Advanced One-Step Synthesis of Benzothiadiazole Heterocycles for Commercial Scale-Up
Advanced One-Step Synthesis of Benzothiadiazole Heterocycles for Commercial Scale-Up
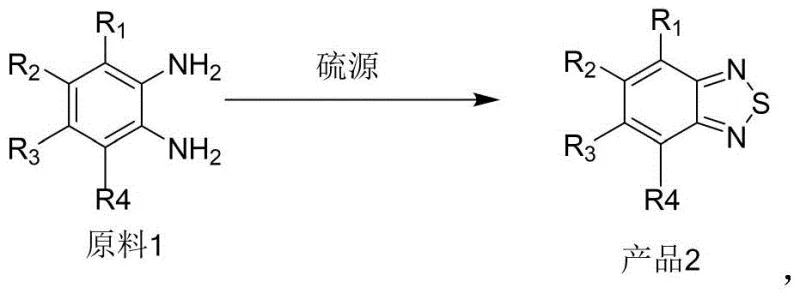
The chemical industry continuously seeks more efficient pathways for constructing heterocyclic scaffolds essential to modern technology. Patent CN115448892B introduces a transformative methodology for synthesizing benzothiadiazole heterocyclic compounds, a class of molecules critical in dye industries, optoelectronic materials, and pharmaceutical applications. This innovation leverages diethylaminosulfur trifluoride (DAST) as a superior sulfur source to convert o-phenylenediamine and its analogues into the target heterocycles through a streamlined one-step reaction. By shifting away from traditional harsh reflux conditions to a mild room-temperature protocol, this technology addresses long-standing inefficiencies in yield and operational complexity, offering a robust solution for the reliable agrochemical intermediate supplier and fine chemical manufacturers seeking process intensification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzothiadiazole core has relied on energetically demanding processes that hinder operational efficiency and safety. Traditional protocols often utilize thionyl chloride as the sulfur source, necessitating prolonged heating under reflux for up to 14 hours to achieve moderate conversion rates around 84 percent. Alternative routes involving non-commercial precursors like N-(2-fluorophenyl)-N-(trimethylsilyl)-λ4-sulfonamide suffer from prohibitively low yields of merely 23 percent due to the complexity of reagent preparation. Furthermore, pyrolysis methods generate excessive by-products with yields dropping to 20 percent, creating significant downstream purification burdens. These legacy techniques impose heavy energy costs and limit the throughput required for cost reduction in electronic chemical manufacturing, making them increasingly obsolete for modern high-volume production demands.
The Novel Approach
The patented methodology revolutionizes this landscape by employing cheap and easily available diethylaminosulfur trifluoride as the sulfur source, enabling a rapid cyclization at ambient temperatures. This approach eliminates the need for extended thermal input, reducing reaction times from hours to mere minutes while boosting yields to impressive levels such as 92 percent in model systems. The simplicity of using industrial-grade o-phenylenediamine derivatives combined with commercially accessible fluorinating agents creates a seamless workflow that drastically simplifies purification. This shift not only enhances the economic viability of producing high-purity OLED material precursors but also aligns with green chemistry principles by minimizing energy consumption and waste generation associated with high-temperature reflux operations.
Mechanistic Insights into DAST-Mediated Cyclization
The efficacy of this transformation lies in the unique reactivity of the sulfur-fluorine bonds within the DAST reagent, which facilitates a gentle yet effective dehydration-cyclization sequence. Mechanistically, the primary amine groups of the o-phenylenediamine substrate act as nucleophiles, attacking the electrophilic sulfur center of the DAST molecule to form an intermediate sulfonium species. This activation step is crucial as it lowers the energy barrier for the subsequent intramolecular ring closure, allowing the reaction to proceed spontaneously at temperatures between 25-40°C without external heating. The elimination of fluoride ions and amine by-products drives the equilibrium forward, ensuring high conversion rates even with sterically hindered substrates containing bulky substituents like tert-butyl or benzoyl groups.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in maintaining product integrity. Unlike harsh acidic or thermal conditions that can promote polymerization or decomposition of sensitive functional groups such as esters or nitriles, this room-temperature protocol preserves the structural fidelity of the starting material. The selectivity of DAST ensures that side reactions are minimized, resulting in a cleaner crude reaction profile that requires less aggressive chromatographic separation. This inherent purity advantage is particularly valuable for the commercial scale-up of complex polymer additives and pharmaceutical intermediates where trace impurities can critically affect downstream performance and regulatory compliance.
How to Synthesize Benzothiadiazole Efficiently
Implementing this synthesis route requires precise control over stoichiometry and solvent selection to maximize the benefits of the novel catalytic system. The standard procedure involves dissolving the o-phenylenediamine starting material in a chlorinated solvent like dichloromethane or a polar aprotic solvent like acetonitrile, followed by the controlled addition of the sulfur source. Maintaining a molar ratio of approximately 1.05:1 between the sulfur source and the substrate ensures complete conversion while minimizing excess reagent waste. The detailed standardized synthesis steps see the guide below, which outlines the specific workup procedures involving aqueous bicarbonate washes to neutralize acidic by-products and isolate the high-purity heterocyclic product.
- Dissolve the o-phenylenediamine starting material in a suitable solvent such as dichloromethane or acetonitrile.
- Add the sulfur source, preferably diethylaminosulfur trifluoride (DAST), to the reaction mixture at a molar ratio of approximately 1.05: 1.
- Stir the reaction at room temperature (25-40°C) for approximately 20 minutes, followed by aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain leaders, this patent represents a significant opportunity to optimize the cost structure and reliability of heterocyclic intermediate sourcing. The transition from energy-intensive reflux methods to a room-temperature process fundamentally alters the operational expenditure profile by removing the need for sustained heating infrastructure and reducing utility consumption. Furthermore, the reliance on widely available industrial reagents like o-phenylenediamine and DAST mitigates supply chain risks associated with specialized or custom-synthesized precursors, ensuring a steady flow of raw materials for continuous manufacturing campaigns.
- Cost Reduction in Manufacturing: The elimination of prolonged heating cycles and the use of inexpensive, commercially available reagents directly translate to lower production costs without compromising yield. By avoiding the need for exotic catalysts or difficult-to-source starting materials, manufacturers can achieve substantial cost savings in raw material procurement and energy usage. The simplified purification process further reduces solvent consumption and labor hours, contributing to a leaner and more economically competitive manufacturing model for high-value fine chemicals.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals such as o-phenylenediamine and standard fluorinating agents ensures that production is not bottlenecked by the availability of niche reagents. This robustness allows for flexible scheduling and rapid response to market demand fluctuations, significantly reducing lead time for high-purity heterocyclic intermediates. The stability of the reaction conditions also minimizes the risk of batch failures due to thermal runaway or equipment malfunction, providing a more predictable and secure supply pipeline for downstream customers.
- Scalability and Environmental Compliance: The mild reaction conditions and simple workup procedures make this process inherently scalable from laboratory benchtop to multi-ton industrial reactors. The reduction in energy demand and the avoidance of harsh corrosive reagents like thionyl chloride align with increasingly stringent environmental regulations, facilitating easier permitting and waste management. This sustainability advantage positions manufacturers as preferred partners for global clients seeking eco-friendly supply chains for advanced electronic and pharmaceutical materials.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology, derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is essential for R&D teams evaluating the feasibility of adopting this route for their specific derivative libraries. The answers reflect the proven capabilities of the method across a diverse range of substrates and conditions.
Q: What are the advantages of using DAST over thionyl chloride for benzothiadiazole synthesis?
A: Using DAST allows the reaction to proceed at room temperature in significantly less time (20 minutes vs 14 hours) compared to thionyl chloride reflux methods, while achieving higher yields (92% vs 84%) and avoiding harsh heating conditions.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method utilizes cheap, commercially available industrial reagents and operates under mild conditions without the need for high-temperature reflux, making it highly suitable for safe and efficient commercial scale-up.
Q: What types of substituents are tolerated on the o-phenylenediamine substrate?
A: The process demonstrates broad substrate scope, tolerating various functional groups including halogens, nitriles, esters, trifluoromethyl, nitro, benzoyl, and alkyl groups at multiple positions on the aromatic ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzothiadiazole Supplier
As the demand for high-performance heterocyclic building blocks grows in the optoelectronic and pharmaceutical sectors, having a partner with deep technical expertise is paramount. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. Our stringent purity specifications and rigorous QC labs guarantee that every batch of benzothiadiazole intermediate meets the exacting standards required for next-generation organic semiconductors and bioactive molecules.
We invite you to leverage our technical capabilities to optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how this advanced DAST-mediated synthesis can enhance your product portfolio and operational efficiency.
