Optimizing Nebivolol Production: Advanced Suzuki Coupling for High-Purity Intermediates
Optimizing Nebivolol Production: Advanced Suzuki Coupling for High-Purity Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for complex chiral molecules, particularly for cardiovascular therapeutics like Nebivolol. Patent CN103087028A discloses a sophisticated process for preparing d-Nebivolol and its enantiomer l-Nebivolol, addressing the significant challenge of managing four unsymmetrical carbon centers. This methodology diverges from traditional routes by leveraging a strategic combination of Grignard chemistry and intramolecular B-alkyl Suzuki coupling. By initiating the synthesis from commercially available 2,2-dimethyl-1,3-dioxolane-4-carbaldehyde, the process establishes a scalable foundation for generating the critical chroman core. The ability to control stereochemistry through diastereomeric separation at intermediate stages ensures the delivery of high-purity active pharmaceutical ingredients (APIs). This technical insight explores the mechanistic depth and commercial viability of this route for global supply chains.
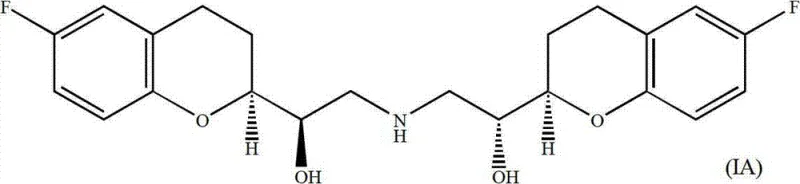
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha,alpha'-[imino-bis(methylene)]bis[6-fluoro-chroman-2-methanol] derivatives has presented substantial hurdles due to the generation of multiple stereoisomers. Prior art, such as European patent applications EP145067 and EP334429, typically involves the reduction of chroman-2-carboxylic acid to an aldehyde, followed by conversion into epoxide mixtures. These conventional pathways often suffer from low overall yields due to the necessity of separating racemic mixtures of (R,S) and (S,R) epoxides from (R,R) and (S,S) variants via column chromatography early in the sequence. Furthermore, the reliance on resolving racemic intermediates inherently limits the maximum theoretical yield to 50% unless dynamic kinetic resolution is employed, which adds complexity and cost. The presence of four stereocenters means that without precise control, up to 16 stereoisomers could theoretically form, complicating purification and increasing waste generation significantly.
The Novel Approach
The disclosed invention introduces a streamlined strategy that bypasses early-stage racemic resolutions by constructing the chiral framework from defined starting materials. Instead of reducing carboxylic acids, the process utilizes a vinyl Grignard addition to a chiral dioxolane aldehyde, immediately establishing side-chain functionality. Crucially, the formation of the chroman ring is achieved through an intramolecular Suzuki coupling of an organoborane intermediate. This approach allows for the generation of diastereomeric mixtures that are chemically distinct enough to be separated efficiently before the final dimerization step. By deferring the final stereochemical definition to later stages and utilizing robust carbon-carbon bond-forming reactions, the novel approach minimizes the loss of valuable chiral material and simplifies the impurity profile, offering a distinct advantage for cost reduction in cardiovascular API manufacturing.
Mechanistic Insights into B-Alkyl Suzuki Cyclization
The cornerstone of this synthetic route is the transformation of the linear ether intermediate into the cyclic chroman structure via a palladium-catalyzed B-alkyl Suzuki reaction. In this mechanism, the allylic side chain of the ether intermediate undergoes hydroboration using reagents such as 9-borabicyclo[3.3.1]nonane (9-BBN). This step installs a boron moiety at the terminal position of the side chain with high regioselectivity. Subsequently, in the presence of a palladium catalyst and base, the carbon-boron bond undergoes transmetallation with the aryl bromide moiety located on the adjacent phenol ring. This intramolecular cross-coupling closes the six-membered chroman ring efficiently. Unlike traditional acid-catalyzed cyclizations which might lead to rearrangement or polymerization, this metal-catalyzed pathway proceeds under mild alkaline conditions, preserving the integrity of the sensitive acetal protecting groups and the established stereocenters.
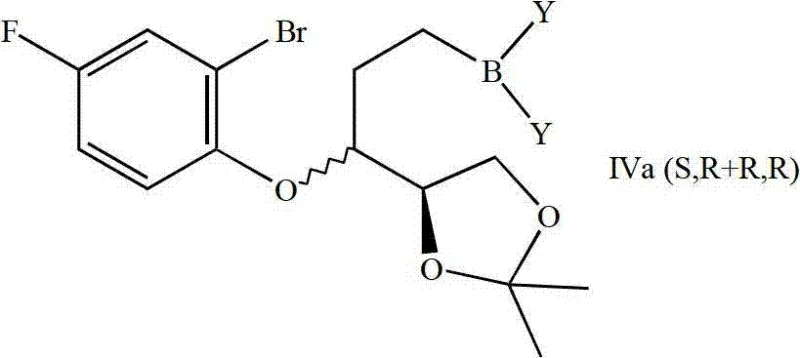
Following the cyclization, the resulting chroman intermediate exists as a mixture of diastereomers, specifically the (S,R+R,R) or (R,S+S,S) forms depending on the starting aldehyde enantiomer. The beauty of this mechanism lies in the physical property differences between these diastereomers, which facilitate their separation via standard chromatographic techniques. This separability is critical for impurity control, as it allows manufacturers to isolate the specific diastereomer required for d-NBV or l-NBV synthesis before proceeding to the final dimerization. The rigorous control over the cyclization step ensures that the final API meets stringent purity specifications required by regulatory bodies, minimizing the risk of genotoxic impurities or off-target stereoisomers that could compromise patient safety.
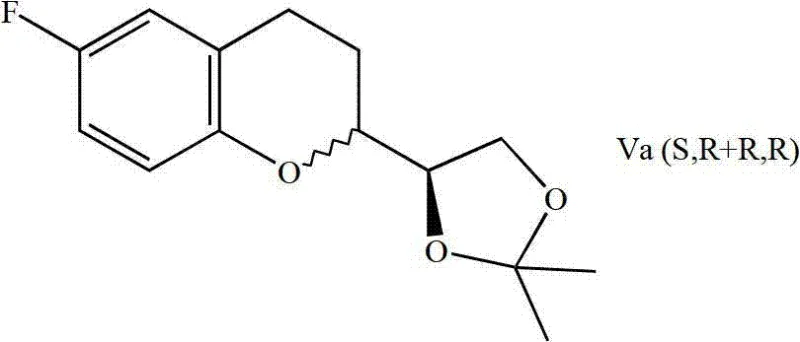
How to Synthesize Nebivolol Intermediates Efficiently
The synthesis begins with the reaction of 2,2-dimethyl-1,3-dioxolane-4-carbaldehyde with a vinyl Grignard reagent at controlled temperatures ranging from -20°C to 25°C. The resulting allylic alcohol is then coupled with 2-bromo-4-fluorophenol under Mitsunobu conditions to form the ether linkage. Following hydroboration and the key Suzuki cyclization, the acetal protecting group is hydrolyzed to reveal the diol, which is subsequently converted to an epoxide. Ring opening with benzylamine and final dimerization yields the protected Nebivolol precursor, which is deprotected to afford the final active compound. Detailed standardized synthetic steps see the guide below.
- React 2,2-dimethyl-1,3-dioxolane-4-carbaldehyde with vinyl Grignard reagent to form the allylic alcohol intermediate.
- Perform Mitsunobu reaction with 2-bromo-4-fluorophenol to establish the ether linkage.
- Execute hydroboration using organoborane compounds like 9-BBN followed by palladium-catalyzed intramolecular Suzuki cyclization to close the chroman ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain directors, the adoption of this synthetic route offers tangible benefits regarding raw material security and process robustness. The reliance on 2,2-dimethyl-1,3-dioxolane-4-carbaldehyde as a starting point is significant because it is a commercially available commodity chemical, reducing dependency on custom-synthesized chiral pools that often suffer from long lead times. Furthermore, the use of standard reagents like vinyl Grignard and palladium catalysts ensures that the process can be executed in multipurpose chemical plants without requiring specialized equipment for cryogenic reactions or high-pressure hydrogenation until the final deprotection stage. This flexibility enhances supply chain reliability by allowing for multi-vendor sourcing of key reagents and mitigating the risk of single-source bottlenecks.
- Cost Reduction in Manufacturing: The process eliminates the need for early-stage resolution of racemic epoxides, which traditionally results in a 50% yield loss of valuable intermediates. By utilizing a convergent strategy where diastereomers are separated after the ring-closing step, the overall material throughput is significantly improved. Additionally, the avoidance of expensive chiral catalysts in the initial steps and the use of recyclable palladium systems contribute to a lower cost of goods sold (COGS). The streamlined workflow reduces the number of unit operations, thereby lowering labor and utility costs associated with extended processing times.
- Enhanced Supply Chain Reliability: Starting from readily available aldehydes and phenols decouples the production schedule from the volatile supply of complex chiral building blocks. The robustness of the Grignard and Suzuki reactions means that batch failure rates are minimized, ensuring consistent output volumes. This stability is crucial for maintaining continuous supply agreements with downstream API manufacturers, as it reduces the likelihood of production delays caused by difficult-to-control exothermic reactions or sensitive enzymatic steps often found in alternative biocatalytic routes.
- Scalability and Environmental Compliance: The synthetic pathway is designed with commercial scale-up of complex pharmaceutical intermediates in mind. The reactions proceed in common solvents like THF, toluene, and DMF, which are easily recovered and recycled in large-scale facilities. The absence of heavy metal contaminants in the final product, thanks to efficient purification after the palladium step, simplifies the regulatory filing process. Moreover, the atom economy of the Suzuki coupling is superior to traditional substitution methods, resulting in less chemical waste and aligning with modern green chemistry principles demanded by environmental regulators.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Nebivolol synthesis process. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility for potential manufacturing partners. Understanding these nuances is essential for evaluating the technology transfer potential and risk assessment.
Q: How does this process control the four chiral centers in Nebivolol?
A: The process utilizes stereoselective starting materials and creates diastereomeric mixtures at key stages (such as the chroman formation) which can be separated via chromatography before final dimerization, ensuring high optical purity of the d-NBV or l-NBV enantiomers.
Q: What are the advantages of the B-alkyl Suzuki cyclization step?
A: This method allows for the mild formation of the chroman ring under alkaline conditions using palladium catalysis, avoiding harsh Lewis acids and providing better functional group tolerance compared to traditional Friedel-Crafts approaches.
Q: Is this synthetic route scalable for commercial production?
A: Yes, the route relies on robust, well-understood reactions like Grignard addition and Pd-catalyzed coupling, which are highly amenable to scale-up from kilogram to multi-ton production with standard industrial equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nebivolol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of stereochemical purity and process scalability in the production of beta-blocker intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We operate stringent purity specifications and maintain rigorous QC labs equipped with chiral HPLC and NMR capabilities to verify the stereochemical integrity of every batch. Our commitment to quality ensures that the Nebivolol intermediates supplied meet the exacting standards required for global regulatory submissions.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains. By leveraging our expertise in palladium-catalyzed couplings and chiral separations, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals for cardiovascular drug production.
