Optimized Industrial Synthesis of Phloroglucinol: Enhancing Purity and Scalability for Global Supply Chains
Optimized Industrial Synthesis of Phloroglucinol: Enhancing Purity and Scalability for Global Supply Chains
The landscape of fine chemical manufacturing is constantly evolving, driven by the urgent need for more efficient, cost-effective, and environmentally sustainable production methodologies. A pivotal advancement in this domain is detailed in patent CN113480413A, which introduces a novel preparation method for phloroglucinol, a critical intermediate widely utilized in the synthesis of flavonoid compounds, antitumor drugs, and various industrial stabilizers. This technology addresses longstanding inefficiencies in the conventional synthesis routes, specifically targeting the excessive reaction times and product degradation issues that have historically plagued manufacturers. By leveraging a refined base-catalyzed substitution strategy in a pseudocumene solvent system, this innovation offers a robust pathway to achieving ultra-high purity standards while drastically optimizing energy consumption and equipment utilization rates.
For global procurement leaders and R&D directors, the implications of this technological shift are profound. The ability to produce high-purity phloroglucinol with reduced cycle times directly translates to enhanced supply chain resilience and lower operational expenditures. Unlike traditional methods that often struggle with product discoloration due to prolonged exposure to high temperatures, this patented approach integrates a sophisticated dual-stage decolorization process. This ensures that the final crystalline product meets the stringent quality requirements necessary for pharmaceutical applications, thereby reducing the risk of batch rejection and downstream processing failures. As we delve deeper into the technical specifics, it becomes evident that this method represents a significant leap forward in the commercial viability of complex aromatic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of phloroglucinol from 2,6-dichlorophenol has been hindered by severe kinetic limitations and thermal instability issues. Conventional reports indicate that the reaction between 2,6-dichlorophenol and strong bases typically requires extensive durations, often spanning 12 to 24 hours even at the laboratory stage. When these processes are scaled up to pilot or industrial levels, the reaction time frequently extends to a staggering 24 to 48 hours to achieve acceptable conversion rates. This prolonged exposure to elevated temperatures creates a hostile environment for the product, leading to significant oxidative degradation and carbonization. Consequently, the resulting crude material often exhibits a dark coloration and a complex impurity profile, necessitating aggressive and costly purification steps that further erode overall yield and profitability.
Furthermore, the extended reaction cycles impose a heavy burden on manufacturing infrastructure. Keeping reaction vessels at high temperatures for days not only spikes energy consumption but also accelerates the wear and tear on expensive reactor linings and agitation systems. From a supply chain perspective, such long batch cycles limit the throughput capacity of a facility, creating bottlenecks that can jeopardize delivery schedules for downstream clients. The combination of high energy costs, equipment maintenance overheads, and the potential for variable product quality due to thermal stress makes the traditional methodology increasingly untenable in a competitive global market where speed and consistency are paramount.
The Novel Approach
In stark contrast to these legacy challenges, the methodology outlined in patent CN113480413A introduces a highly optimized reaction protocol that fundamentally redefines the efficiency of phloroglucinol synthesis. By precisely controlling the reaction environment within a pseudocumene solvent system and maintaining a strict temperature window of 150-160°C, the process successfully compresses the reaction timeline to a mere 1 to 10 hours. Remarkably, even during large-scale amplification tests in industrial reactors, the reaction time remains consistently between 8 and 10 hours, representing a dramatic reduction compared to the multi-day cycles of the past. This acceleration is achieved without compromising conversion efficiency, allowing manufacturers to significantly increase their batch turnover rates and overall production capacity.
Beyond speed, the novel approach places a premium on product integrity through an advanced post-treatment regimen. The integration of a dual-stage decolorization process, utilizing sodium hydrosulfite and activated carbon in both organic and aqueous phases, effectively removes colored byproducts and trace impurities that typically persist in crude batches. This results in a final product that presents as a white crystalline powder with exceptional clarity, eliminating the need for repetitive recrystallization steps. The strategic adjustment of pH to 2-3 during acidification further ensures the complete precipitation of the target molecule while minimizing waste generation. Collectively, these innovations deliver a manufacturing route that is not only faster but also inherently cleaner and more economically sustainable.
Mechanistic Insights into Base-Catalyzed Substitution and Purification
The core of this technological breakthrough lies in the precise manipulation of nucleophilic aromatic substitution dynamics under high-temperature conditions. The reaction initiates with the deprotonation of 2,6-dichlorophenol by potassium hydroxide, generating a reactive phenoxide species within the pseudocumene medium. Under the applied thermal energy of 150-160°C, this intermediate undergoes a series of transformations, potentially involving benzyne-like intermediates or direct nucleophilic displacement, ultimately leading to the introduction of hydroxyl groups at the meta positions. The choice of pseudocumene as the solvent is critical, as it provides the necessary thermal stability and solubility profile to sustain the reaction kinetics without promoting premature decomposition. The molar ratio of potassium hydroxide to substrate, optimized between 1:3.1 and 1:9, ensures an excess of base to drive the equilibrium towards the fully substituted trihydroxybenzene structure.
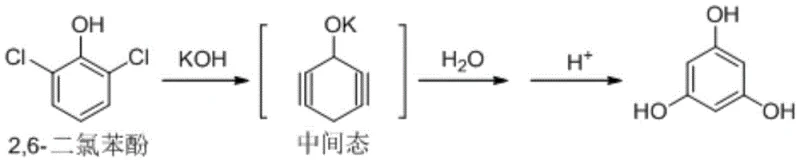
Following the primary synthesis, the control of impurities becomes the defining factor for pharmaceutical-grade quality. The patent details a meticulous purification sequence where the crude reaction mixture is first subjected to phase separation upon cooling to 135-140°C. The aqueous layer, containing the potassium salt of phloroglucinol, is then acidified to precipitate the free acid form. Crucially, the subsequent decolorization steps are designed to target specific chromophores formed during the high-temperature reaction. The first decolorization in ethyl acetate removes bulk organic impurities, while the second decolorization in an aqueous reflux system targets residual polar contaminants. This orthogonal purification strategy ensures that genotoxic impurities and unknown single impurities are suppressed to levels below 0.002%, guaranteeing a final HPLC purity that consistently exceeds 99.99%.
How to Synthesize Phloroglucinol Efficiently
Implementing this advanced synthesis route requires strict adherence to the defined thermal and stoichiometric parameters to ensure reproducibility and safety. The process begins with the preparation of a clear solution of 2,6-dichlorophenol in pseudocumene, which is then introduced into a pre-heated suspension of potassium hydroxide. Maintaining the nitrogen atmosphere throughout the heating and reaction phases is essential to prevent oxidative side reactions that could compromise the whiteness of the final crystal. Operators must carefully monitor the internal temperature to stay within the 150-160°C window, as deviations can impact the reaction rate and impurity profile. Once the reaction is complete, the controlled addition of purified water and subsequent acidification steps must be executed with precision to maximize recovery yields.
- Dissolve 2,6-dichlorophenol in pseudocumene and prepare a potassium hydroxide suspension in the same solvent under nitrogen protection.
- Heat the system to 150-160°C, add the phenol solution, and maintain reflux for 1-10 hours to complete the conversion.
- Cool to 135-140°C, add water for phase separation, acidify the aqueous layer to pH 2-3, and perform double decolorization using sodium hydrosulfite and activated carbon.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this optimized synthesis method offers tangible strategic benefits that extend far beyond simple chemical yield. The most immediate impact is observed in the realm of operational expenditure, where the drastic reduction in reaction time directly correlates with lowered energy costs. By shortening the heating cycle from nearly two days to under ten hours, facilities can achieve a substantial decrease in utility consumption per kilogram of product produced. Furthermore, the accelerated batch cycle allows for a higher frequency of production runs within the same timeframe, effectively increasing the asset utilization rate of existing reactor farms without the need for capital-intensive infrastructure expansion. This efficiency gain provides a buffer against market volatility, enabling suppliers to respond more agilely to fluctuations in demand.
- Cost Reduction in Manufacturing: The elimination of prolonged heating periods and the reduction in byproduct formation lead to significant cost savings across the production value chain. By minimizing the formation of tarry residues and oxidized impurities, the process reduces the load on downstream purification units, lowering the consumption of solvents and decolorizing agents like activated carbon. Additionally, the improved selectivity of the reaction means that less raw material is wasted on side products, enhancing the overall atom economy. These cumulative efficiencies result in a more competitive cost structure, allowing for better pricing flexibility in negotiations with downstream pharmaceutical partners while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The robustness of this method at the pilot and commercial scale ensures a consistent and predictable supply of high-purity phloroglucinol. Traditional methods often suffer from batch-to-batch variability due to the sensitivity of long reaction times to minor thermal fluctuations. In contrast, the shortened reaction window of this novel approach minimizes the opportunity for process drift, ensuring that every batch meets the stringent specifications required for API synthesis. This reliability reduces the risk of supply disruptions and quality disputes, fostering stronger, long-term partnerships between chemical manufacturers and their clients who depend on uninterrupted material flow for their own production schedules.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, the process demonstrates excellent scalability and a reduced ecological footprint. The efficient use of reagents and the minimization of waste streams align with modern green chemistry principles, simplifying the permitting and compliance landscape for manufacturing sites. The specific control of pH during acidification prevents the overuse of concentrated hydrochloric acid, reducing the volume of saline wastewater generated. Moreover, the production of a white, high-purity crystal reduces the need for reprocessing or disposal of off-spec material, contributing to a cleaner and more sustainable manufacturing operation that meets the increasingly rigorous ESG criteria of global corporate buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced phloroglucinol preparation technology. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these nuances is critical for R&D teams assessing process transfer and procurement officers evaluating supplier capabilities.
Q: How does the new preparation method improve upon traditional phloroglucinol synthesis?
A: The patented method significantly reduces reaction time from the traditional 24-48 hours in pilot scales down to 8-10 hours, while simultaneously minimizing product oxidation and carbonization through precise temperature control and dual-stage decolorization.
Q: What represents the critical purity specifications achieved in this process?
A: Through the implementation of a rigorous two-step decolorization protocol using sodium hydrosulfite and activated carbon, the process achieves an HPLC purity exceeding 99.99%, with genotoxic impurities undetected and total impurities controlled below 0.01%.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process has been validated for scalability, maintaining consistent reaction kinetics and product quality even when transitioning from laboratory glassware to large enamel and stainless steel reaction kettles, ensuring reliable supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phloroglucinol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of this patented synthesis are fully realized in a GMP-compliant environment. We are committed to delivering phloroglucinol that adheres to stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our infrastructure is designed to handle the specific thermal and solvent requirements of this process, guaranteeing a supply of material that is both chemically superior and commercially viable.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can integrate into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits specific to your volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate concretely how our manufacturing prowess can support your R&D and production goals. Let us collaborate to secure a stable, high-quality supply of this critical intermediate for your next generation of pharmaceutical or fine chemical products.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
