Advanced Solid-Phase Synthesis of Somatostatin KE108: A Safer, Scalable Route for Global Supply Chains
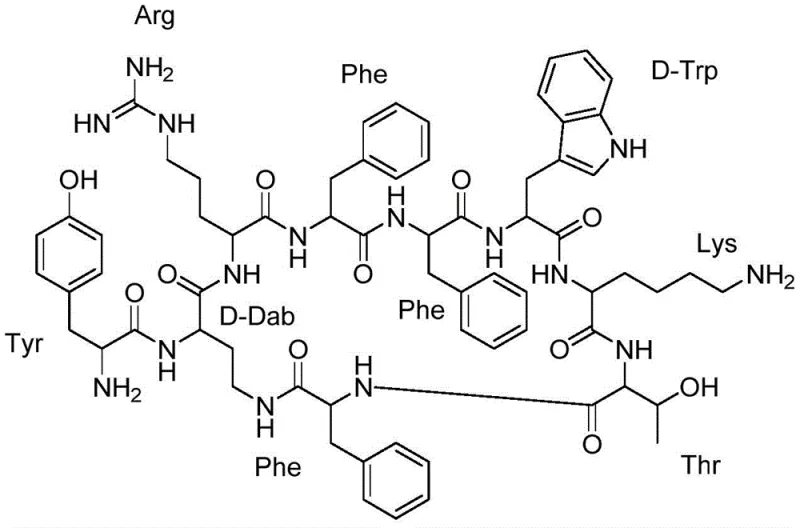
The pharmaceutical industry's demand for high-affinity somatostatin receptor ligands, particularly the pan-somatostatin antagonist KE108, has driven intense research into more efficient manufacturing protocols. Patent CN112538103B, published in late 2022, introduces a transformative solid-phase synthesis strategy that fundamentally alters the production landscape for this complex nonapeptide. Unlike traditional methods that rely on hazardous hydrogenation steps to remove benzyl-based protecting groups, this novel approach utilizes an Alloc (Allyloxycarbonyl) protection strategy on the critical D-Dab residue. This shift not only mitigates significant safety risks associated with high-pressure hydrogen gas but also streamlines the workflow by enabling orthogonal deprotection under mild conditions. For global supply chain leaders and R&D directors, this patent represents a pivotal advancement in cost reduction in peptide manufacturing, offering a route that is inherently safer, more scalable, and chemically robust for large-scale commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of somatostatin analogs like KE108 has been plagued by the reliance on Cbz (Carboxybenzyl) protecting groups for the side-chain amino functionality of diaminobutyric acid (Dab) residues. The removal of Cbz groups necessitates catalytic hydrogenation, typically employing palladium on carbon (Pd/C) under hydrogen pressure. This requirement introduces severe operational bottlenecks: it demands specialized high-pressure reactor equipment, rigorous safety protocols to manage explosive hydrogen gas, and complex filtration steps to remove trace heavy metal catalysts. Furthermore, hydrogenation conditions can sometimes lead to unwanted side reactions, such as the reduction of other sensitive functional groups within the peptide backbone, compromising the overall purity and yield. These factors collectively inflate the capital expenditure (CAPEX) and operational expenditure (OPEX) for manufacturers, making the commercial scale-up of complex peptide intermediates economically challenging and logistically fragile.
The Novel Approach
The methodology disclosed in CN112538103B elegantly circumvents these hurdles by substituting the Cbz group with an Alloc protecting group on the gamma-amino position of the D-Dab residue. This strategic modification enables the use of a palladium(0)-catalyzed deprotection system using phenylsilane as a scavenger, a reaction that proceeds smoothly at atmospheric pressure and room temperature. This eliminates the need for hydrogen gas entirely, drastically simplifying the equipment requirements and enhancing workplace safety. Additionally, the patent details a highly optimized cyclization protocol using PyAOP and HOAt, which ensures efficient ring closure between the Dab side chain and the Phe carboxyl group with minimal racemization. This holistic redesign of the synthetic route results in a process that is not only chemically superior but also perfectly aligned with the needs of a reliable somatostatin supplier aiming for consistent, high-volume output.
Mechanistic Insights into Alloc Deprotection and PyAOP-Mediated Cyclization
The core chemical innovation lies in the orthogonal deprotection of the Alloc group. In this mechanism, tetrakis(triphenylphosphine)palladium(0) acts as the catalyst, coordinating with the allyl moiety of the Alloc group to form a pi-allyl palladium complex. Phenylsilane then serves as a nucleophilic scavenger, attacking this complex to release the free amine on the D-Dab side chain while generating volatile byproducts that are easily removed. This mild condition is crucial because it preserves the integrity of other acid-labile protecting groups (like Boc and tBu) present on the peptide chain during the linear assembly phase. By avoiding the harsh reducing environment of hydrogenation, the process maintains the stereochemical purity of the chiral centers, which is paramount for the biological activity of the final somatostatin analog. This level of control over the deprotection step is a key differentiator for any high-purity peptide intermediate intended for clinical applications.
Following the linear assembly and deprotection, the cyclization step is critical for forming the constrained lactam ring that defines the bioactive conformation of KE108. The patent specifies the use of PyAOP (a uronium-type coupling reagent) in conjunction with HOAt (an additive that suppresses racemization) and N-methylmorpholine (NMM) as the base. This combination is superior to standard carbodiimides because PyAOP forms a highly reactive active ester that facilitates rapid amide bond formation between the sterically hindered side-chain amine of Dab and the alpha-carboxyl of the N-terminal Phe. The reaction is conducted in dilute dichloromethane solutions to favor intramolecular cyclization over intermolecular polymerization. This precise control over reaction kinetics ensures that the macrocyclization proceeds with high efficiency, minimizing the formation of linear oligomers and other difficult-to-remove impurities, thus simplifying downstream purification.
How to Synthesize Somatostatin KE108 Efficiently
The synthesis of this complex nonapeptide requires precise adherence to the solid-phase protocol outlined in the patent to ensure optimal yield and purity. The process begins with the loading of the first amino acid onto the resin, followed by iterative cycles of deprotection and coupling using Fmoc chemistry. The critical divergence from standard protocols occurs after the full linear sequence is assembled: the specific removal of the Alloc group prior to cleavage allows for a solution-phase cyclization that is both robust and scalable.
- Load Fmoc-Phe-OH onto chloride resin and sequentially condense protected amino acids including Fmoc-D-Dab(Alloc)-OH.
- Remove the Alloc protecting group using tetrakis(triphenylphosphine)palladium(0) and phenylsilane in DCM.
- Cleave the linear peptide from resin, then cyclize using PyAOP/HOAt/NMM to form the lactam ring between Dab and Phe.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hydrogen-free synthesis route offers profound strategic benefits beyond mere chemical elegance. By eliminating the requirement for high-pressure hydrogenation, manufacturers can significantly reduce the safety infrastructure costs associated with production facilities. This translates directly into a more resilient supply chain, as the process is less susceptible to regulatory shutdowns or safety incidents that often plague hydrogen-dependent operations. Furthermore, the use of readily available reagents like phenylsilane and standard palladium catalysts ensures that raw material sourcing remains stable and cost-effective, avoiding the volatility associated with specialized hydrogenation catalysts. This stability is essential for maintaining reducing lead time for high-purity peptide intermediates in a competitive global market.
- Cost Reduction in Manufacturing: The elimination of hydrogenation equipment and the associated safety protocols leads to substantial capital savings. Additionally, the milder reaction conditions reduce energy consumption and extend the lifespan of production vessels. The high efficiency of the PyAOP-mediated cyclization minimizes raw material waste by reducing the formation of byproducts, thereby lowering the overall cost of goods sold (COGS) without compromising on quality standards.
- Enhanced Supply Chain Reliability: The simplified process flow, characterized by fewer unit operations and the removal of hazardous gas handling, enhances operational continuity. Manufacturers can achieve faster batch turnover rates, ensuring a steady flow of material to downstream drug product facilities. This reliability is critical for pharmaceutical partners who require just-in-time delivery of key intermediates to maintain their own clinical or commercial production schedules.
- Scalability and Environmental Compliance: The absence of heavy metal hydrogenation catalysts simplifies waste treatment and disposal, aligning the process with increasingly stringent environmental regulations. The use of standard organic solvents like DCM and DMF, which are easily recovered and recycled, further supports sustainable manufacturing practices. This eco-friendly profile makes the process highly scalable from pilot batches to multi-kilogram commercial production without encountering the regulatory hurdles often associated with heavy metal residues.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Somatostatin KE108 based on the patented methodology. Understanding these details helps stakeholders evaluate the feasibility and advantages of integrating this supply source into their procurement strategy.
Q: Why is the Alloc protecting group preferred over Cbz for D-Dab in this synthesis?
A: The Alloc (Allyloxycarbonyl) group allows for orthogonal deprotection using mild palladium catalysis and silane scavengers, completely eliminating the need for hazardous high-pressure hydrogenation required by Cbz groups, thereby enhancing process safety and scalability.
Q: What specific condensing agents ensure high-efficiency cyclization for this peptide?
A: The patent specifies a combination of PyAOP (7-Azabenzotriazol-1-yloxy)tris(pyrrolidino)phosphonium hexafluorophosphate) and HOAt (1-Hydroxy-7-azabenzotriazole) with N-methylmorpholine, which significantly reduces racemization and improves cyclization yields compared to standard agents like HCTU or DIC.
Q: How does this process impact the purity profile of the final Somatostatin analog?
A: By avoiding harsh hydrogenation conditions and utilizing highly selective coupling reagents, the process minimizes side reactions and impurity formation, resulting in a crude peptide that is easier to purify to pharmaceutical-grade specifications via HPLC.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Somatostatin KE108 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and safe manufacturing processes for complex peptide therapeutics. Our technical team has extensively analyzed the innovations presented in CN112538103B and possesses the expertise to implement this advanced Alloc-protection strategy effectively. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets stringent purity specifications. Our state-of-the-art rigorous QC labs are equipped to verify the identity and purity of every batch, guaranteeing that the final product is free from the impurities often associated with older synthesis methods.
We invite pharmaceutical partners to collaborate with us to leverage this superior synthesis route for your somatostatin projects. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and discuss route feasibility assessments that can accelerate your development timelines and optimize your supply chain economics.
