Advanced Chemical Synthesis of Orlistat Intermediates: A Scalable Alternative to Fermentation
Advanced Chemical Synthesis of Orlistat Intermediates: A Scalable Alternative to Fermentation
The global demand for effective anti-obesity therapeutics has placed immense pressure on supply chains to deliver high-purity active pharmaceutical ingredients (APIs) and their precursors reliably. Orlistat, a potent gastrointestinal lipase inhibitor, remains a cornerstone in weight management therapy, yet its production has historically been bottlenecked by inefficient biological fermentation processes. Patent CN1321114C introduces a transformative chemical methodology that bypasses these biological limitations through a concise, eight-step synthetic route. This innovation leverages advanced asymmetric catalysis to construct the complex chiral architecture of the molecule with superior efficiency. By shifting from microbial fermentation to a fully chemical synthesis, manufacturers can achieve drastic improvements in throughput and cost-efficiency. This report analyzes the technical merits of this route, highlighting its potential for reliable pharmaceutical intermediates supplier networks seeking to optimize their portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Orlistat and its precursors relied heavily on fermentation technologies utilizing strains such as Streptomyces toxinii. While biologically elegant, these methods are plagued by severe economic and operational inefficiencies that hinder large-scale adoption. The patent background explicitly notes that fermentation yields are abysmally low, often producing merely 0.15 grams of lipostatin per liter of culture medium after an arduous 138-hour incubation period. Such low titers necessitate massive fermentation tanks and extensive downstream processing to isolate the product from the complex broth, driving up the cost of goods sold (COGS) significantly. Furthermore, biological systems are inherently variable, susceptible to contamination, and difficult to scale linearly without compromising yield. Alternative chemical routes proposed by other entities, such as the twelve-step synthesis involving selective reduction of dihydropyran-2-ones, also suffer from excessive length and the use of prohibitively expensive chiral reagents for controlling the C5 hydroxyl stereochemistry.
The Novel Approach
In stark contrast to these legacy methods, the methodology disclosed in CN1321114C streamlines the entire manufacturing workflow into a robust eight-step sequence. The core innovation lies in the direct asymmetric aldol condensation between lauryl aldehyde and specific enol silyl ethers. This single step effectively establishes the critical carbon backbone and the primary chiral center with high fidelity, eliminating the need for lengthy resolution steps found in older chemical routes. By utilizing readily available commodity chemicals like lauryl aldehyde as the starting material, the process decouples production from the volatility of biological feedstocks. The route strategically employs protecting group chemistry and stereoselective reductions to build complexity efficiently. This reduction in step count from twelve to eight not only improves the overall cumulative yield but also drastically simplifies the purification burden at each stage. For procurement teams, this translates to a more predictable and cost-effective supply chain for high-purity Orlistat intermediates.
Mechanistic Insights into Chiral Titanium-BINOL Catalyzed Aldol Condensation
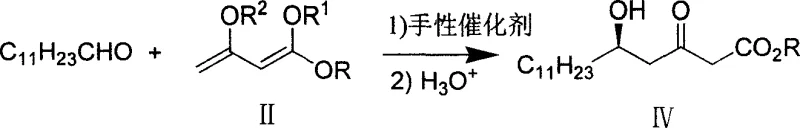
The cornerstone of this synthetic strategy is the initial enantioselective aldol reaction, which sets the absolute stereochemistry for the entire molecule. The process utilizes sophisticated chiral catalysts derived from binaphthyl ligands coordinated with transition metals such as Titanium (Ti), Copper (Cu), or Zirconium (Zr). Specifically, the patent highlights the efficacy of (S)-BINOL-Ti(OPr-i)2 type complexes. These catalysts operate by forming a rigid chiral pocket that directs the approach of the enol silyl ether to the aldehyde electrophile. The coordination geometry ensures that the nucleophilic attack occurs exclusively from one face of the carbonyl group, resulting in an enantiomeric excess (ee) greater than 97 percent. This high level of stereocontrol at the outset is critical because it minimizes the formation of unwanted diastereomers in subsequent steps, thereby simplifying the impurity profile. The use of catalytic amounts (molar ratios as low as 1/2000 to 1/4000) further underscores the efficiency of this system, avoiding the waste associated with stoichiometric chiral auxiliaries.
Beyond the initial bond formation, the subsequent steps rely on substrate-controlled diastereoselectivity to install the remaining chiral centers. For instance, the introduction of the hexyl side chain via enolate alkylation is performed at cryogenic temperatures (-78°C) to kinetically favor the desired stereoisomer. The existing chiral centers at C3 and C5 exert a strong directing effect on the incoming alkyl halide, ensuring that the new C2 center is formed with the correct configuration. This interplay between catalyst-controlled and substrate-controlled stereochemistry allows for the precise construction of the molecule's three contiguous chiral centers without the need for intermediate resolution. From a quality control perspective, this mechanistic rigor ensures that the final API meets stringent regulatory specifications for chiral purity, reducing the risk of batch rejection due to isomeric impurities.
How to Synthesize Orlistat Efficiently
The execution of this eight-step synthesis requires precise control over reaction parameters, particularly temperature and stoichiometry, to maximize yield and purity. The process begins with the generation of the chiral titanium catalyst in situ under inert atmosphere, followed by the slow addition of reactants to manage exotherms. Subsequent protection and reduction steps utilize standard reagents but require careful monitoring to prevent over-reaction or epimerization. The critical alkylation step demands strict temperature maintenance at -78°C to preserve diastereoselectivity. Finally, the assembly of the leucine side chain via Mitsunobu coupling requires anhydrous conditions to ensure high conversion. For detailed operational parameters, safety data, and specific workup procedures, please refer to the standardized synthesis guide below.
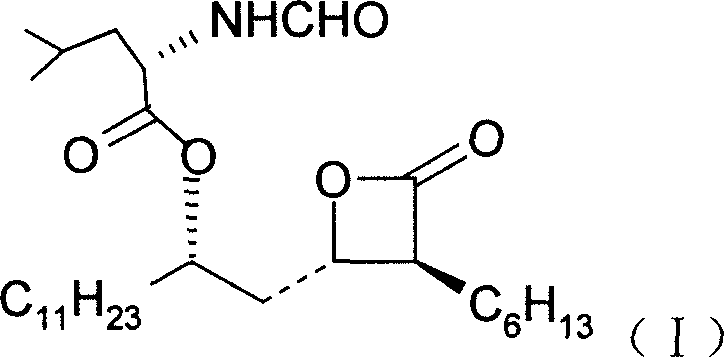
- Perform asymmetric catalytic aldol condensation between lauryl aldehyde and enol silyl ether using a chiral Ti-BINOL catalyst to obtain (5R)-5-hydroxy-3-oxo hexadecanoic acid methyl ester.
- Protect the hydroxyl group of the resulting beta-keto ester using a protecting agent such as p-methoxybenzyl chloride to form the protected intermediate.
- Conduct asymmetric catalytic hydrogenation using a chiral bisphosphine metal complex (Ru/Rh/Pd) to stereoselectively reduce the ketone to a hydroxyl group.
- Introduce a hexyl group at the alpha-carbon of the ester via enolate alkylation using n-BuLi and 1-iodohexane at low temperatures (-78°C) to control stereochemistry.
- Hydrolyze the methyl ester under basic conditions (KOH/MeOH) to generate the corresponding carboxylic acid intermediate.
- Induce intramolecular cyclization using TsCl and pyridine to form the chiral beta-lactone ring structure.
- Remove the hydroxyl protecting group via catalytic hydrogenation (Pd/C) to reveal the free hydroxyl functionality on the lactone side chain.
- Complete the synthesis by coupling the lactone alcohol with N-formyl-L-leucine under Mitsunobu conditions (PPh3, DEAD) to yield Orlistat.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain directors and procurement managers, the shift from fermentation to this optimized chemical synthesis represents a strategic opportunity to de-risk the supply of weight-loss drug intermediates. The reliance on fermentation creates a single point of failure dependent on biological strain performance and facility capacity. In contrast, this chemical route utilizes a modular approach where each step can be scaled independently in standard multipurpose reactors. The raw materials, including lauryl aldehyde and various silyl ethers, are commodity chemicals available from multiple global vendors, ensuring supply continuity even if one source faces disruption. This diversification of the supply base is crucial for maintaining production schedules in the face of market volatility. Furthermore, the elimination of the fermentation step removes the need for specialized bioprocessing infrastructure, allowing production to be outsourced to a wider range of CDMOs with organic synthesis capabilities.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the drastic reduction in process steps and the elimination of expensive biological media. By shortening the synthesis from twelve steps to eight, the cumulative yield is significantly improved, meaning less raw material is wasted to produce the same amount of final product. Additionally, the use of catalytic chiral induction rather than stoichiometric chiral reagents lowers the material cost per kilogram substantially. The removal of the fermentation stage also eliminates the high energy and water consumption associated with sterilization and aeration, leading to lower utility costs. These factors combine to create a leaner cost structure that can withstand pricing pressure in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: Chemical synthesis offers a level of predictability that fermentation cannot match. Batch cycles in this process are measured in hours or days rather than the nearly six days required for microbial growth, allowing for faster turnover and quicker response to demand spikes. The robustness of the chemical reactions means that batch-to-batch variability is minimized, reducing the incidence of out-of-specification results that delay shipments. Moreover, the intermediates generated in the early steps are stable and can be stockpiled, creating a buffer inventory that protects against downstream disruptions. This reliability is essential for partners requiring just-in-time delivery of high-purity lipase inhibitor intermediates.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial tonnage is straightforward due to the use of conventional unit operations like distillation, crystallization, and filtration. Unlike fermentation, which generates vast amounts of biological waste sludge, this chemical route produces waste streams that are easier to characterize and treat. The high atom economy of the aldol condensation and the efficient use of reagents contribute to a greener process profile. The ability to run reactions at higher concentrations compared to dilute fermentation broths also reduces solvent usage per unit of product. These environmental advantages facilitate easier regulatory approval and align with the sustainability goals of modern pharmaceutical manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific advantages outlined in the patent documentation and are intended to clarify the feasibility of adopting this technology for commercial production. Understanding these nuances is vital for R&D teams evaluating technology transfer and for procurement officers negotiating supply agreements. The answers reflect the consensus on best practices for maximizing the value of this intellectual property.
Q: Why is the chemical synthesis method preferred over fermentation for Orlistat production?
A: Fermentation processes using Streptomyces strains typically suffer from extremely low yields (approx. 0.15 g/L) and long cycle times (138 hours), leading to high production costs. The chemical route described in CN1321114C shortens the synthesis to 8 steps with higher efficiency and uses readily available raw materials like lauryl aldehyde.
Q: How is stereochemical purity controlled in this synthesis route?
A: High enantiomeric excess (>97% ee) is achieved in the first step using chiral BINOL-Titanium complexes for the aldol condensation. Subsequent steps utilize substrate-controlled stereoselectivity during the alkylation at -78°C and chiral hydrogenation catalysts to maintain the integrity of the multiple chiral centers.
Q: What are the scalability advantages of this 8-step process?
A: Unlike fermentation which requires massive bioreactors and complex downstream purification, this chemical process relies on standard organic unit operations (condensation, hydrogenation, crystallization). The reduction from 12 steps to 8 steps significantly improves overall yield and reduces the accumulation of impurities, making it highly suitable for multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Orlistat Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in securing the global supply of essential medicines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are equipped with rigorous QC labs and state-of-the-art analytical instrumentation to verify that every batch meets stringent purity specifications, including chiral purity and residual solvent limits. Our commitment to quality assurance means that we can consistently deliver Orlistat intermediates that comply with international pharmacopeial standards, providing our partners with the confidence they need to file regulatory dossiers.
We invite you to collaborate with us to optimize your supply chain for weight management therapeutics. Our engineering team is ready to perform a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By leveraging our expertise in asymmetric catalysis and process intensification, we can help you reduce lead time for high-purity pharmaceutical intermediates while minimizing total landed cost. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your next project.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
