Advanced Acylation Technology for High-Purity 7-Acylamino-Cephalosporin Derivatives Manufacturing
Advanced Acylation Technology for High-Purity 7-Acylamino-Cephalosporin Derivatives Manufacturing
The pharmaceutical industry constantly seeks robust synthetic routes that balance high yield with exceptional purity, particularly for complex beta-lactam antibiotics. Patent CN1019300B introduces a transformative methodology for the preparation of carboxylic acid amides, specifically targeting the acylation of amines using carboxylic acid 2-benzothiazolylthioesters. Unlike traditional protocols that necessitate the conversion of amine salts to free bases, this innovation allows the amine to react directly in its acid addition salt form. This breakthrough is particularly significant for the synthesis of 7-acylamino-cephalosporin derivatives, where the free base form often exhibits instability. By leveraging this novel approach, manufacturers can achieve high yields of high-purity antibacterial agents while streamlining the production workflow, positioning this technology as a cornerstone for reliable pharmaceutical intermediates supplier networks aiming for process intensification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acylation of amines with 2-benzothiazolylthioesters has relied heavily on the availability of the amine in its free base form. In standard practice, if the starting material is an acid addition salt, which is common for stabilizing sensitive amines like cephalosporins, a neutralization step is mandatory. This typically involves adding at least one equivalent of an organic base, such as a tertiary amine or DBU, to the reaction mixture to liberate the reactive free amine. This extra step introduces several complications: it increases the stoichiometric complexity, requires additional reagents that must later be removed, and exposes the sensitive beta-lactam core to basic conditions that can trigger degradation or epimerization. Furthermore, the presence of excess organic base can complicate downstream purification, leading to higher levels of residual impurities and increased solvent consumption during workup, ultimately driving up the cost reduction in API manufacturing challenges.
The Novel Approach
The methodology disclosed in the patent fundamentally shifts this paradigm by demonstrating that acid addition salts of amines can react directly with 2-benzothiazolylthioesters without prior neutralization. This unexpected reactivity eliminates the need for organic bases entirely, simplifying the reaction matrix significantly. The process utilizes the inherent nucleophilicity of the amine salt in the presence of the highly activated thioester, allowing the acylation to proceed efficiently under mild conditions. This is especially advantageous for 7-amino-cephalosporin derivatives which are prone to decomposition in free base forms. By maintaining the amine in its salt form throughout the reaction, the structural integrity of the molecule is preserved, leading to superior product quality. 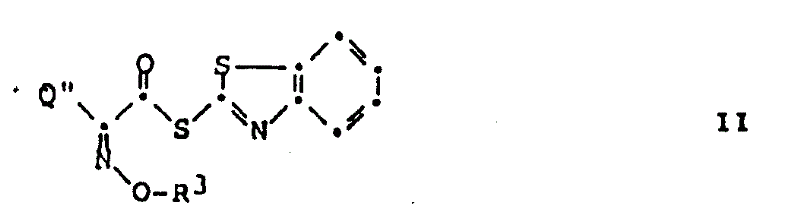
Mechanistic Insights into Direct Salt Acylation
The mechanistic underpinning of this reaction involves the interaction between the protonated amine species and the electrophilic carbonyl carbon of the 2-benzothiazolylthioester. While conventional wisdom suggests that a free lone pair is required for nucleophilic attack, the high reactivity of the benzothiazolyl leaving group facilitates the displacement even in the presence of the counterion. The reaction likely proceeds through an equilibrium where a small fraction of the free amine is generated in situ or through a direct concerted mechanism assisted by the solvent environment. This subtle balance prevents the accumulation of free base, thereby mitigating degradation pathways common in cephalosporin chemistry. The use of solvents like dichloromethane further stabilizes the transition state, ensuring that the acylation occurs selectively at the 7-amino position without affecting other sensitive functional groups on the cephem nucleus.
Impurity control is a critical aspect of this mechanism, particularly for regulatory compliance in antibiotic production. By avoiding strong organic bases, the formation of base-catalyzed byproducts, such as ring-opened species or stereoisomers, is drastically reduced. The reaction conditions, typically maintained between 15°C and 30°C, are mild enough to prevent thermal degradation yet sufficient to drive the reaction to completion within a reasonable timeframe. The resulting products, often isolated as stable acid addition salts themselves, exhibit excellent crystallinity and purity profiles. 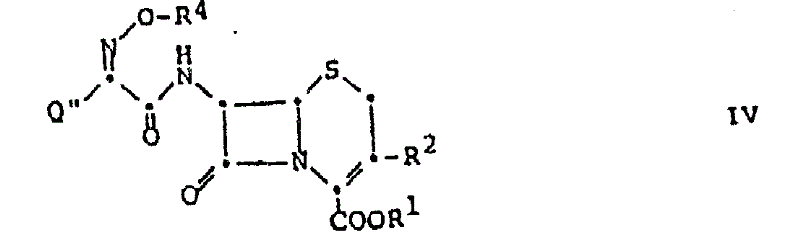
How to Synthesize 7-Acylamino-Cephalosporin Efficiently
The synthesis protocol derived from this patent offers a straightforward path to producing complex cephalosporin intermediates with minimal operational overhead. The process begins with the suspension of the amine acid addition salt, such as a p-toluenesulfonate or hydrochloride salt, in a suitable inert solvent. The acylating agent is then introduced directly, and the mixture is stirred under controlled temperatures. This elimination of the neutralization step not only saves time but also reduces the chemical load on the reactor. For detailed operational parameters, stoichiometry, and specific workup procedures tailored to your specific substrate, please refer to the standardized synthesis guide below.
- Suspend the 7-amino-cephalosporin acid addition salt (e.g., p-toluenesulfonate or hydrochloride) in an inert organic solvent such as dichloromethane.
- Add the carboxylic acid 2-benzothiazolylthioester acylating agent directly to the suspension at a temperature range of 15°C to 30°C.
- Stir the reaction mixture for 1.5 to 3 hours, then perform aqueous workup with sodium acetate solution followed by crystallization to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology addresses several pain points traditionally associated with cephalosporin manufacturing. The elimination of organic bases like DBU, which are costly and require careful handling, translates directly into raw material savings. Furthermore, the simplified workup procedure reduces the volume of solvents and washing solutions required, lowering waste disposal costs and environmental impact. For procurement managers, this means a more predictable cost structure and reduced dependency on specialized reagents that may face supply volatility. The ability to run reactions at room temperature also reduces energy consumption associated with heating or cooling, contributing to a greener and more economical manufacturing footprint.
- Cost Reduction in Manufacturing: The removal of the neutralization step eliminates the need for expensive organic bases and the associated quenching processes. This streamlining reduces the total number of unit operations, leading to substantial cost savings in labor and utilities. Additionally, the higher purity of the crude product minimizes the need for extensive recrystallization or chromatography, further enhancing the economic viability of the process for large-scale production.
- Enhanced Supply Chain Reliability: By utilizing stable acid addition salts as starting materials, the process mitigates risks associated with the degradation of sensitive free base amines during storage and transport. This stability ensures a consistent supply of high-quality intermediates, reducing the likelihood of batch failures. The robustness of the reaction conditions also allows for greater flexibility in sourcing raw materials, as the process is less sensitive to minor variations in reagent quality.
- Scalability and Environmental Compliance: The use of common solvents like dichloromethane and the absence of hazardous strong bases make this process highly scalable from pilot plant to commercial tonnage. The reduced generation of saline waste from neutralization steps simplifies effluent treatment, aiding in compliance with stringent environmental regulations. This scalability ensures that the technology can meet the demands of global markets for high-purity pharmaceutical intermediates without compromising on safety or sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this acylation technology. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this route for their specific product pipelines. The answers are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for technical decision-making.
Q: Why is reacting the amine as an acid addition salt advantageous?
A: Reacting the amine as an acid addition salt eliminates the need for pre-neutralization with organic bases like DBU or triethylamine. This is particularly beneficial for amines that are unstable in their free base form, such as certain 7-amino-cephalosporin derivatives, preventing degradation and improving overall yield.
Q: What solvents are compatible with this acylation method?
A: The process is versatile and can be conducted in various inert organic solvents where raw materials are at least partially soluble. Preferred solvents include halogenated lower hydrocarbons like dichloromethane, lower alcohols, and N,N-dialkyl fatty acid amides, with dichloromethane being identified as the optimal choice for reaction efficiency.
Q: Does this method improve impurity profiles compared to conventional routes?
A: Yes, by avoiding the use of excess organic bases and the associated neutralization steps, the formation of base-catalyzed side products is minimized. The direct reaction of the salt form allows for milder conditions, typically at room temperature, which preserves the integrity of sensitive beta-lactam rings and results in high-purity final products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Acylamino-Cephalosporin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and pure synthetic routes in the competitive landscape of antibiotic manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the direct salt acylation method are translated into reliable supply chains. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 7-acylamino-cephalosporin meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of beta-lactam chemistry with precision, delivering intermediates that facilitate the production of safe and effective medicines.
We invite you to collaborate with us to leverage this advanced technology for your specific requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain for high-purity pharmaceutical intermediates.
