Scaling High-Purity OBMF Production: A Breakthrough in Biomass-Derived Polyamide Precursors
Scaling High-Purity OBMF Production: A Breakthrough in Biomass-Derived Polyamide Precursors
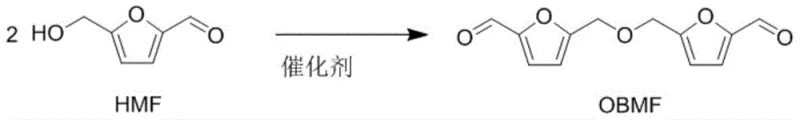
The global shift towards sustainable materials has intensified the search for bio-based alternatives to petroleum-derived monomers. Patent CN112300101B introduces a transformative methodology for synthesizing bis-(5-formyl furfuryl) ether (OBMF), a critical intermediate for high-performance polyamides and pharmaceutical agents. This technology leverages robust metal triflate catalysts to dehydrate 5-hydroxymethylfurfural (HMF), a readily available biomass derivative, achieving unprecedented yields and selectivity. For R&D directors and procurement strategists, this represents a pivotal opportunity to secure supply chains for advanced materials while adhering to green chemistry principles. The process eliminates the need for stringent water removal systems, significantly lowering operational complexity and capital expenditure associated with traditional etherification routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of OBMF has been plagued by inefficiencies that hinder commercial scalability. Traditional methods typically rely on homogeneous organic acids like p-toluenesulfonic acid (p-TSA) in solvents such as toluene. Even with the aid of Dean-Stark apparatuses to continuously remove water—a byproduct that inhibits equilibrium—the reported yields rarely exceed 58%. Furthermore, these acidic conditions often promote undesirable side reactions, including the alkylation of the solvent itself, which complicates downstream purification and compromises the purity profile required for electronic-grade polymers. Alternative routes, such as the Williamson ether synthesis involving 5-chloromethyl-2-furfural, suffer from even lower yields of approximately 42.7% and generate stoichiometric amounts of salt waste, creating significant environmental disposal burdens and inflating production costs.
The Novel Approach
The patented methodology overturns these limitations by employing metal triflates, such as Ytterbium(III) triflate [Yb(OTf)3], as Lewis acid catalysts. Unlike traditional Brønsted acids, these metal salts exhibit exceptional hydrolytic stability, allowing the reaction to proceed efficiently without the need for aggressive water removal techniques. This innovation not only streamlines the reactor setup but also dramatically enhances the reaction yield, pushing conversion rates to nearly quantitative levels under optimized conditions. The ability to operate effectively in the presence of moisture is particularly advantageous when handling biomass-derived feedstocks like HMF, which often contain residual water. This robustness translates directly into reduced energy consumption and simplified process control, making the technology highly attractive for large-scale manufacturing environments.
Mechanistic Insights into Metal Triflate-Catalyzed Etherification
The core of this technological advancement lies in the unique coordination chemistry of metal triflates. These compounds function as powerful Lewis acids that activate the hydroxyl group of the HMF molecule, facilitating the nucleophilic attack by a second HMF molecule to form the ether linkage. Crucially, the triflate anion is non-coordinating and extremely stable, preventing the catalyst from decomposing in the presence of the water generated during the dehydration step. This mechanism ensures that the active catalytic species remains intact throughout the reaction cycle, maintaining high turnover frequencies. For process chemists, this means that the reaction kinetics are not inhibited by product accumulation, allowing for higher substrate concentrations and shorter reaction times compared to equilibrium-limited acid-catalyzed processes.
Furthermore, the selectivity of this catalytic system is remarkable, minimizing the formation of humins and other polymeric byproducts that typically foul reactors in biomass conversion. The mild reaction conditions, typically ranging from 70°C to 140°C, prevent the thermal degradation of the sensitive furan ring structure. This preservation of structural integrity is vital for downstream applications, particularly in the synthesis of polyamides where monomer purity dictates the mechanical and thermal properties of the final polymer. The catalyst's tolerance to Lewis bases also suggests a broad substrate scope, potentially allowing for the functionalization of OBMF with various amines or other nucleophiles to create a diverse library of bio-based specialty chemicals.
How to Synthesize Bis-(5-formyl furfuryl) ether Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the economic and technical benefits outlined in the patent. The process is designed to be operationally simple, utilizing standard reactor configurations found in most fine chemical facilities. By optimizing the ratio of substrate to catalyst and selecting the appropriate solvent system, manufacturers can achieve consistent high-quality output. The following guide summarizes the critical operational steps derived from the patent examples to ensure successful replication and scale-up of this high-value transformation.
- Prepare the reaction mixture by dissolving 5-hydroxymethylfurfural (HMF) in an organic solvent such as toluene at a concentration of 10 to 100 g/L.
- Heat the solution to a reaction temperature between 70°C and 140°C, preferably around 110°C, under reflux conditions.
- Add a metal triflate catalyst (e.g., Yb(OTf)3) at a mass ratio of HMF to catalyst between 10: 1 and 50:1, and stir for 6 to 24 hours.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this metal triflate-catalyzed process offers profound strategic advantages beyond mere technical performance. The primary value driver is the drastic simplification of the production workflow, which directly correlates to reduced operational expenditures. By eliminating the need for complex water-removal infrastructure like Dean-Stark traps or molecular sieves, the capital intensity of the manufacturing line is significantly lowered. Moreover, the high selectivity of the reaction reduces the burden on purification units, such as distillation columns or crystallization tanks, leading to lower energy usage and higher overall throughput. This efficiency gain allows for more competitive pricing structures in the volatile market for bio-based intermediates.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the recyclability of the catalyst. Experimental data indicates that the metal triflate catalyst can be recovered and reused multiple times without a discernible loss in activity or selectivity. This circular usage pattern drastically reduces the consumption of expensive rare-earth or transition metal salts, which are often a major cost component in catalytic processes. Additionally, the ability to use crude biomass-derived HMF without rigorous drying pre-treatment further trims raw material preparation costs, creating a leaner and more resilient cost structure for the final OBMF product.
- Enhanced Supply Chain Reliability: Sourcing high-purity intermediates is often bottlenecked by the availability of specialized reagents. However, this method utilizes HMF, which is increasingly produced at industrial scales from abundant agricultural waste like corn stover and bagasse. This shifts the supply base from finite petrochemical resources to renewable, widely available biomass, mitigating the risk of feedstock price volatility. The robustness of the catalyst also means that production schedules are less susceptible to delays caused by reagent sensitivity or strict moisture control requirements, ensuring a more reliable delivery timeline for downstream polymer manufacturers.
- Scalability and Environmental Compliance: As regulatory pressures mount regarding industrial waste and carbon footprints, this technology positions manufacturers favorably. The reduction in salt waste compared to Williamson ether synthesis and the elimination of corrosive liquid acids minimize the environmental impact of the production facility. The process aligns with green chemistry metrics, facilitating easier permitting and compliance with increasingly stringent environmental regulations in key markets like Europe and North America. This sustainability profile is a powerful differentiator when supplying to multinational corporations with aggressive net-zero targets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this OBMF synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: Why are metal triflates preferred over traditional organic acids for OBMF synthesis?
A: Traditional organic acids like p-TSA often result in low yields (around 58%) and require water removal equipment like Dean-Stark traps. Metal triflates offer superior water tolerance, maintaining high catalytic activity even in the presence of moisture, which simplifies the process and boosts yields to over 95%.
Q: Can the catalyst be reused in industrial applications?
A: Yes, the patent demonstrates that metal triflate catalysts can be easily recovered by distilling off the solvent and product, leaving the catalyst in the reactor. Experimental data shows the catalyst maintains high activity and selectivity over at least 5 reuse cycles without significant degradation.
Q: What are the primary applications of Bis-(5-formyl furfuryl) ether?
A: OBMF serves as a versatile bio-based building block. It is primarily used as a monomer for synthesizing high-performance polyamides with excellent thermal and electrical conductivity for aerospace and electronics. Additionally, it acts as a precursor for pharmaceutical agents, including antiviral and anti-tuberculosis drugs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable OBMF Supplier
The transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven scaling capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, offering comprehensive CDMO services tailored to the complex needs of bio-based chemical manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields observed in the lab are faithfully reproduced in the plant. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of OBMF meets the exacting standards required for high-performance polyamide and pharmaceutical applications.
We invite industry leaders to collaborate with us to unlock the full potential of this biomass-derived platform. By leveraging our process optimization expertise, we can help you achieve substantial cost savings and supply chain security. Please contact our technical procurement team to request a Customized Cost-Saving Analysis specific to your volume requirements. We are ready to provide specific COA data and detailed route feasibility assessments to support your next project, ensuring a seamless integration of this advanced intermediate into your value chain.
