Advanced Lenalidomide Manufacturing: Optimizing Yield and Purity for Global Pharmaceutical Supply Chains
Advanced Lenalidomide Manufacturing: Optimizing Yield and Purity for Global Pharmaceutical Supply Chains
The pharmaceutical landscape for oncology treatments continues to evolve, with Lenalidomide standing as a cornerstone therapy for multiple myeloma and myelodysplastic syndromes. As global demand for this critical active pharmaceutical ingredient (API) surges, the efficiency and robustness of its manufacturing process become paramount for supply chain stability. Patent CN102838586A introduces a refined synthetic methodology that addresses historical bottlenecks in Lenalidomide production, specifically targeting yield enhancement and operational simplicity. This technical disclosure outlines a two-step sequence involving a condensation reaction followed by a catalytic reduction, operating under mild conditions that avoid the extremes of high temperature and high pressure often associated with traditional heterocyclic synthesis. By optimizing the post-treatment techniques, this approach not only secures a total yield of approximately 65% but also guarantees a finished product purity exceeding 99.9%, meeting the stringent regulatory standards required for injectable and oral solid dosage forms in major markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in patent WO2010100476, have historically relied on complex purification sequences that inadvertently compromise overall process efficiency. A significant drawback in these conventional routes involves the intricate after-treatment technologies employed after each reaction step, particularly during the final purification stage. In many legacy processes, the Lenalidomide molecule is prone to forming hydrochloride salts during repeated purification cycles, a phenomenon that necessitates additional neutralization and conversion steps. These extra operations not only extend the production cycle time significantly but also introduce opportunities for product loss, thereby depressing the total recovery rate to levels often below 60%. Furthermore, the reliance on harsh conditions or cumbersome isolation procedures in older methods increases the generation of hazardous waste and complicates the scalability of the process, making it less attractive for large-scale commercial manufacturing where throughput and environmental compliance are critical KPIs for procurement teams.
The Novel Approach
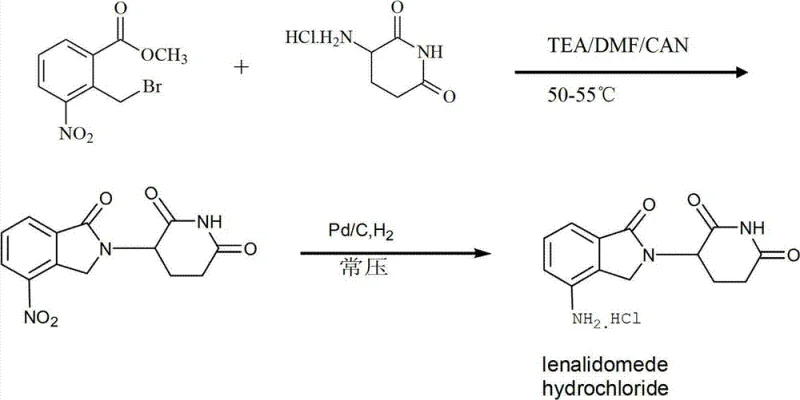
The innovative strategy presented in CN102838586A fundamentally reengineers the workflow by simplifying the isolation and purification protocols while maintaining rigorous quality control. Instead of engaging in complex salt formation and conversion cycles, this novel approach utilizes a direct precipitation and washing technique immediately following the condensation reaction. By cooling the reaction solution to room temperature and introducing it into water under stirring, the intermediate precipitates efficiently, allowing for straightforward filtration. The subsequent washing steps with water and alcohol reagents effectively remove soluble impurities and residual reactants without degrading the sensitive isoindoline backbone. This streamlined workflow minimizes unit operations, reduces solvent consumption, and critically, preserves the integrity of the product, leading to a marked improvement in total yield. The visual representation of this optimized pathway highlights the direct transformation from the nitro-intermediate to the final amino-product, underscoring the elegance of the design.
Mechanistic Insights into Condensation and Catalytic Hydrogenation
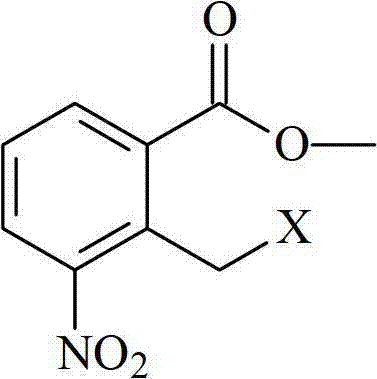
The core of this synthesis lies in the precise control of the nucleophilic substitution reaction between 3-aminopiperidine-2,6-dione hydrochloride and the halogenated benzoate derivative. The reaction mechanism proceeds via an SN2-type displacement where the primary amine of the piperidine dione attacks the halomethyl group of the aromatic substrate. To facilitate this, the process employs a weakly alkaline environment, typically generated in situ by adding acid binding agents such as triethylamine, sodium bicarbonate, or N,N-diisopropylethylamine. These bases serve a dual purpose: they neutralize the hydrochloric acid released during the reaction, preventing the protonation of the nucleophilic amine which would render it inactive, and they maintain the pH within a range that favors the formation of the isoindoline ring system. The choice of solvent, often a mixture of DMF and acetonitrile, is crucial as it provides the necessary polarity to dissolve the ionic starting materials while stabilizing the transition state of the substitution reaction at moderate temperatures ranging from 50°C to 55°C.
Following the formation of the nitro-intermediate, 3-(7-nitro-3-oxo-1H-isoindol-2-yl)piperidine-2,6-dione, the process transitions to a catalytic hydrogenation step to reduce the nitro group to the corresponding amine. This transformation is achieved using a palladium on carbon (Pd/C) catalyst under a hydrogen atmosphere at atmospheric pressure, a condition that is significantly safer and more energy-efficient than high-pressure hydrogenation methods. The structural integrity of the starting halogenated material, as depicted in the chemical diagrams, is vital for the success of the initial coupling; the presence of the electron-withdrawing nitro group ortho to the halomethyl moiety activates the benzylic position for nucleophilic attack. The subsequent reduction must be carefully monitored to ensure complete conversion of the nitro group without affecting the lactam carbonyls or causing hydrogenolysis of the newly formed C-N bond. The final crystallization at -20°C leverages the differential solubility of the product versus impurities, ensuring that the final API meets the >99.9% purity specification required for clinical applications.
How to Synthesize Lenalidomide Efficiently
Implementing this synthesis route requires strict adherence to the specified stoichiometry and temperature profiles to maximize the yield of the nitro-intermediate and the final API. The process begins with the preparation of the reaction mixture in a dry vessel under nitrogen protection to prevent moisture interference, followed by the controlled addition of the acid binding agent to manage exotherms. Detailed standard operating procedures for the filtration, washing, and drying steps are essential to replicate the high purity outcomes reported in the patent data. For a comprehensive breakdown of the specific reagent quantities, reaction times, and equipment specifications required to execute this protocol safely and effectively, please refer to the standardized synthesis guide provided below.
- Condense 3-aminopiperidine-2,6-dione hydrochloride with 2-halomethyl-3-nitro-methyl benzoate in DMF/acetonitrile with triethylamine at 50-55°C.
- Isolate the nitro-intermediate by pouring into water, filtering, and washing with water and alcohol reagents.
- Perform catalytic hydrogenation using Pd/C to reduce the nitro group, followed by concentration and crystallization at -20°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this optimized synthesis route offers substantial strategic benefits for procurement managers and supply chain directors seeking to mitigate risk and control costs in the production of oncology intermediates. The elimination of high-temperature and high-pressure requirements translates directly into reduced capital expenditure on specialized reactor vessels and safety systems, allowing for production in standard glass-lined or stainless steel facilities that are more readily available in the global CDMO network. Furthermore, the simplified workup procedure, which relies on basic filtration and washing rather than complex chromatography or multi-step salt conversions, drastically reduces the consumption of organic solvents and the volume of hazardous waste generated. This reduction in material intensity not only lowers the direct cost of goods sold (COGS) but also streamlines the environmental permitting process, ensuring uninterrupted supply continuity even in regions with strict environmental regulations.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the significant simplification of the downstream processing stages. By avoiding the formation of hydrochloride salts during purification, the method eliminates the need for additional reagents and processing time associated with salt breaking and freebasing steps. This reduction in unit operations leads to a direct decrease in labor costs and utility consumption, such as steam for heating and chilled water for cooling cycles. Additionally, the improved total yield of approximately 65% means that less raw material is required to produce the same amount of finished API, effectively lowering the input cost per kilogram of Lenalidomide produced and enhancing the overall margin profile for the manufacturing partner.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable starting materials, such as 3-aminopiperidine-2,6-dione hydrochloride and substituted benzoates, which are commercially sourced from established chemical suppliers. The robustness of the reaction conditions, which tolerate slight variations in temperature and mixing without compromising product quality, reduces the risk of batch failures that can lead to costly delays. Moreover, the ability to perform the hydrogenation step at atmospheric pressure removes the dependency on high-pressure hydrogen infrastructure, which can be a bottleneck in some manufacturing sites, thereby increasing the number of qualified suppliers capable of producing this critical intermediate.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial tonnage is facilitated by the linear nature of the reaction kinetics and the simplicity of the isolation steps. The use of common solvents like DMF, acetonitrile, and methanol allows for efficient solvent recovery and recycling systems, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing operation. The high purity of the crude product prior to recrystallization minimizes the load on wastewater treatment facilities, as fewer organic impurities need to be degraded or removed. This environmental efficiency ensures long-term regulatory compliance and protects the supply chain from potential shutdowns due to environmental violations, securing a steady flow of high-quality Lenalidomide for the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Lenalidomide synthesis technology. These answers are derived directly from the experimental data and claims within patent CN102838586A, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding these nuances is critical for R&D teams planning technology transfer and for procurement specialists negotiating supply agreements based on process capability.
Q: What is the primary advantage of this Lenalidomide synthesis method over prior art?
A: The method described in CN102838586A eliminates complex purification steps found in previous patents (like WO2010100476), specifically avoiding the formation of hydrochloride salts during purification which previously caused yield loss. This results in a total yield of approximately 65% with purity exceeding 99.9%.
Q: What are the critical reaction conditions for the condensation step?
A: The condensation reaction requires weakly alkaline conditions, typically achieved using acid binding agents like triethylamine or sodium bicarbonate. The reaction is conducted in solvents such as DMF or acetonitrile at temperatures ranging from 20°C to the reflux temperature, optimally around 50-55°C.
Q: How is the final purity of Lenalidomide ensured in this process?
A: High purity (>99.9%) is achieved through a specific recrystallization protocol. The crude product is dissolved in methanol or acetonitrile, heated to reflux, hot-filtered, and then allowed to crystallize at low temperatures (-20°C), effectively removing impurities without needing complex chromatographic separations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lenalidomide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex oncology drugs like Lenalidomide depends on a partnership grounded in technical excellence and supply chain reliability. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify identity, assay, and impurity profiles against the highest international pharmacopeial standards. Our facility is equipped to handle the specific solvent systems and catalytic hydrogenation requirements outlined in this patent, guaranteeing consistent batch-to-batch quality.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain to achieve significant operational efficiencies. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this methodology for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring that your development programs proceed without interruption and with the highest quality materials available in the market.
