Advanced Non-Nitration Synthesis of Phenylenediamine for High-Purity Polymer and Pharma Applications
Advanced Non-Nitration Synthesis of Phenylenediamine for High-Purity Polymer and Pharma Applications
The global demand for high-purity phenylenediamine derivatives, critical precursors for polyimides, aramid fibers, and pharmaceutical intermediates, has necessitated a paradigm shift in manufacturing technologies. Patent CN111763149A introduces a groundbreaking preparation method that fundamentally alters the synthetic landscape by replacing hazardous nitration processes with a safe, efficient rearrangement strategy. This innovation addresses the long-standing industry pain points of waste acid pollution and the explosive risks associated with polynitrobenzene byproducts. By utilizing phthalate esters as the initial raw materials, this technology offers a cleaner, more sustainable pathway to both m-phenylenediamine and p-phenylenediamine. For R&D directors and supply chain leaders, this represents a significant opportunity to enhance process safety profiles while securing a reliable phenylenediamine supplier capable of meeting stringent environmental regulations without compromising on yield or purity specifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of p-phenylenediamine and m-phenylenediamine has relied heavily on chlorobenzene or benzene feedstocks subjected to aggressive mixed acid nitration followed by reduction. This traditional workflow is fraught with severe operational hazards, including the generation of unstable polynitro byproducts that pose significant explosion risks during storage and handling. Furthermore, the nitration process produces vast quantities of waste acid, creating a massive environmental burden and escalating disposal costs for manufacturers. The subsequent reduction steps, whether utilizing iron powder, alkali sulfide, or catalytic hydrogenation, introduce their own complexities; iron powder reduction is technologically laggard with excessive solid waste, while hydrogenation requires expensive noble metal catalysts and high-pressure equipment. These factors collectively result in a fragile supply chain vulnerable to regulatory crackdowns and fluctuating raw material costs, making cost reduction in polymer additive manufacturing increasingly difficult under legacy protocols.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a two-step sequence beginning with the reaction of phthalate esters with hydroxylamine. This initial transformation converts readily available dimethyl isophthalate or dimethyl terephthalate into the corresponding benzene-dicarboxhydroxamic acid or its inorganic salt. This step is conducted under mild conditions, typically between 0 to 40°C, avoiding the extreme thermal stresses of nitration. The subsequent rearrangement of these hydroxamic acid derivatives directly yields the target phenylenediamine structure. This methodology effectively bypasses the formation of nitro-groups entirely, thereby eliminating the root cause of explosion hazards and waste acid generation. The process flow is significantly streamlined, offering a robust alternative for the commercial scale-up of complex pharmaceutical intermediates and specialty chemicals where safety and environmental compliance are paramount.
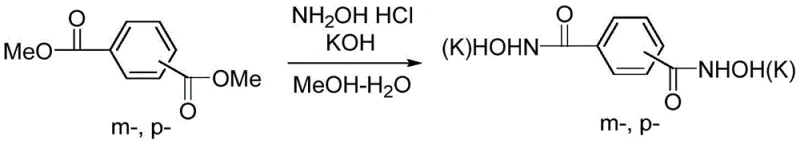
Mechanistic Insights into Hydroxamic Acid Rearrangement
The core of this technological breakthrough lies in the rearrangement mechanism of the di-hydroxamic acid intermediates. In the first stage, the nucleophilic attack of hydroxylamine on the ester carbonyls generates the di-potassium salt of the hydroxamic acid. This intermediate is stable enough to be isolated or carried forward directly, providing flexibility in process design. The patent highlights that the reaction can be performed in an alcohol-water solution, facilitating the dissolution of inorganic bases like potassium hydroxide while maintaining the solubility of the organic ester. The stoichiometry is carefully controlled, with a molar ratio of phthalate ester to hydroxylamine maintained between 1.0:2.0 and 1.0:2.5 to ensure complete conversion without excessive reagent waste. This precision in the first step is critical for minimizing impurities that could carry over into the final product, ensuring the high-purity phenylenediamine required for sensitive electronic or pharmaceutical applications.
The second stage involves the thermal rearrangement of the hydroxamate salt, a process akin to the Lossen rearrangement, which proceeds through an isocyanate-like transition state before hydrolyzing to the amine. The patent data reveals that the choice of solvent and catalyst dramatically influences the reaction kinetics and operating conditions. While the rearrangement can occur at elevated temperatures (up to 200°C) under pressure, the introduction of specific catalysts such as cesium carbonate allows the reaction to proceed at atmospheric pressure and lower temperatures (around 110°C). The solvent system, typically a mixture of acetonitrile and water, plays a dual role: water aids in dissolving the ionic hydroxamate salt, while the nitrile component acts as an activator for the rearrangement. Optimizing the volume ratio of acetonitrile to water is essential; too little water prevents dissolution, while too little nitrile slows the reaction rate. This mechanistic understanding allows for fine-tuning the process to maximize yield and minimize energy consumption.
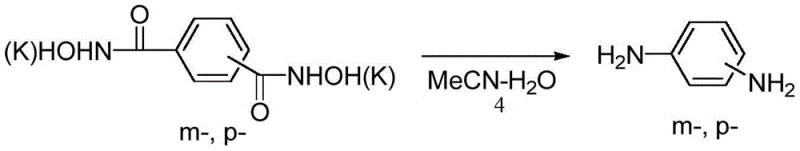
How to Synthesize Phenylenediamine Efficiently
Implementing this synthesis route requires careful attention to the preparation of the hydroxamic acid intermediate and the specific conditions of the rearrangement step. The process begins with the neutralization of hydroxylamine hydrochloride with an inorganic base, followed by reaction with the phthalate ester in a methanol-water medium. After the formation of the hydroxamate salt, the solvent is removed, and the solid residue is subjected to the rearrangement conditions. The versatility of the method allows for the production of either the free base or the inorganic salt of phenylenediamine depending on the post-reaction workup, offering downstream processing flexibility. For detailed operational parameters, including specific temperature ramps, stirring rates, and purification techniques like extraction and recrystallization, operators should refer to the standardized protocols derived from the patent examples.
- React phthalate ester (dimethyl isophthalate or terephthalate) with hydroxylamine hydrochloride and base (KOH) in methanol-water to form the di-potassium salt of benzene-dicarboxhydroxamic acid.
- Subject the hydroxamate salt to rearrangement in an acetonitrile-water solvent system, optionally using cesium carbonate catalyst at 100-170°C.
- Neutralize the reaction mixture with hydrochloric acid, extract with ethyl acetate, and purify to obtain high-purity m- or p-phenylenediamine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this non-nitration technology translates into tangible strategic benefits beyond mere chemical efficiency. The elimination of mixed acid nitration removes the need for specialized corrosion-resistant equipment and extensive safety infrastructure required to handle explosive polynitro compounds. This simplification of the plant requirements leads to substantial capital expenditure savings and reduces the ongoing operational costs associated with safety monitoring and regulatory compliance. Furthermore, the avoidance of heavy metal catalysts or iron powder sludge significantly lowers the cost of waste treatment, addressing one of the most persistent financial drains in traditional aromatic amine manufacturing. The supply chain becomes more resilient as the process relies on commodity chemicals like phthalate esters and hydroxylamine, which are widely available and less subject to the geopolitical volatility often seen with specialized nitrating agents.
- Cost Reduction in Manufacturing: The new process eliminates the need for expensive high-pressure hydrogenation reactors and the associated noble metal catalysts often required in traditional reduction steps. By enabling atmospheric pressure operation through the use of cesium carbonate catalysts, the technology drastically reduces energy consumption and equipment maintenance costs. Additionally, the removal of the nitration step means there is no need for costly waste acid neutralization and disposal systems, leading to significant operational expenditure savings. The overall simplification of the workflow reduces labor hours and increases batch throughput, further enhancing the economic viability of producing high-value diamines.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is inherently more stable because dimethyl terephthalate and dimethyl isophthalate are bulk commodities produced on a massive scale for the polyester industry. This abundance ensures that manufacturers are not held hostage by the supply constraints of niche nitrating agents or chlorinated benzenes. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent ratios without catastrophic failure, ensures consistent production schedules. This reliability is crucial for reducing lead time for high-purity phenylenediamine derivatives, allowing downstream customers in the aerospace and electronics sectors to maintain their own production timelines without interruption.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, transitioning smoothly from laboratory gram-scale experiments to multi-ton commercial production without fundamental changes in chemistry. The absence of toxic chlorine gas or sodium hypochlorite, which are required in some alternative non-nitration routes like Hofmann rearrangement, simplifies the environmental permitting process. The cleaner reaction profile results in a simpler impurity spectrum, making purification easier and reducing the volume of organic solvent waste generated during workup. This alignment with green chemistry principles positions manufacturers favorably against tightening global environmental regulations, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel phenylenediamine synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, safety improvements, and process flexibility. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing facilities or for procurement specialists assessing the quality consistency of suppliers adopting this method.
Q: How does this new method improve safety compared to traditional nitration?
A: Traditional methods utilize mixed acid nitration which generates hazardous polynitro byproducts prone to explosion and significant waste acid pollution. This novel route uses phthalate esters and hydroxylamine, completely eliminating the nitration step and associated explosion risks.
Q: What are the key advantages for large-scale manufacturing?
A: The process allows for operation at atmospheric pressure when using specific catalysts like cesium carbonate, reducing equipment costs. It also avoids the use of heavy metal reduction catalysts or iron powder, simplifying waste treatment and lowering environmental compliance costs.
Q: Can this method produce both meta and para isomers?
A: Yes, the method is versatile. By selecting either dimethyl isophthalate or dimethyl terephthalate as the starting raw material, manufacturers can selectively produce m-phenylenediamine or p-phenylenediamine with high yields exceeding 90%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylenediamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is critical for the future of the fine chemical industry. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the phthalate ester rearrangement method are translated into reliable supply solutions. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of phenylenediamine delivered. We understand that for R&D directors, consistency is key, and our commitment to process validation ensures that the impurity profiles remain stable across large-scale campaigns, supporting your downstream synthesis of polyimides or active pharmaceutical ingredients.
We invite you to collaborate with us to leverage these technological advancements for your specific application needs. Whether you require custom synthesis of m-phenylenediamine for agrochemical intermediates or p-phenylenediamine for high-performance polymers, our technical team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our adoption of non-nitration technologies can enhance your supply chain security and reduce your total cost of ownership.
