Revolutionizing Somatostatin KE108 Production: A Safer, Hydrogen-Free Synthetic Route for Commercial Scale
Revolutionizing Somatostatin KE108 Production: A Safer, Hydrogen-Free Synthetic Route for Commercial Scale
The pharmaceutical landscape for somatostatin analogs is evolving rapidly, driven by the need for safer and more efficient manufacturing processes for complex peptides like KE108. Patent CN112538103B, published in late 2022, introduces a groundbreaking methodology for the preparation of somatostatin KE108, a potent pan-somatostatin receptor agonist with significant potential in oncology and endocrine disorder treatments. This technical disclosure moves away from traditional, hazard-prone synthetic routes that rely on hydrogenation, proposing instead a sophisticated solid-phase peptide synthesis (SPPS) strategy utilizing Alloc-protected amino acids. For R&D directors and supply chain leaders, this shift represents a critical optimization in process safety and scalability, eliminating the need for high-pressure hydrogen gas while maintaining high synthesis efficiency. The core innovation lies in the strategic protection of the D-Dab amino acid side chain, which facilitates a smoother cyclization workflow and significantly reduces operational risks associated with conventional catalytic hydrogenation steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex cyclic peptides like KE108 has been plagued by significant safety and efficiency bottlenecks, particularly during the deprotection phases. Traditional routes often employ Cbz (carboxybenzyl) protecting groups for the diamino butyric acid (Dab) residue, which necessitates the use of molecular hydrogen (H2) and heterogeneous metal catalysts for removal. This requirement introduces severe safety hazards, including explosion risks and the need for specialized high-pressure equipment, which complicates facility compliance and increases capital expenditure. Furthermore, the conventional sequence often involves synthesizing the majority of the chain, performing cyclization, and then adding the final tyrosine residue, leading to a fragmented and multi-step workflow that accumulates impurities and lowers overall yield. The reliance on hydrogenation also poses challenges in removing trace metal residues to meet stringent pharmaceutical standards, adding costly purification steps that delay time-to-market for clinical candidates.
The Novel Approach
The methodology disclosed in CN112538103B fundamentally restructures the synthetic logic by integrating an Alloc (allyloxycarbonyl) protecting group on the D-Dab side chain from the outset. This strategic choice enables the removal of the protecting group using a mild palladium-catalyzed reaction with phenylsilane, completely bypassing the need for dangerous hydrogen gas. By synthesizing the entire nine-amino acid sequence on the resin prior to cyclization, the process streamlines the workflow into a cohesive linear-to-cyclic transition. This "all-on-resin" elongation followed by solution-phase cyclization not only simplifies the operational protocol but also enhances the chemical integrity of the peptide backbone. The elimination of hydrogenation steps translates directly into a safer manufacturing environment, reducing the regulatory burden on facility safety audits and allowing for more flexible production scheduling without the constraints of high-pressure reactor availability.
Mechanistic Insights into Alloc Deprotection and Head-to-Side-Chain Cyclization
The chemical elegance of this new process is best understood through the specific mechanistic interactions during the deprotection and cyclization stages. The use of tetrakis(triphenylphosphine)palladium(0) [Pd(PPh3)4] in conjunction with phenylsilane acts as a highly efficient scavenger system for the allyl group. In this catalytic cycle, the palladium(0) species coordinates with the allyl moiety of the Alloc group, forming a pi-allyl palladium complex that is subsequently attacked by the hydride source (phenylsilane). This reaction cleanly releases the free amine on the D-Dab side chain while generating volatile byproducts that are easily removed, ensuring minimal contamination of the peptide resin. This orthogonality is crucial because it leaves other acid-labile protecting groups (like Boc and tBu) intact during the deprotection phase, preserving the structural fidelity of the growing peptide chain until the final global cleavage.
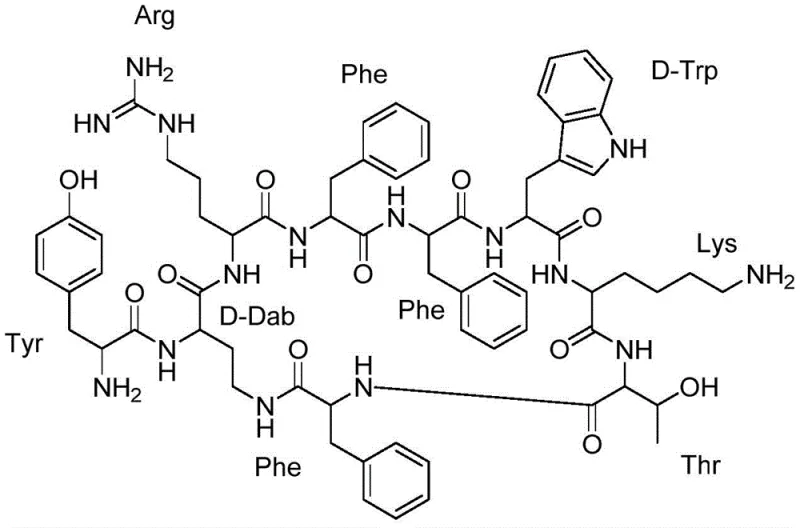
Following the deprotection, the cyclization mechanism relies on the precise activation of the carboxyl group of the N-terminal Phenylalanine (Phe) to react with the newly exposed epsilon-amino group of the D-Dab residue. The patent highlights that standard coupling reagents often fail in this macrocyclization due to steric hindrance and conformational constraints. However, the combination of PyAOP (7-azabenzotriazol-1-yloxy)tris(pyrrolidino)phosphonium hexafluorophosphate) and HOAt (1-hydroxy-7-azabenzotriazole) in the presence of N-methylmorpholine (NMM) provides the necessary activation energy and suppression of racemization. This specific reagent system facilitates the formation of the lactam bridge between the side chain and the backbone, locking the peptide into its bioactive conformation. The success of this cyclization is paramount, as comparative examples in the patent demonstrate that substituting PyAOP with HCTU or HATU results in complete failure to cyclize, underscoring the sensitivity of this step to reagent selection.
How to Synthesize Somatostatin KE108 Efficiently
Implementing this synthesis route requires strict adherence to the optimized stoichiometry and solvent systems described in the patent to ensure reproducibility and high purity. The process begins with the swelling of chloride resin and the anchoring of the first Fmoc-Phe-OH, followed by iterative cycles of deprotection and coupling using a 4-fold excess of protected amino acids. Critical attention must be paid to the Alloc deprotection step, where the ratio of palladium catalyst to phenylsilane must be carefully controlled to prevent incomplete deprotection or side reactions. Once the linear peptide is cleaved from the resin using hexafluoroisopropanol (HFIP), the crude material is subjected to the specific PyAOP-mediated cyclization conditions at room temperature for approximately 24 hours. The detailed standardized synthesis steps, including exact reagent quantities and reaction times for each phase of the production, are outlined below.
- Load Fmoc-Phe-OH onto chloride resin and perform sequential Fmoc-SPPS elongation using protected amino acids including Fmoc-D-Dab(Alloc)-OH.
- Remove the Alloc protecting group from the D-Dab side chain using tetrakis(triphenylphosphine)palladium(0) and phenylsilane in DCM, avoiding hazardous hydrogen gas.
- Cleave the fully protected linear peptide from the resin using hexafluoroisopropanol (HFIP) and dichloromethane.
- Perform head-to-side-chain cyclization between the D-Dab amine and Phe carboxyl using PyAOP/HOAt and N-methylmorpholine.
- Execute global deprotection using a TFA-based cleavage cocktail (TFA/Water/Phenol/TIPS) followed by HPLC purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this hydrogen-free synthesis route offers substantial strategic benefits beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the safety infrastructure required for production. By eliminating the need for high-pressure hydrogen gas and the associated explosion-proof facilities, manufacturers can significantly reduce capital overhead and insurance costs. This reduction in operational complexity allows for more agile production scheduling and lowers the barrier for contract manufacturing organizations (CMOs) to adopt the process, thereby increasing the available supply base for this critical intermediate. Furthermore, the streamlined workflow reduces the total number of unit operations, which correlates directly with lower labor costs and reduced consumption of solvents and reagents per kilogram of final product.
- Cost Reduction in Manufacturing: The economic impact of removing hydrogenation cannot be overstated; it eliminates the need for expensive noble metal catalysts typically used in high-pressure reactors and the subsequent rigorous testing required to ensure residual metal levels comply with ICH Q3D guidelines. Additionally, the high efficiency of the PyAOP/HOAt cyclization system minimizes the formation of deletion sequences and linear byproducts, which reduces the load on downstream purification columns and improves the overall recovery yield of the final API. This efficiency gain translates to a lower cost of goods sold (COGS) and a more competitive pricing structure for the finished pharmaceutical product.
- Enhanced Supply Chain Reliability: The reliance on commercially available, stable reagents like phenylsilane and standard Fmoc-amino acids ensures a robust supply chain that is less susceptible to disruptions compared to processes requiring specialized gases or custom catalysts. The solid-phase nature of the initial synthesis allows for easy scaling from gram to kilogram quantities by simply increasing resin bed volume, providing a clear path for capacity expansion as clinical demand grows. This scalability ensures that partners can reliably meet the fluctuating demands of clinical trials and eventual commercial launch without the long lead times associated with retrofitting facilities for hazardous chemistry.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the process generates less hazardous waste by avoiding heavy metal catalysts and high-pressure gas emissions. The use of standard organic solvents like DCM and DMF, while requiring proper management, fits within established waste treatment protocols familiar to most fine chemical manufacturers. The simplified workup procedures, such as precipitation with ethyl ether and straightforward filtration, reduce the energy intensity of the process, aligning with modern green chemistry principles and helping corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel somatostatin synthesis pathway. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering clarity on why specific reagents and conditions are non-negotiable for success. Understanding these nuances is essential for technology transfer teams aiming to replicate the high purity and yield reported in the intellectual property.
Q: Why is the Alloc protecting group preferred over Cbz for KE108 synthesis?
A: The Alloc (allyloxycarbonyl) group allows for orthogonal deprotection using palladium catalysis and phenylsilane under mild conditions. Unlike the traditional Cbz group which requires hazardous hydrogen gas (H2) and metal catalysts for removal, the Alloc strategy eliminates explosion risks and simplifies the safety profile for large-scale manufacturing.
Q: What specific condensing agents are critical for the cyclization step?
A: Experimental data indicates that PyAOP combined with HOAt and N-methylmorpholine (NMM) is essential for successful cyclization. Comparative studies showed that common agents like HCTU, HATU, or DIC/Oxyma failed to achieve the necessary head-to-side-chain cyclization between D-Dab and Phe, resulting in no target product formation.
Q: How does this process impact the purity profile of the final API intermediate?
A: By utilizing a robust solid-phase synthesis followed by solution-phase cyclization and rigorous HPLC purification (using water/acetonitrile with 0.1% TFA), the process achieves high purity. The specific cleavage cocktail (TFA/Phenol/TIPS) effectively removes acid-labile protecting groups (Pbf, Boc, tBu) without degrading the sensitive peptide backbone.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Somatostatin KE108 Supplier
As the demand for targeted somatostatin therapies continues to rise, securing a manufacturing partner with deep expertise in complex peptide chemistry is vital for project success. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring innovative molecules like KE108 to market. Our state-of-the-art facilities are equipped to handle the specific requirements of solid-phase peptide synthesis and solution-phase cyclization, ensuring stringent purity specifications are met through our rigorous QC labs and advanced analytical capabilities. We understand the critical nature of timeline and quality in pharmaceutical development, and our team is dedicated to optimizing every step of the value chain to deliver superior results.
We invite you to collaborate with us to evaluate the feasibility of this advanced synthetic route for your specific pipeline needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that details how implementing this hydrogen-free process can optimize your budget without compromising quality. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our technical prowess can accelerate your journey from bench to bedside.
