Advanced 5-Step Synthetic Route for High-Purity Lenalidomide Intermediates and Commercial Scale-Up
Advanced 5-Step Synthetic Route for High-Purity Lenalidomide Intermediates and Commercial Scale-Up
The pharmaceutical landscape for immunomodulatory drugs has been significantly shaped by the introduction of lenalidomide, a potent agent approved for treating multiple myeloma and myelodysplastic syndromes. As detailed in patent CN103497175A, a novel and highly efficient method for preparing this critical active pharmaceutical ingredient (API) has been developed, addressing many of the longstanding challenges associated with its manufacture. This innovative process utilizes 3-amino-2-methylbenzoic acid as the primary starting material, offering a streamlined pathway that contrasts sharply with earlier, more cumbersome synthetic strategies. For R&D directors and procurement specialists seeking a reliable lenalidomide intermediate supplier, understanding the technical nuances of this route is essential for securing a stable supply chain. The method promises not only high synthesis efficiency but also superior purity profiles, which are paramount for meeting stringent regulatory requirements in global markets.
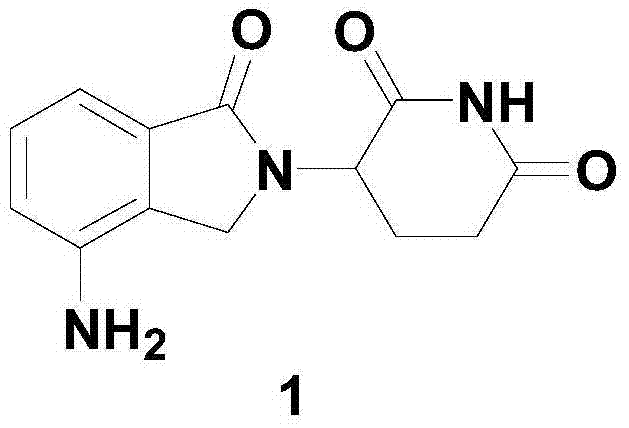
Lenalidomide, chemically known as 3-(7-amino-3-oxo-1H-isoindol-2-yl)piperidine-2,6-dione, represents a significant evolution from its predecessor thalidomide, offering enhanced anticancer potential with a improved safety profile regarding teratogenicity. However, the industrial realization of such complex heterocyclic compounds often faces hurdles related to yield, safety, and environmental impact. The disclosed invention provides a robust solution by simplifying the technological operation and ensuring mild reaction conditions throughout the synthesis. This approach effectively mitigates the risks associated with high-pressure or high-temperature reactions, thereby enhancing production safety. Furthermore, the absence of difficult-to-separate compounds in the preparation process means that the final product can be obtained with high purity without resorting to expensive and time-consuming purification techniques, making it an ideal candidate for cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of lenalidomide and its precursors has been plagued by complex preparation processes that involve multiple steps with low overall atom economy. Traditional routes often necessitate the use of hazardous reagents, extreme reaction conditions, and specialized equipment that increases capital expenditure and operational risk. A significant bottleneck in many legacy methods is the generation of by-products that are structurally similar to the target molecule, making separation extremely difficult and often requiring column chromatography, which is impractical for multi-tonne production. These inefficiencies lead to high production costs and extended lead times, creating vulnerabilities in the supply chain for high-purity lenalidomide. Additionally, the environmental burden of waste disposal from such inefficient processes has become a major concern for modern chemical manufacturers aiming for sustainability.
The Novel Approach
In stark contrast, the method described in CN103497175A introduces a rational design that prioritizes simplicity and scalability. The core innovation lies in the strategic functionalization of the aromatic ring followed by a clean cyclization step. As illustrated in the comprehensive reaction scheme below, the process flows logically from esterification to protection, halogenation, cyclization, and finally deprotection. This linear progression minimizes the handling of unstable intermediates and reduces the cumulative loss of material at each stage. The use of readily available starting materials ensures that the supply chain remains resilient against market fluctuations. Moreover, the ability to purify intermediates via simple recrystallization rather than chromatography is a game-changer for commercial scale-up of complex pharmaceutical intermediates, drastically reducing solvent consumption and processing time while maintaining exceptional product quality.
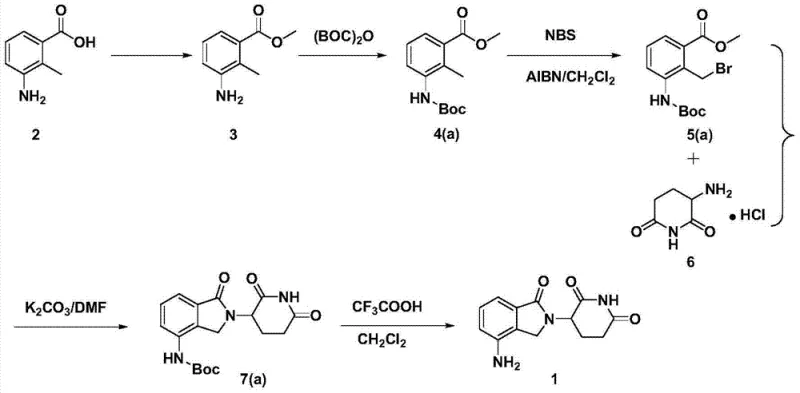
Mechanistic Insights into the 5-Step Synthetic Pathway
The success of this synthetic route relies on precise control over reaction mechanisms at each stage, beginning with the esterification of 3-amino-2-methylbenzoic acid. In this initial step, the carboxylic acid group is activated by an acid catalyst, such as p-toluenesulfonic acid or sulfuric acid, facilitating nucleophilic attack by a monohydric alcohol like methanol. This transformation is crucial as it converts the polar acid into a more lipophilic ester, which improves solubility in organic solvents for subsequent steps. Following esterification, the amino group is protected using agents like di-tert-butyl dicarbonate (Boc2O) or benzyl chloroformate. This protection is mechanistically vital to prevent the nucleophilic amino group from interfering with the subsequent radical halogenation, ensuring regioselectivity at the benzylic position. The choice of protecting group also influences the final deprotection strategy, offering flexibility between acidolysis and hydrogenolysis depending on the specific process requirements.
The subsequent halogenation step employs a radical mechanism, typically utilizing N-bromosuccinimide (NBS) in the presence of a radical initiator like azobisisobutyronitrile (AIBN). This reaction selectively substitutes a hydrogen atom on the benzylic methyl group with a bromine atom, creating a highly reactive electrophilic center. This bromomethyl intermediate is then poised for the critical cyclization reaction with 3-amino-2,6-piperidinedione hydrochloride. In the presence of a base such as potassium carbonate, the amino group of the piperidine dione acts as a nucleophile, displacing the bromide ion in an SN2-type substitution to form the isoindoline ring system. This cyclization constructs the core scaffold of lenalidomide with high fidelity. Finally, the removal of the protecting group reveals the free amino functionality, completing the synthesis. The meticulous control of impurities throughout these mechanistic steps ensures that the final high-purity lenalidomide meets rigorous pharmacopeial standards without the need for complex downstream processing.
How to Synthesize Lenalidomide Efficiently
The synthesis of lenalidomide via this patented route offers a clear, step-by-step protocol that balances chemical efficiency with operational practicality. By adhering to the specific molar ratios and temperature controls outlined in the patent, manufacturers can achieve consistent results with minimal batch-to-batch variation. The process is designed to be robust, tolerating slight variations in reagent quality while still delivering high yields. For technical teams looking to implement this chemistry, the detailed standardized synthesis steps provided below serve as a foundational guide for process optimization and validation. This structured approach ensures that the transition from laboratory scale to pilot plant and eventually to full commercial production is seamless and predictable.
- Perform esterification of 3-amino-2-methylbenzoic acid with methanol using an acid catalyst to form the methyl ester.
- Protect the amino group using di-tert-butyl dicarbonate (Boc2O) or benzyl chloroformate to prevent side reactions.
- Execute radical halogenation on the methyl group using N-bromosuccinimide (NBS) and an initiator like AIBN.
- Conduct cyclization by reacting the brominated intermediate with 3-amino-2,6-piperidinedione hydrochloride in the presence of a base.
- Finalize the synthesis by removing the protecting group via acidolysis or hydrogenation to yield lenalidomide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic methodology offers profound advantages that directly address the pain points of procurement managers and supply chain heads. The primary benefit stems from the utilization of commodity chemicals as starting materials, which are abundantly available in the global market. This accessibility eliminates the dependency on exotic or proprietary reagents that often create bottlenecks and price volatility in the supply chain. Furthermore, the simplified operational workflow reduces the demand for highly specialized labor and complex equipment, translating into significant operational expenditure savings. The elimination of column chromatography not only speeds up production cycles but also drastically reduces the volume of organic solvents required, aligning with green chemistry principles and reducing waste disposal costs. These factors collectively contribute to a more resilient and cost-effective supply chain for pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom economy and the avoidance of expensive purification technologies. By replacing column chromatography with recrystallization, the process significantly lowers the consumption of silica gel and large volumes of elution solvents, which are major cost drivers in fine chemical synthesis. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling, further driving down utility costs. The high yields reported in the patent examples indicate that raw material utilization is optimized, minimizing waste and maximizing the output per unit of input. This efficiency allows for a substantial reduction in the cost of goods sold (COGS), providing a competitive edge in the pricing of the final API.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of robust and well-understood chemical transformations that are less prone to failure. The starting material, 3-amino-2-methylbenzoic acid, is a stable solid that can be sourced from multiple vendors, mitigating the risk of single-source dependency. The process does not require moisture-sensitive reagents or inert atmosphere conditions for every step, simplifying logistics and storage requirements. This robustness ensures that production schedules can be maintained even under fluctuating environmental conditions or minor supply disruptions. Consequently, partners can rely on a steady flow of high-purity lenalidomide intermediates, ensuring that downstream drug formulation timelines are met without delay.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but this route is inherently scalable due to its homogeneous reaction phases and manageable exotherms. The absence of dangerous or virulent reagents simplifies the permitting process and reduces the need for extensive safety infrastructure, accelerating the timeline for plant commissioning. From an environmental standpoint, the reduced solvent usage and the ability to recycle mother liquors from recrystallization steps lower the facility's environmental footprint. This alignment with environmental, social, and governance (ESG) goals is increasingly important for multinational corporations seeking sustainable partners. The process facilitates the commercial scale-up of complex pharmaceutical intermediates while maintaining strict adherence to environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this lenalidomide synthesis route. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for evaluating the feasibility of adopting this technology within your existing manufacturing framework. The insights provided here aim to clarify the operational benefits and technical capabilities of the process.
Q: What are the key advantages of this lenalidomide synthesis route compared to conventional methods?
A: This novel process utilizes readily available starting materials like 3-amino-2-methylbenzoic acid and avoids complex purification steps such as column chromatography. The reaction conditions are mild, eliminating the need for special equipment, and the intermediates can often be purified via simple recrystallization, significantly enhancing operational safety and efficiency.
Q: How does this method ensure high purity for pharmaceutical applications?
A: The process is designed to minimize the formation of difficult-to-separate impurities. By employing specific protecting groups like Boc or Cbz and optimizing the cyclization conditions, the route ensures that both intermediates and the final lenalidomide product can achieve high purity standards suitable for API manufacturing through straightforward workup procedures.
Q: Is this synthetic pathway suitable for large-scale industrial production?
A: Yes, the method is specifically engineered for scalability. It avoids dangerous or virulent reagents and does not require specialized reaction vessels. The simplicity of the operational steps, combined with the use of common solvents and catalysts, makes it highly adaptable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lenalidomide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the production of life-saving oncology medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical companies. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation. Our facility is equipped to handle the specific requirements of the lenalidomide synthesis route, from the initial esterification to the final crystallization, guaranteeing consistency and quality in every batch we deliver to our clients.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can be integrated into your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this more efficient methodology. We encourage you to request specific COA data and route feasibility assessments to verify our capabilities against your internal standards. Let us collaborate to optimize your production of lenalidomide, ensuring a secure and cost-effective supply of this vital therapeutic agent for patients worldwide.
