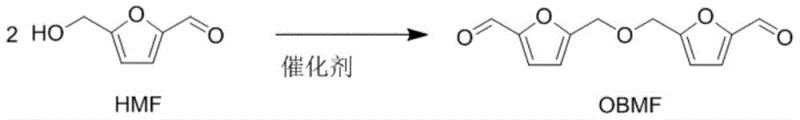
Scalable Production of Bis-(5-formyl furfuryl) Ether Using Robust Metal Triflate Catalysts for Advanced Polymer Applications
The global demand for sustainable, bio-based polymer precursors is driving a paradigm shift in fine chemical manufacturing, moving away from petrochemical dependence toward renewable feedstocks. A pivotal advancement in this sector is detailed in Chinese Patent CN112300101B, which discloses a highly efficient method for preparing bis-(5-formyl furfuryl) ether (OBMF) from 5-hydroxymethylfurfural (HMF). This technology addresses critical bottlenecks in biomass valorization by utilizing robust metal triflate catalysts that overcome the water sensitivity issues plaguing traditional Lewis acids. For R&D directors and procurement strategists, this patent represents a significant opportunity to secure a reliable polymer precursor supplier capable of delivering high-purity intermediates with exceptional process stability. The method not only achieves yields as high as 98% but also simplifies downstream processing, making it an ideal candidate for cost reduction in polyamide manufacturing and other high-value material sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of OBMF has been hindered by inefficient catalytic systems and harsh reaction conditions that compromise both yield and scalability. Conventional routes typically rely on homogeneous organic acids, such as p-toluenesulfonic acid (p-TSA), or base-mediated Williamson etherification. When using p-TSA in toluene, even with the aid of a Dean-Stark apparatus to continuously remove water, the yield of OBMF rarely exceeds 58%. Furthermore, these acidic conditions often promote undesirable side reactions, such as the alkylation of reactants with the solvent, leading to complex impurity profiles that are difficult and expensive to separate. Alternatively, the Williamson reaction pathway, which involves reacting HMF with 5-chloromethyl-2-furfural in the presence of excess alkali, suffers from even lower efficiency, with reported yields stagnating around 42.7%. These legacy methods impose significant burdens on supply chain heads due to low atom economy, high waste generation, and the necessity for extensive purification steps to meet the stringent purity specifications required for electronic or pharmaceutical applications.
The Novel Approach
In stark contrast, the novel approach outlined in the patent leverages the unique properties of metal trifluoromethanesulfonates (metal triflates) to drive the dehydration and etherification of HMF with unprecedented efficiency. Unlike traditional Lewis acids that are rapidly deactivated by moisture, metal triflates such as Yb(OTf)3, Sc(OTf)3, and Zr(OTf)4 possess remarkable water tolerance, allowing the reaction to proceed smoothly without the need for rigorous anhydrous conditions or complex water-removal equipment. This technological leap enables the conversion of biomass-derived HMF into OBMF with yields reaching up to 98%, effectively doubling the output compared to conventional acid-catalyzed methods. The process operates under mild conditions, typically between 70°C and 140°C, and utilizes common organic solvents like toluene, which facilitates easy product isolation via distillation. This robustness translates directly into commercial scale-up of complex polymer additives and intermediates, offering a streamlined pathway that minimizes operational complexity while maximizing throughput.
Mechanistic Insights into Metal Triflate-Catalyzed Etherification
The superior performance of this synthesis route is rooted in the specific coordination chemistry of the metal triflate catalysts. These compounds act as strong Lewis acids that activate the hydroxyl group of the HMF molecule, facilitating the nucleophilic attack by a second HMF molecule to form the ether linkage. Crucially, the triflate anion is non-coordinating and extremely stable, which prevents the catalyst from decomposing in the presence of the water byproduct generated during the dehydration step. This mechanism ensures that the catalytic cycle remains uninterrupted, maintaining high turnover numbers throughout the reaction duration. The ability of these catalysts to function effectively in the presence of Lewis bases containing N, O, P, or S atoms further broadens their applicability, allowing for potential tandem reactions or modifications without catalyst poisoning. This mechanistic stability is a key differentiator for any reliable agrochemical intermediate supplier or pharma partner looking to integrate bio-based building blocks into their existing synthetic pipelines without compromising on reaction reliability.
Furthermore, the impurity control mechanism inherent in this catalytic system is vital for producing high-purity OLED material or pharmaceutical intermediates. The high selectivity of the metal triflate catalysts suppresses the formation of humins and other polymeric byproducts that typically arise from the uncontrolled polymerization of furanic compounds under acidic conditions. By minimizing these side reactions, the process yields a crude product with significantly fewer impurities, thereby reducing the load on downstream purification units. The simplicity of the work-up procedure, which often involves merely distilling off the solvent and product while leaving the catalyst behind, ensures that the final OBMF product meets rigorous quality standards. This level of purity is essential for downstream applications where trace impurities could detrimentally affect the thermal or electrical properties of the resulting polyamides or the efficacy of drug candidates derived from this scaffold.
How to Synthesize Bis-(5-formyl furfuryl) ether Efficiently
The synthesis protocol described in the patent offers a straightforward and reproducible method for generating OBMF, suitable for both laboratory optimization and industrial production. The process begins by charging a reactor with 5-hydroxymethylfurfural and a selected organic solvent, such as toluene, to achieve a substrate concentration between 10 to 100 g/L. The mixture is then heated to the desired reaction temperature, with 110°C identified as the optimal balance between reaction rate and energy efficiency. Once the target temperature is reached, the metal triflate catalyst, preferably Yb(OTf)3, is introduced at a mass ratio of HMF to catalyst of approximately 50:1. The reaction proceeds under stirring for a period of 6 to 24 hours, with 12 hours typically sufficient to reach maximum conversion. Detailed standardized synthesis steps see the guide below.
- Charge a reactor with 5-hydroxymethylfurfural (HMF) and an organic solvent such as toluene, ensuring a concentration between 10 to 100 g/L.
- Heat the mixture to a reaction temperature ranging from 70 to 140°C, with 110°C being optimal for balancing yield and energy consumption.
- Add a metal triflate catalyst, specifically Yb(OTf)3, at a mass ratio of HMF to catalyst of approximately 50: 1, and stir for 12 hours.
- Upon completion, distill off the solvent and product to isolate high-purity OBMF, leaving the catalyst in the reactor for direct reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this metal triflate-catalyzed process offers substantial strategic benefits beyond mere technical performance. The primary advantage lies in the dramatic improvement in process efficiency, which directly correlates to reduced manufacturing costs and enhanced supply security. By eliminating the need for expensive and sensitive anhydrous reagents or complex water-removal setups like Dean-Stark traps, the operational expenditure (OpEx) associated with the synthesis is significantly lowered. The robustness of the catalyst against water contamination means that raw material specifications for HMF can be relaxed slightly, allowing for the use of cost-effective biomass-derived feedstocks without compromising the final yield. This flexibility is crucial for maintaining margin stability in a volatile raw material market and supports the broader industry goal of sustainable sourcing.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the exceptional recyclability of the catalyst. Unlike homogeneous organic acids that are consumed or neutralized during the work-up, the metal triflate catalyst remains in the reactor after the product and solvent are distilled off. This allows for direct reuse in subsequent batches without the need for complex regeneration processes. Experimental data indicates that the catalyst can be recycled multiple times with negligible loss in activity, maintaining yields above 94% even after five cycles. This drastic reduction in catalyst consumption eliminates a major variable cost component and reduces the environmental footprint associated with catalyst disposal, aligning with green chemistry principles and corporate sustainability goals.
- Enhanced Supply Chain Reliability: The reliance on biomass-derived HMF as the sole carbon source decouples the production of OBMF from fossil fuel volatility, offering a more stable long-term supply chain. HMF can be conveniently produced from abundant agricultural and forestry wastes such as straw, corn stalks, and bagasse, ensuring a continuous and diverse feedstock base. Furthermore, the use of common solvents like toluene, rather than hazardous chlorinated solvents like dichloromethane (which has high vapor pressure and regulatory restrictions), simplifies logistics and storage requirements. This shift towards safer, more abundant materials reduces the risk of supply disruptions caused by regulatory changes or raw material shortages, ensuring consistent delivery schedules for downstream customers.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is exceptionally well-suited for commercial expansion. The high selectivity of the reaction minimizes the generation of hazardous waste streams, simplifying effluent treatment and reducing compliance costs. The ability to operate at moderate temperatures (70-140°C) and atmospheric pressure reduces the engineering complexity and capital expenditure (CapEx) required for reactor design compared to high-pressure hydrogenation or cryogenic processes. The simplicity of the separation step—essentially a distillation—allows for easy integration into existing fine chemical manufacturing facilities. This ease of scale-up ensures that production volumes can be rapidly increased from pilot scale to hundreds of tons annually to meet growing market demand for bio-based polyamides and advanced materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of adopting this synthesis route. Understanding these details is essential for technical teams evaluating the feasibility of integrating OBMF into their product portfolios.
Q: Why are metal triflates preferred over traditional organic acids for OBMF synthesis?
A: Traditional organic acids like p-TSA often result in low yields (around 58%) and require complex water removal setups like Dean-Stark traps. Metal triflates, particularly Yb(OTf)3, exhibit strong water tolerance, maintaining high catalytic activity even without rigorous dehydration, leading to yields up to 98%.
Q: Can the catalyst be recovered and reused in this process?
A: Yes, the metal triflate catalyst demonstrates excellent stability and recyclability. After the reaction, the solvent and product can be distilled off, leaving the catalyst in the reactor. Experimental data shows the catalyst can be reused for at least 5 cycles with yields remaining between 94% and 97%.
Q: What are the primary applications of the synthesized OBMF?
A: OBMF serves as a versatile bio-based building block. It is primarily used as a precursor for synthesizing high-performance polyamides, polyurethanes, and crown ethers. Additionally, due to its antiviral and antioxidant properties, it holds potential as an intermediate for pharmaceutical applications, including hepatitis B virus drug precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-(5-formyl furfuryl) ether Supplier
As the market for bio-based high-performance materials accelerates, securing a partner with deep technical expertise and proven manufacturing capabilities is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging advanced catalytic technologies like the metal triflate system to deliver superior quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of OBMF meets the exacting standards required for aerospace materials, microelectronics, and pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall production costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing processes can support your innovation goals and drive your business forward in the competitive landscape of sustainable chemicals.
