Advanced Synthetic Route for Linagliptin Intermediates: Enhancing Purity and Scalability
The global pharmaceutical landscape is continuously evolving, driven by the urgent need for more effective treatments for chronic conditions such as type 2 diabetes. Linagliptin, a potent DPP-IV inhibitor, has emerged as a cornerstone therapy due to its favorable safety profile and efficacy. However, the manufacturing of its key intermediates often presents significant challenges regarding safety, cost, and environmental impact. Patent CN107936023B introduces a groundbreaking synthetic methodology that addresses these critical bottlenecks. This technical insight report analyzes the novel oxidative coupling strategy disclosed in the patent, highlighting its potential to redefine the supply chain dynamics for pharmaceutical intermediates. By shifting away from hazardous nitrosation protocols towards a streamlined oxidative cyclization, this technology offers a robust pathway for producing high-purity linagliptin precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the critical linagliptin intermediate, specifically the 8-iodo-1-methylxanthine derivative (Compound II), has relied on convoluted multi-step sequences that pose substantial risks to both personnel and the environment. As illustrated in prior art references such as Route Two, the conventional process typically initiates with 6-amino-1-methyluracil, which must undergo a dangerous nitrosation reaction using sodium nitrite. This step is inherently unstable, carrying the risk of explosive decomposition and generating toxic nitrogen oxide byproducts. Furthermore, the subsequent transformation requires a series of discrete operations including reduction, cyclization, iodination, and finally substitution. Each additional unit operation introduces opportunities for yield loss, impurity accumulation, and increased solvent consumption. The cumulative effect of these inefficiencies results in a process that is not only economically burdensome but also difficult to scale safely under Good Manufacturing Practice (GMP) conditions.
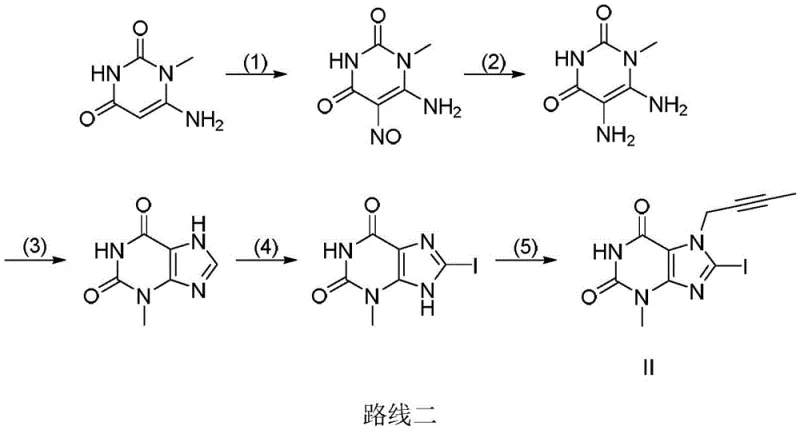
The Novel Approach
In stark contrast to the legacy methodologies, the invention disclosed in CN107936023B proposes a radically simplified two-step strategy that bypasses the hazardous nitrosation entirely. The core innovation lies in the direct construction of the xanthine scaffold via an oxidative coupling cyclization reaction. Starting from commercially abundant 1-methyluracil and urea, the process utilizes an iodine source and an oxidant under mild acid catalysis to directly forge the requisite carbon-nitrogen bonds and install the iodine atom in a single pot. This telescoped approach drastically reduces the number of isolation steps and minimizes the handling of reactive intermediates. The subsequent alkylation with 1-bromo-2-butyne proceeds under温和 conditions to deliver the final target molecule. This streamlined workflow not only enhances the overall throughput but also significantly lowers the E-factor (environmental factor) of the manufacturing process, aligning perfectly with modern green chemistry principles.
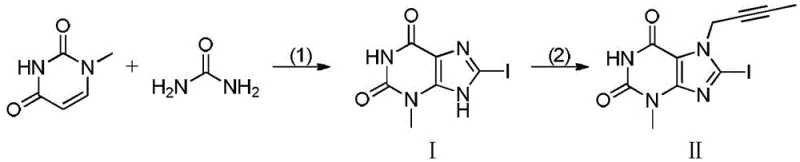
Mechanistic Insights into Oxidative Coupling Cyclization
The heart of this technological advancement is the oxidative coupling mechanism that facilitates the formation of the purine-like core. In the first step, 1-methyluracil reacts with urea in the presence of an iodide source, such as potassium iodide, and an oxidant like tert-butyl hydroperoxide (TBHP). The acid catalyst, preferably sulfuric acid, activates the carbonyl groups, promoting nucleophilic attack by urea. Simultaneously, the oxidant mediates the dehydrogenative coupling, while the iodide species acts as both a catalyst and an iodinating agent, ensuring the regioselective introduction of the iodine atom at the C-8 position. This tandem cyclization-iodination sequence is highly efficient, proceeding at temperatures between 60°C and 90°C in solvents like dioxane. The mechanistic elegance ensures that the reaction trajectory favors the desired heterocyclic formation over potential polymerization or degradation pathways, resulting in a crude product of exceptional quality.
Impurity control is intrinsically built into this reaction design. By avoiding the harsh acidic and reducing conditions associated with traditional nitrosation-reduction sequences, the formation of genotoxic impurities and complex side products is minimized. The use of specific bases like N,N-diisopropylethylamine (DIPEA) in the second substitution step further ensures that the alkylation occurs selectively at the N-7 position without affecting other sensitive functional groups. The patent data indicates that simple recrystallization from dichloromethane and cyclohexane is sufficient to achieve purity levels exceeding 99%. This high level of chemical fidelity is crucial for downstream processing, as it reduces the burden on purification units and ensures that the final API meets stringent regulatory specifications for residual solvents and related substances.
How to Synthesize Linagliptin Intermediate Efficiently
The practical implementation of this synthetic route is straightforward and amenable to standard reactor configurations found in most fine chemical facilities. The process begins with the charging of 1-methyluracil, urea, potassium iodide, and TBHP into a reactor containing dioxane, followed by the controlled addition of sulfuric acid. The mixture is heated to approximately 75°C and maintained for 16 to 20 hours to ensure complete conversion to the 8-iodo intermediate (Compound I). Upon completion, the workup involves a standard aqueous wash sequence to remove inorganic salts and acid residues. The second stage involves dissolving the purified Compound I in acetone, adding DIPEA as an acid scavenger, and reacting with 1-bromo-2-butyne at 50°C.
- React 1-methyluracil with urea, an iodine source (e.g., KI), and an oxidant (e.g., TBHP) under acid catalysis to form the 8-iodo intermediate (Compound I).
- Perform a substitution reaction between Compound I and 1-bromo-2-butyne in the presence of a base to yield the final linagliptin intermediate (Compound II).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates into tangible strategic advantages beyond mere technical feasibility. The elimination of sodium nitrite, a regulated and potentially hazardous material, simplifies the logistics of raw material sourcing and storage. Facilities no longer need to invest in specialized containment systems for explosive reagents, thereby reducing capital expenditure and insurance costs. Furthermore, the reliance on commodity chemicals like urea and 1-methyluracil ensures a stable and resilient supply base, mitigating the risks associated with vendor lock-in or geopolitical disruptions in the supply of exotic reagents.
- Cost Reduction in Manufacturing: The consolidation of multiple reaction steps into a concise two-step sequence inherently drives down manufacturing costs. By removing the need for intermediate isolations, reductions, and separate iodination steps, the process significantly reduces labor hours, solvent usage, and energy consumption. The high yields reported in the patent examples suggest a substantial improvement in material efficiency, meaning less raw material is wasted per kilogram of finished product. This operational efficiency allows for a more competitive pricing structure for the final pharmaceutical intermediate, enabling better margin management for downstream API manufacturers.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply chain continuity. The tolerance of the process to slight variations in temperature and stoichiometry reduces the likelihood of batch failures, which are a common cause of supply delays in complex organic synthesis. Additionally, the shorter cycle time—from raw material input to finished intermediate—means that inventory turnover is faster, allowing suppliers to respond more agilely to fluctuations in market demand. This reliability is critical for maintaining the continuous production schedules required by major pharmaceutical clients.
- Scalability and Environmental Compliance: As regulatory pressure mounts regarding waste disposal and carbon footprints, this green chemistry approach offers a distinct compliance advantage. The reduction in hazardous waste streams, particularly those containing nitroso compounds and heavy metal residues often found in alternative catalytic systems, simplifies wastewater treatment protocols. The process is inherently scalable, having been demonstrated to work effectively with standard organic solvents and reagents that are easily sourced in bulk quantities. This scalability ensures that the transition from pilot plant to commercial tonnage production can be achieved with minimal process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities.
Q: How does this new method improve safety compared to traditional routes?
A: Traditional methods rely on sodium nitrite for nitrosation, which poses explosion risks and generates hazardous waste. This novel route utilizes oxidative coupling with TBHP and iodide sources, eliminating the need for unstable nitrous acid compounds and significantly enhancing operational safety.
Q: What are the purity levels achievable with this synthetic pathway?
A: The patented process demonstrates exceptional control over impurities, achieving product purity of up to 99% after simple recrystallization steps. The mild reaction conditions minimize side reactions, ensuring a clean impurity profile suitable for stringent pharmaceutical standards.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method is designed for scalability. It uses readily available raw materials like 1-methyluracil and urea, operates at moderate temperatures (60-90°C), and avoids complex purification steps, making it highly viable for commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Linagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving medications like linagliptin depends on a secure and high-quality supply of critical intermediates. Our technical team has thoroughly analyzed the oxidative coupling pathway described in CN107936023B and is fully prepared to execute this advanced chemistry at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project requirements are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest international standards.
We invite you to collaborate with us to leverage this innovative synthetic route for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in pharmaceutical intermediates can drive value and efficiency for your organization.
