Advanced Synthesis of 6-Fluoro-3,4-Dihydro-2H-1-Benzopyran-2-Carboxylic Acid for Cardiovascular Drug Manufacturing
Advanced Synthesis of 6-Fluoro-3,4-Dihydro-2H-1-Benzopyran-2-Carboxylic Acid for Cardiovascular Drug Manufacturing
The pharmaceutical industry constantly seeks more efficient pathways for producing critical cardiovascular drug intermediates, and patent CN102250050B presents a significant breakthrough in this domain. This intellectual property details a robust synthetic methodology for 6-fluoro-3,4-dihydro-2H-1-benzopyran-2-carboxylic acid and its alkyl esters, which serve as pivotal precursors for nebivolol. Unlike traditional approaches that rely heavily on costly noble metal catalysts, this innovation leverages a streamlined three-step reaction sequence starting from gamma-butyrolactone. The process is characterized by mild reaction conditions, high atom economy, and the strategic avoidance of complex hydrogenation steps. For R&D directors and procurement specialists, this represents a tangible opportunity to optimize supply chains and reduce the overall cost of goods sold for beta-blocker manufacturing. The technical depth of this patent provides a clear roadmap for scaling production while maintaining stringent quality standards required for active pharmaceutical ingredient (API) synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-dihydro-2H-1-benzopyran-2-carboxylic acid derivatives has been fraught with economic and technical inefficiencies that burden large-scale manufacturing operations. Most reported literature describes a pathway that first constructs a 4-oxo-3,4-dihydro-2H-1-benzopyran-2-carboxylic acid scaffold, which subsequently requires a reduction step to reach the desired saturation level. This reduction is typically achieved using palladium on carbon (Pd/C) catalyzed hydrogenation, a process that introduces significant cost volatility due to the fluctuating price of palladium. Furthermore, alternative routes described in patents such as EP264586 involve the use of trimethylsilyl cyanide, a reagent that is not only expensive but also poses handling challenges due to its toxicity and moisture sensitivity. Other methods, like those in EP331078, utilize Fries rearrangement strategies which often suffer from inherently low yields and require harsh Lewis acid conditions that complicate downstream waste treatment. The cumulative effect of these legacy processes is a longer production timeline, higher impurity profiles requiring extensive purification, and a dependency on specialized hydrogenation equipment that limits flexibility in multi-purpose plants.

The Novel Approach
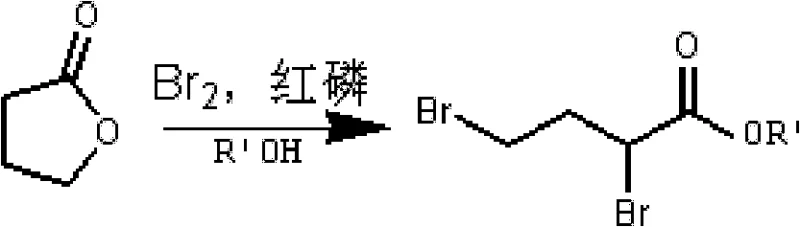
In stark contrast to these cumbersome legacy protocols, the methodology disclosed in CN102250050B offers a direct and economically superior alternative that bypasses the need for catalytic hydrogenation entirely. The novel approach initiates with the conversion of gamma-butyrolactone into a 2,4-dibromobutyric acid alkyl ester, a transformation that utilizes inexpensive bromine and red phosphorus. This intermediate then undergoes a selective etherification with p-fluorophenol to install the aromatic ring, followed by a decisive intramolecular cyclization. This final ring-closing step is catalyzed by a molten mixture of inorganic salts and Lewis acids, effectively constructing the chromene core in a single operation without generating the ketone intermediate that necessitates reduction in older routes. By eliminating the Pd/C step, the process removes the risk of heavy metal contamination, thereby simplifying the regulatory compliance burden for API manufacturers. The result is a shorter synthetic sequence with fewer unit operations, directly translating to reduced capital expenditure and operational complexity for facilities aiming to produce high-purity cardiovascular drug intermediates.
Mechanistic Insights into Lewis Acid Catalyzed Cyclization
The core chemical innovation of this patent lies in the final cyclization step, where a 4-bromo-2-(4-fluorophenoxy)butyrate derivative is converted into the target chromene structure under Lewis acid catalysis. This transformation is facilitated by heating a mixture of inorganic salts, such as sodium chloride, and Lewis acids like aluminum trichloride to a molten state. In this high-temperature ionic environment, the Lewis acid activates the carbonyl group or facilitates the departure of the bromide leaving group, promoting an intramolecular Friedel-Crafts type alkylation. The presence of the inorganic salt serves to lower the melting point of the reaction medium, allowing the reaction to proceed at manageable temperatures between 80°C and 200°C while maintaining a homogeneous phase that ensures efficient heat transfer. This mechanism avoids the formation of the 4-oxo intermediate entirely, jumping straight to the saturated or partially saturated ring system depending on the specific workup, although the patent indicates the direct formation of the dihydro-chromene acid. The selectivity of this cyclization is crucial, as it minimizes the formation of regioisomers or polymerization byproducts that often plague acid-catalyzed aromatic substitutions. Understanding this mechanism allows process chemists to fine-tune the stoichiometry of the Lewis acid to maximize yield while minimizing the generation of aluminum-containing waste streams.
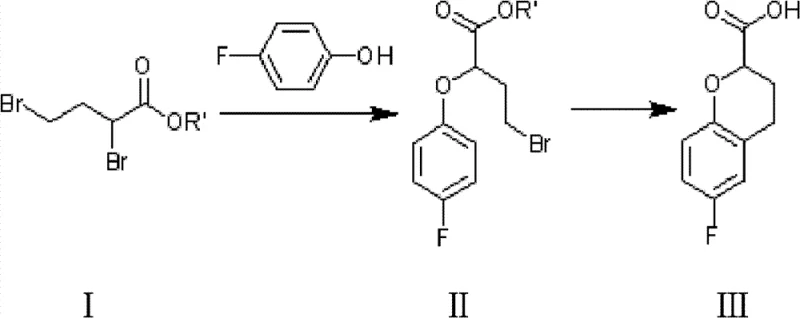
Preceding the cyclization, the synthesis of the linear precursor involves a highly controlled bromination and esterification sequence starting from gamma-butyrolactone. The reaction of gamma-butyrolactone with bromine in the presence of red phosphorus generates an alpha,beta-dibromo species which is immediately trapped by an alcohol to form the ester. This step is critical for setting up the correct carbon chain length and functionalization pattern required for the subsequent etherification. The use of red phosphorus acts as a catalyst to facilitate the bromination, ensuring that the reaction proceeds with high conversion rates without the need for radical initiators that could lead to uncontrolled side reactions. Following this, the nucleophilic substitution with p-fluorophenol is conducted under basic conditions, where the phenoxide ion attacks the less hindered terminal bromide of the dibromo ester. This regioselectivity is vital for ensuring that the phenoxy group is installed at the correct position (the 4-position of the butyrate chain) to enable the subsequent 6-endo-trig cyclization. The precise control over these early-stage intermediates ensures that the final cyclization step proceeds with high efficiency, resulting in a final product with a purity profile that meets the rigorous demands of pharmaceutical applications.
How to Synthesize 6-Fluoro-3,4-Dihydro-2H-1-Benzopyran-2-Carboxylic Acid Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly during the high-temperature cyclization phase where thermal control is paramount. The process begins with the preparation of the dibromo ester, followed by the etherification to link the aromatic ring, and concludes with the Lewis acid mediated ring closure. Each step has been optimized in the patent examples to demonstrate scalability, with specific attention paid to solvent selection and workup procedures that facilitate isolation. For process engineers, the ability to run the cyclization in a molten salt medium offers a unique advantage in terms of reaction kinetics, though it requires equipment capable of handling corrosive Lewis acids at elevated temperatures. The detailed experimental conditions provided in the patent serve as a foundational guide for developing a robust GMP manufacturing process. For the complete standardized operating procedure and specific stoichiometric ratios, please refer to the technical guide below.
- React gamma-butyrolactone with liquid bromine and red phosphorus, followed by esterification with alcohol to obtain 2,4-dibromobutyric acid alkyl ester.
- Perform nucleophilic substitution between 2,4-dibromobutyric acid alkyl ester and p-fluorophenol in the presence of a base to form 4-bromo-2-(4-fluorophenoxy)butyrate.
- Execute intramolecular cyclization using a molten mixture of inorganic salts and Lewis acids (e.g., AlCl3/NaCl) to yield the final chromene carboxylic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound benefits for procurement managers and supply chain directors focused on cost reduction in API manufacturing. The most significant economic driver is the complete elimination of palladium catalysts, which are subject to extreme market volatility and represent a substantial portion of raw material costs in traditional hydrogenation routes. By replacing this with abundant and inexpensive Lewis acids like aluminum chloride and common inorganic salts, the variable cost of production is drastically lowered. Furthermore, the removal of the hydrogenation step simplifies the facility requirements, as there is no longer a need for high-pressure hydrogen reactors or the specialized safety infrastructure associated with them. This reduction in capital intensity allows for more flexible manufacturing arrangements and lowers the barrier to entry for secondary suppliers, enhancing overall supply chain resilience. The use of commodity chemicals like gamma-butyrolactone and p-fluorophenol ensures that raw material sourcing is not a bottleneck, as these are produced globally at massive scales for various industrial applications.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the substitution of noble metal catalysts with base metal Lewis acids. In traditional methods, the cost of palladium on carbon and the subsequent metal scavenging resins required to meet residual metal specifications can be prohibitive. This new method circumvents those expenses entirely, leading to substantial cost savings per kilogram of the intermediate. Additionally, the higher yields reported in the patent examples for the cyclization step mean that less raw material is wasted, further improving the overall process mass intensity. The simplified purification workflow, which avoids complex chromatographic separations often needed to remove metal traces, reduces solvent consumption and waste disposal costs. These factors combine to create a leaner, more cost-effective manufacturing process that improves margin potential for the final API.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly enhanced by relying on a raw material portfolio that is diverse and globally available. Gamma-butyrolactone and p-fluorophenol are high-volume commodity chemicals with multiple qualified suppliers worldwide, reducing the risk of single-source dependency. In contrast, specialized reagents like trimethylsilyl cyanide or specific hydrogenation catalysts often have limited supply chains that can be disrupted by geopolitical or logistical issues. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, also means that production is less susceptible to minor fluctuations in utility availability. This reliability is critical for pharmaceutical companies that require guaranteed continuity of supply to meet regulatory filing commitments and market demand for cardiovascular medications. The ability to scale this process from pilot plant to commercial tonnage without changing the fundamental chemistry further de-risks the supply chain.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than conventional routes, aligning with modern green chemistry principles and regulatory expectations. By avoiding the use of toxic cyanide reagents and expensive heavy metals, the hazardous waste profile of the process is simplified, making wastewater treatment more straightforward and cost-effective. The molten salt cyclization method, while requiring energy for heating, operates efficiently and minimizes the volume of organic solvents needed compared to solution-phase reactions. This reduction in solvent usage directly correlates to lower VOC emissions and reduced fire hazards in the manufacturing plant. Scalability is inherent in the design, as the batch sizes for the bromination and etherification steps can be easily increased, and the cyclization can be adapted for continuous processing if desired. The combination of safety, environmental compliance, and ease of scale-up makes this technology an attractive asset for long-term production planning.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and comparative analysis provided in the patent documentation. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing networks. The answers reflect the practical realities of running this chemistry at scale, focusing on yield, purity, and operational safety.
Q: What is the primary advantage of this synthesis method over conventional routes?
A: The primary advantage is the elimination of the expensive Pd/C catalytic hydrogenation step required in traditional methods. This new route utilizes a Lewis acid catalyzed cyclization, significantly reducing catalyst costs and simplifying the purification process by avoiding heavy metal residues.
Q: What are the key starting materials for this process?
A: The process relies on readily available and cost-effective raw materials, specifically gamma-butyrolactone, liquid bromine, and p-fluorophenol. These commodities are widely accessible in the global chemical supply chain, ensuring stable production continuity.
Q: How is high purity achieved in the final product?
A: High purity is achieved through a combination of selective reaction conditions and effective purification techniques. The final ester product can be purified via recrystallization using solvents like n-hexane, consistently achieving purity levels exceeding 97% as demonstrated in patent examples.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Fluoro-3,4-Dihydro-2H-1-Benzopyran-2-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the competitive landscape of cardiovascular drug development. Our technical team has extensively analyzed the pathway described in CN102250050B and possesses the expertise to implement this advanced chemistry at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 6-fluoro-3,4-dihydro-2H-1-benzopyran-2-carboxylic acid meets the highest quality standards required for API synthesis. We are committed to being a reliable pharmaceutical intermediate supplier that adds value through technical excellence and operational reliability.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this non-hydrogenation method. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our material against your internal benchmarks. Let us collaborate to optimize your production costs and secure a sustainable supply of this critical nebivolol intermediate.
