Advanced Synthesis of High-Purity Cefuroxime Acid for Global Pharmaceutical Manufacturing
Advanced Synthesis of High-Purity Cefuroxime Acid for Global Pharmaceutical Manufacturing
The global demand for second-generation cephalosporin antibiotics continues to drive the need for robust, scalable, and high-purity intermediate synthesis routes. Patent CN102134252B introduces a transformative preparation method for cefuroxime acid, a critical precursor for cefuroxime sodium and cefuroxime axetil. This technology addresses long-standing challenges in impurity control, specifically targeting the persistent lactone by-products that compromise product stability and color in conventional manufacturing. By leveraging a sophisticated ternary solvent extraction system combined with precise temperature-controlled nucleophilic additions, this process achieves a final product purity of greater than 99% with yields exceeding 91%. For R&D directors and procurement strategists, understanding the mechanistic advantages of this route is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent pharmacopoeial standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for cefuroxime acid, typically starting from 7-aminocephalosporanic acid (7-ACA), have historically struggled with significant impurity profiles that degrade final drug quality. In standard processes, the intermediate 3-deformamido cefuroxime acid (DCC) is generated, but subsequent steps often fail to adequately remove side-reaction products. Specifically, the formation of a lactone by-product is a pervasive issue, with liquid chromatography detecting levels between 3.2% and 3.8% in crude outputs. Conventional aqueous phase crystallization is fundamentally inefficient at separating these organic unsaponifiable impurities from the target molecule. Furthermore, the resulting filter cakes from aqueous crystallization retain excessive moisture, often surpassing 50% water content, which necessitates prolonged drying at elevated temperatures. This thermal stress not only increases energy consumption but also induces degradation, leading to products with deep coloration, reduced purity (often around 90.96%), and compromised shelf-life stability.
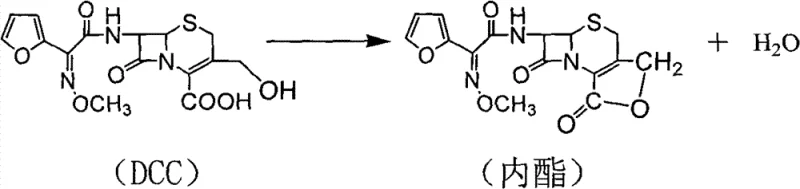
The Novel Approach
The patented methodology overcomes these deficiencies through a strategic shift from simple aqueous crystallization to a multi-stage liquid-liquid extraction protocol utilizing a tailored ternary solvent system. Instead of attempting to crystallize the product directly from an impure aqueous solution, the process first converts the cefuroxime acid into its water-soluble sodium salt using sodium bicarbonate. This salification step allows for the selective partitioning of impurities. By introducing a specific mixture of dichloromethane, ethyl acetate, and tetrahydrofuran, the process effectively dissolves and removes the troublesome lactone by-products and other organic unsaponifiables into the organic phase, leaving the desired sodium salt in the aqueous layer. Following this purification, the aqueous phase is re-acidified, and the pure cefuroxime acid is extracted back into the ternary solvent system. This "wash-and-recover" strategy ensures that water-soluble impurities are left behind in the aqueous waste stream while the organic phase contains highly purified product ready for crystallization.
Mechanistic Insights into N-Acylation and Ternary Extraction
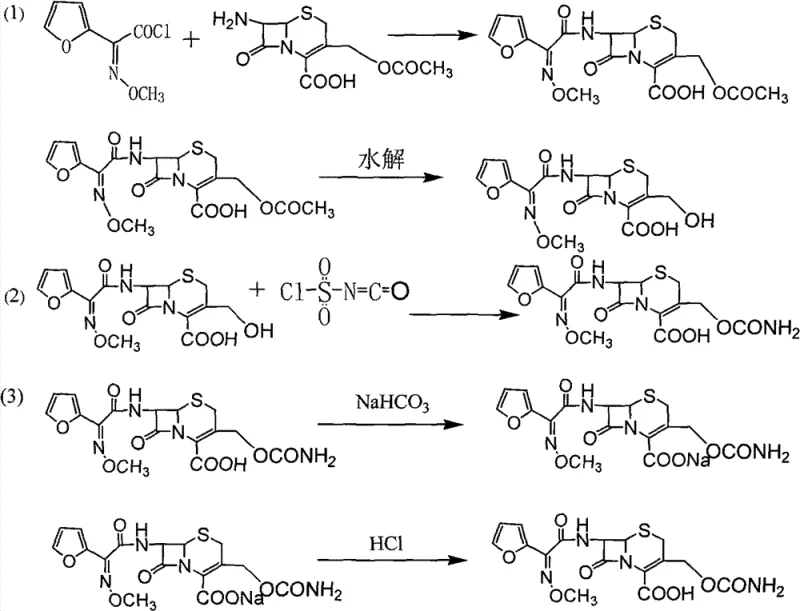
The core of this synthesis relies on precise control over the nucleophilic addition reactions and the physicochemical properties of the extraction solvents. The initial formation of the DCC intermediate involves an N-acylation at the 7-position of the 7-ACA nucleus using furoyl acetyl chloride, followed by a controlled hydrolysis of the 3-acetyl group using sodium hydroxide at low temperatures ranging from -20°C to -60°C. This低温 environment is critical to prevent the premature opening of the beta-lactam ring. Subsequently, the DCC intermediate undergoes a nucleophilic addition with chlorosulfonyl isocyanate (CSI) in tetrahydrofuran. The reaction conditions are extremely sensitive, requiring temperatures between -30°C and -80°C during the dropwise addition of CSI to manage the exotherm and prevent side reactions. The resulting chlorosulfonyl cefuroxime acid is then hydrolyzed to yield the free acid, which serves as the substrate for the novel purification sequence.
The purification mechanism exploits the differential solubility of the ionized versus neutral forms of the molecule. By adjusting the pH to between 5.0 and 8.0 using sodium bicarbonate, the cefuroxime acid is converted to its sodium salt, which is highly soluble in water but insoluble in the organic ternary mixture. Conversely, the lactone impurity and other non-polar by-products remain soluble in the organic phase (dichloromethane/ethyl acetate/THF). This allows for a clean separation where the organic layer, containing the impurities, is discarded. Upon re-acidification of the aqueous phase to a pH of 1.0 to 4.0 using hydrochloric acid, the cefuroxime acid reverts to its neutral, lipophilic form. A second extraction with the ternary solvent now pulls the pure product into the organic phase, leaving inorganic salts and water-soluble contaminants in the aqueous raffinate. This dual-extraction logic is the key to achieving the reported purity of ≥99%.
How to Synthesize Cefuroxime Acid Efficiently
Implementing this synthesis route requires strict adherence to the stoichiometric ratios and temperature profiles outlined in the patent data to ensure reproducibility and safety. The process begins with the preparation of the DCC intermediate, followed by the critical CSI reaction and the multi-step extraction workup. Operators must pay close attention to the volume ratios of the ternary solvent system, specifically maintaining a balance where tetrahydrofuran constitutes 20-50%, dichloromethane 20-60%, and ethyl acetate 10-30% of the mixture to optimize phase separation and impurity removal. The detailed standardized operating procedures for scaling this reaction from laboratory to commercial production are provided below.
- Perform N-acylation of 7-ACA with furoyl acetyl chloride followed by low-temperature hydrolysis to obtain the intermediate DCC.
- React DCC with chlorosulfonyl isocyanate (CSI) in tetrahydrofuran at -30°C to -80°C, followed by hydrolysis to form the cefuroxime acid solution.
- Purify via salification with sodium bicarbonate, extract impurities using a dichloromethane/ethyl acetate/THF ternary solvent, acidify, and recrystallize.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers substantial strategic benefits beyond mere technical superiority. The ability to consistently produce cefuroxime acid with purity levels exceeding 99% significantly reduces the risk of batch rejection during downstream formulation, thereby enhancing overall supply chain reliability. Traditional methods that yield products with high moisture content and unstable impurity profiles often lead to unpredictable shelf-life issues, forcing manufacturers to hold larger safety stocks or face costly recalls. By eliminating the lactone impurity at the source through effective extraction, this method ensures a stable, white crystalline powder that meets rigorous quality specifications without the need for extensive re-processing or remediation.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven by the elimination of inefficient purification steps and the optimization of solvent usage. Unlike conventional methods that may require multiple recrystallizations to approach acceptable purity levels, this single-pass ternary extraction strategy streamlines the workflow, reducing labor hours and utility consumption. Furthermore, the patent explicitly notes that the solvents used in the ternary system can be recycled and reused, which drastically lowers the raw material costs associated with solvent procurement and waste disposal. The reduction in drying time and temperature also translates to direct energy savings, contributing to a lower overall cost of goods sold (COGS) for the intermediate.
- Enhanced Supply Chain Reliability: The robustness of the synthesis pathway ensures a consistent supply of high-quality intermediates, which is critical for maintaining uninterrupted production of finished cephalosporin antibiotics. The process utilizes readily available starting materials such as 7-ACA and common industrial solvents like tetrahydrofuran and ethyl acetate, mitigating the risk of raw material shortages. Additionally, the high yield reported (greater than 91%) means that less starting material is required to produce the same amount of final product, improving material efficiency and reducing the strain on upstream supply chains. This efficiency allows suppliers to offer more competitive lead times and maintain steady inventory levels even during periods of high market demand.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns well with modern green chemistry principles by minimizing waste generation and maximizing solvent recovery. The ability to recycle the ternary solvent mixture reduces the volume of hazardous waste requiring treatment, simplifying compliance with environmental regulations. The process is designed for scalability, with clear parameters for temperature and pH control that can be easily translated from pilot plant to multi-ton commercial reactors. This scalability ensures that the technology can support large-volume contracts without compromising on the purity or stability of the cefuroxime acid, making it an ideal solution for long-term supply agreements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity synthesis route. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process capabilities and quality outcomes. Understanding these details is vital for technical teams evaluating the feasibility of integrating this intermediate into their existing manufacturing workflows.
Q: How does the ternary solvent system improve cefuroxime acid purity?
A: The patented process utilizes a specific ratio of tetrahydrofuran, dichloromethane, and ethyl acetate to selectively extract lactone by-products and unsaponifiable impurities before the final crystallization, achieving purity levels exceeding 99%.
Q: What are the critical temperature controls during the CSI reaction step?
A: To ensure optimal nucleophilic addition and prevent degradation, the reaction between the DCC intermediate and chlorosulfonyl isocyanate must be maintained strictly between -30°C and -80°C during the dropwise addition phase.
Q: Why is the removal of the lactone impurity critical for stability?
A: Conventional aqueous crystallization fails to remove lactone by-products (often 3.2-3.8%), which persist in the final product causing poor stability and discoloration; the new extraction method effectively eliminates these species.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefuroxime Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of your final antibiotic formulation depends entirely on the integrity of your starting intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated ternary extraction techniques described in patent CN102134252B can be executed with precision at an industrial scale. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch of cefuroxime acid meets the >99% purity threshold, free from the detrimental lactone impurities that plague inferior grades. Our commitment to quality assurance guarantees that you receive a stable, white crystalline product suitable for the most demanding pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for second-generation cephalosporins. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can enhance your production efficiency and reduce your total landed costs.
