Scalable PTC-Catalyzed Synthesis of Linagliptin Intermediates for Commercial API Production
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value antidiabetic agents, and Patent CN103319483A presents a transformative approach to producing the critical intermediate for Linagliptin. This intellectual property details a novel preparation method for Compound V, a key xanthine derivative, utilizing advanced phase transfer catalysis (PTC) to overcome significant bottlenecks in traditional manufacturing. By shifting away from conventional polar aprotic solvents, this technology enables a greener, more efficient synthesis that aligns perfectly with modern regulatory and environmental standards. The core innovation lies in the ability to execute multiple alkylation and substitution steps in non-water-soluble media, facilitating solvent recovery and drastically reducing hazardous waste generation. For global supply chain leaders, this represents a pivotal opportunity to secure a more sustainable and cost-effective source of high-purity pharmaceutical intermediates.
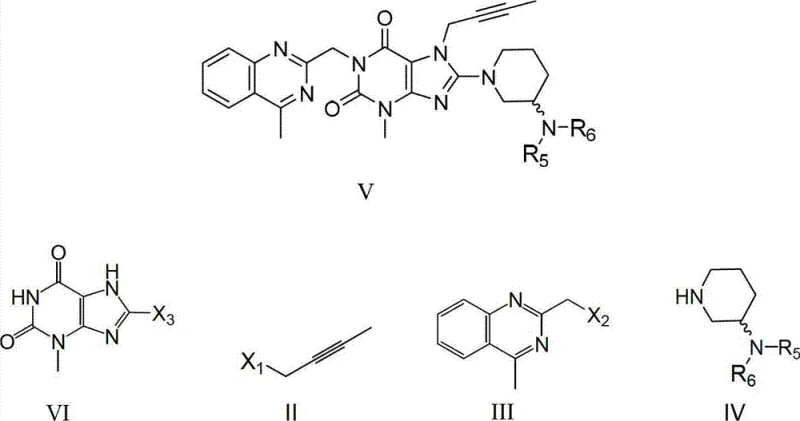
As a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this patent is crucial for evaluating its commercial viability. The process described allows for the industrial preparation of Linagliptin with excellent chemical and optical purity, addressing the growing demand for DPP-IV inhibitors. The methodology supports both stepwise and one-pot implementations, offering flexibility depending on existing infrastructure. By leveraging specific quaternary ammonium or phosphonium salts as catalysts, the reaction kinetics are enhanced, allowing for milder conditions and higher selectivity. This technical breakthrough not only improves yield but also ensures that the final product meets stringent quality specifications required for active pharmaceutical ingredient (API) production, making it an essential reference for any organization focused on cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Linagliptin intermediates has relied heavily on water-soluble solvents such as N,N-dimethylformamide (DMF), dimethyl sulfoxide (DMSO), or N-methylpyrrolidone (NMP). These traditional processes suffer from severe logistical and environmental drawbacks, primarily because the solvents are difficult to recover and recycle efficiently. In standard operations, intermediates must be isolated at each stage, often requiring the addition of water to precipitate the product, which generates massive volumes of contaminated wastewater. Furthermore, the purification of the final product typically necessitates column chromatography, a technique that is notoriously difficult to scale up for industrial production due to high solvent consumption and low throughput. These factors contribute to extended production cycles, elevated energy consumption, and a significant carbon footprint, rendering conventional routes economically and environmentally unsustainable for large-scale commercialization.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes phase transfer catalysts to enable reactions in non-water-soluble organic solvents like tetrahydrofuran (THF), toluene, or various acetates. This fundamental shift allows for the implementation of a one-pot synthesis strategy where multiple reaction steps occur sequentially in the same vessel without isolating intermediates. The ability to use recoverable solvents means that distillation can be employed to recycle the reaction medium, substantially lowering raw material costs and waste disposal fees. Moreover, the PTC system enhances regioselectivity, effectively suppressing the formation of unwanted O-alkylated impurities that are common in traditional methods. This results in a crude product with purity exceeding 95%, eliminating the need for complex chromatographic purification and streamlining the path from raw materials to the final high-purity linagliptin intermediates.
Mechanistic Insights into Phase Transfer Catalyzed Alkylation
The success of this synthetic route hinges on the precise application of phase transfer catalysts, which facilitate the transport of ionic species into the organic phase where the reaction occurs. The mechanism involves the formation of lipophilic ion pairs between the catalyst cation and the nucleophilic anions generated from the xanthine core and the amine components. This interaction significantly increases the reactivity of the nucleophiles towards the electrophilic alkylating agents, such as 1-bromo-2-butyne and 2-chloromethyl-4-methylquinazoline. By carefully selecting catalysts like tributylmethylammonium chloride or tetrabutylphosphonium bromide, the process achieves high conversion rates at moderate temperatures. The presence of co-catalysts, such as potassium iodide, further accelerates the reaction by enhancing the leaving group ability of the halides. This synergistic catalytic effect ensures that the N-alkylation at the 7-position and 1-position, followed by the C-substitution at the 8-position, proceeds with exceptional efficiency and minimal side reactions.
Impurity control is another critical aspect of this mechanism, particularly regarding the suppression of O-alkylation. In conventional basic conditions, the oxygen atoms on the xanthine ring can compete with nitrogen atoms for alkylation, leading to difficult-to-remove byproducts. The PTC environment modifies the local electronic environment and solvation shell around the nucleophile, favoring N-attack over O-attack. This selectivity is vital for maintaining the integrity of the heterocyclic core and ensuring that the final API meets rigorous pharmacopeial standards. The patent data indicates that this method can control O-alkylated impurities to less than 1%, a level that is achievable without extensive purification steps. This mechanistic advantage translates directly into operational simplicity, as the crude product can often be purified through simple crystallization or washing, rather than resource-intensive chromatography, thereby supporting the commercial scale-up of complex heterocycles.
How to Synthesize Linagliptin Intermediate Efficiently
The practical implementation of this technology involves a streamlined sequence that maximizes atom economy and operational safety. The process begins with the N7-alkylation of the xanthine precursor, followed by N1-alkylation with the quinazoline moiety, and concludes with the introduction of the chiral piperidine side chain. Detailed experimental data within the patent demonstrates that these steps can be telescoped into a single reactor setup, minimizing material handling and exposure. The use of common organic solvents and readily available bases like potassium carbonate further enhances the accessibility of this route for contract development and manufacturing organizations (CDMOs). For technical teams looking to adopt this methodology, the following guide outlines the critical operational parameters derived from the patent examples to ensure optimal yield and purity.
- Perform N7-alkylation of 8-bromo-3-methylxanthine with 1-bromo-2-butyne using a phase transfer catalyst and base in a non-polar solvent.
- Conduct N1-alkylation by adding 2-chloromethyl-4-methylquinazoline directly to the reaction mixture under reflux conditions.
- Complete the synthesis via C8-substitution with a protected 3-aminopiperidine derivative to yield the final xanthine precursor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this PTC-catalyzed process offers tangible strategic benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and enhanced supply security. By eliminating the need for intermediate isolation and column chromatography, the facility footprint required for production is significantly reduced, allowing for higher throughput within existing infrastructure. The ability to recover and reuse solvents creates a closed-loop system that mitigates the volatility of raw material pricing and reduces dependency on external waste management services. This resilience is crucial for maintaining consistent supply chains in the face of regulatory changes regarding solvent emissions and environmental compliance.
- Cost Reduction in Manufacturing: The economic impact of switching to this PTC-mediated route is profound, primarily driven by the elimination of expensive and difficult-to-recycle solvents like DMF and DMSO. Traditional processes incur high costs associated with solvent purchase, hazardous waste disposal, and energy-intensive drying steps. In contrast, the new method utilizes solvents like toluene or ethyl acetate, which are cheaper and can be distilled and reused multiple times without significant loss of quality. Additionally, the removal of column chromatography from the purification train saves substantial amounts of silica gel and elution solvents, which are major cost drivers in fine chemical synthesis. These cumulative savings result in a significantly lower cost of goods sold (COGS), providing a competitive edge in the global market for high-purity linagliptin intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex multi-step syntheses that require numerous unit operations and long cycle times. This innovative one-pot approach consolidates multiple reaction steps into a single vessel, drastically reducing the total production time and the risk of batch failure during transfers. The robustness of the PTC system ensures consistent reaction performance even with slight variations in raw material quality, leading to more predictable delivery schedules. Furthermore, the reagents and catalysts used are commercially available and stable, reducing the risk of supply disruptions associated with specialized or custom-synthesized reagents. This reliability makes the process ideal for securing long-term contracts and ensuring reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often reveals hidden challenges related to heat transfer and mixing, particularly in viscous solvent systems. The use of low-viscosity, non-polar solvents in this PTC method improves mixing efficiency and heat dissipation, making the reaction inherently safer and easier to scale to multi-ton quantities. From an environmental perspective, the reduction in waste generation aligns with Green Chemistry principles, helping manufacturers meet increasingly strict environmental regulations without costly retrofits. The minimized waste stream lowers the burden on effluent treatment plants and reduces the overall environmental footprint of the manufacturing site. This sustainability profile is increasingly valued by downstream partners and regulators, enhancing the marketability of the final API.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this technology compares to existing standards. Understanding these nuances is essential for R&D teams evaluating process feasibility and procurement officers assessing vendor capabilities. The answers highlight the specific advantages of phase transfer catalysis in the context of xanthine derivative synthesis.
Q: How does the PTC method improve solvent recovery compared to traditional DMF processes?
A: Traditional methods rely on water-soluble solvents like DMF or DMSO which are difficult to recycle. The PTC method enables the use of non-water-soluble solvents like toluene or THF, allowing for efficient distillation and reuse, significantly reducing waste.
Q: What is the impact of this technology on O-alkylation impurities?
A: The strategic use of phase transfer catalysts suppresses the formation of O-alkylated byproducts, controlling these impurities to below 1%, which simplifies downstream purification and eliminates the need for column chromatography.
Q: Is the one-pot synthesis suitable for large-scale industrial production?
A: Yes, the one-pot methodology minimizes unit operations by avoiding intermediate isolation. This reduces equipment requirements, shortens production cycles, and enhances overall process safety and scalability for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Linagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the pharmaceutical sector. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the PTC-catalyzed Linagliptin route are translated into reality with precision. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of intermediate supports the safety and efficacy of the final drug product. Our infrastructure is designed to handle complex heterocyclic chemistry safely and efficiently, positioning us as a strategic partner for your long-term supply needs.
We invite you to engage with our technical procurement team to discuss how this improved synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic potential of switching to this greener manufacturing process. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume needs. Together, we can optimize your supply chain for Linagliptin production, ensuring reliability, quality, and sustainability in every shipment.
