.
- Prepare a solution or suspension of the 3-hydroxymethylceph-3-ene precursor in a C1-C4 alkyl carbonate solvent, such as dimethyl carbonate or diethyl carbonate, maintaining a concentration between 1-20% by weight.
- Under an inert atmosphere and at controlled low temperatures (preferably 0-10°C), add an activated isocyanate, such as chlorosulfonyl isocyanate, to the precursor solution at a molar ratio of 1: 1 to 5:1.
- Monitor the reaction progress via HPLC until substrate conversion is complete, then quench the mixture with water or acidic aqueous solution to recover the high-purity Cefuroxime acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the carbonate-based synthesis route described in CN1364169A offers tangible strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic reduction of operational risk associated with solvent handling. By eliminating highly flammable solvents like acetone and THF, facilities can significantly lower their insurance premiums and reduce the costs associated with explosion-proof infrastructure and specialized fire suppression systems. This inherent safety feature translates directly into cost reduction in antibiotic intermediate manufacturing, as the overhead for safety compliance and hazard mitigation is substantially decreased. Furthermore, the lower toxicity of carbonates simplifies waste disposal protocols, reducing the fees paid to third-party hazardous waste treatment vendors and minimizing the environmental footprint of the production site.
- Cost Reduction in Manufacturing: The switch to carbonate solvents eliminates the need for expensive and energy-intensive solvent recovery systems required for low-boiling, hazardous volatiles. Since dimethyl carbonate and diethyl carbonate are less volatile and more stable, solvent loss during processing is minimized, leading to substantial cost savings on raw material replenishment. Additionally, the high reaction yields reported in the patent, reaching up to 95% in optimized examples, ensure that the consumption of the valuable cephalosporin precursor is maximized, reducing the cost per kilogram of the final active pharmaceutical ingredient. The process efficiency also means shorter batch cycles, allowing for better utilization of reactor capacity and labor resources without compromising quality standards.
- Enhanced Supply Chain Reliability: Sourcing and transporting hazardous solvents often involve complex logistics and regulatory hurdles that can disrupt supply continuity. Carbonate solvents are generally easier to transport and store, classified with lower hazard ratings compared to Class B flammable liquids. This ease of logistics ensures a more reliable supply of raw materials, reducing the risk of production stoppages due to solvent shortages or transportation delays. Moreover, the robustness of the reaction conditions, which tolerate a range of temperatures and concentrations, makes the process less sensitive to minor variations in raw material quality, further stabilizing the supply chain against upstream fluctuations.
- Scalability and Environmental Compliance: Scaling up chemical processes often amplifies safety risks, but the carbonate-mediated pathway mitigates this through its high flash point and low toxicity profile. This makes the technology ideal for commercial scale-up of complex pharmaceutical intermediates, as the safety margins remain wide even at multi-ton scales. From an environmental perspective, the use of greener solvents aligns with increasingly stringent global regulations on volatile organic compound (VOC) emissions. Adopting this process positions the manufacturer as a leader in sustainable chemistry, facilitating easier permitting for plant expansions and enhancing the corporate brand image among eco-conscious partners and stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carbonate-based synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN1364169A, providing a factual basis for decision-making. Understanding these details is crucial for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing the long-term viability of the supply source.
Q: What are the primary safety advantages of using carbonate solvents in Cefuroxime synthesis?
A: According to patent CN1364169A, replacing traditional solvents like acetone or THF with dimethyl carbonate or diethyl carbonate significantly reduces fire hazards due to much higher flash points (e.g., 21.7°C for DMC vs -18°C for acetone) and drastically lowers toxicity profiles (oral LD50 in mice increases from ~2800 mg/kg for THF to 13000 mg/kg for DMC).
Q: Which activated isocyanates are suitable for this carbamylation process?
A: The patent specifies that various activated isocyanates can be used, but chlorosulfonyl isocyanate is particularly preferred for achieving high yields (up to 95%) and excellent purity when reacted with the 3-hydroxymethyl precursor in carbonate media.
Q: What purity levels can be achieved with this novel synthetic route?
A: Experimental data within the patent demonstrates that the recovered Cefuroxime acid exhibits high HPLC purity greater than 95%, with structural confirmation via 1H-NMR, 13C-NMR, and mass spectrometry matching literature standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefuroxime Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer and more efficient synthetic routes for high-value antibiotics like Cefuroxime acid. Our technical team has extensively analyzed the carbonate-mediated carbamylation process and possesses the expertise to implement this technology at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Cefuroxime acid meets the highest pharmacopeial standards required by global regulatory bodies.
We invite potential partners to engage with our technical procurement team to discuss how this innovative process can be tailored to your specific supply needs. By collaborating with us, you can leverage our engineering capabilities to achieve a Customized Cost-Saving Analysis that quantifies the benefits of switching to this greener solvent system. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of safety, quality, and economic efficiency.
