Advanced Synthetic Route for Landiolol Hydrochloride Intermediates Enhancing Commercial Scalability
Advanced Synthetic Route for Landiolol Hydrochloride Intermediates Enhancing Commercial Scalability
The pharmaceutical landscape for ultra-short-acting beta-blockers has been significantly advanced by the development of Landiolol Hydrochloride, a potent agent for managing intraoperative tachyarrhythmias. As detailed in patent CN100506814C, a refined synthetic methodology has been established to address the critical bottlenecks associated with the commercial production of this complex cardiovascular active pharmaceutical ingredient (API). This technical insight report analyzes the novel five-step synthesis pathway that leverages chiral pool strategies and safer coupling reagents to deliver high-purity intermediates. By shifting away from hazardous reagents and inefficient purification techniques, this process offers a robust framework for reliable pharmaceutical intermediate supplier partnerships aiming to secure the supply chain for critical care medications.
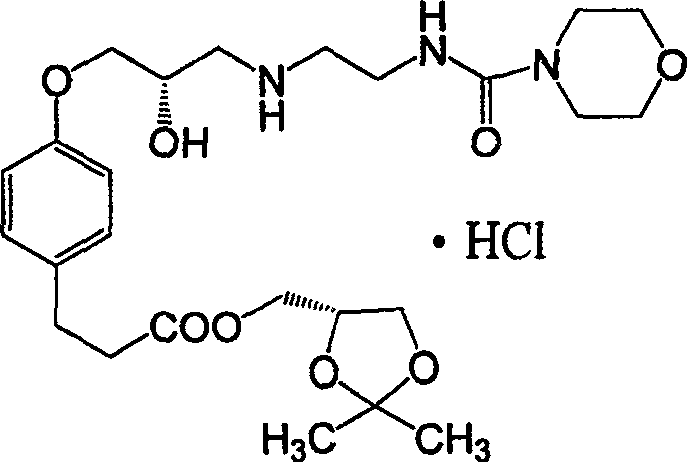
The structural complexity of Landiolol Hydrochloride necessitates precise stereochemical control, particularly at the 2S-position of the dioxolane ring and the propoxy side chain. The patented method introduces a strategic modification in the early stages of synthesis by utilizing S(+)-epichlorohydrin as a foundational chiral building block. This approach not only simplifies the introduction of chirality but also mitigates the risk of racemization that often plagues multi-step syntheses of beta-adrenergic antagonists. For R&D directors and process chemists, understanding the nuances of this pathway is essential for evaluating the feasibility of technology transfer and the potential for cost reduction in pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Landiolol and its precursors has been fraught with significant operational and safety challenges that hinder efficient commercial scale-up. Traditional routes, such as those disclosed in earlier Japanese patents, often rely on the use of 3-chloro-1,2-propanediol, which requires additional resolution steps to achieve the necessary optical purity, thereby increasing material costs and waste generation. Furthermore, conventional methodologies frequently employ phenyl chloroformate as a coupling agent for the morpholine moiety; this reagent is not only highly toxic and corrosive but also poses severe handling risks in a large-scale reactor environment. The presence of residual toxic impurities from such reagents necessitates rigorous and expensive downstream purification protocols to meet stringent regulatory standards for injectable drugs.
Another critical bottleneck in prior art methods is the reliance on column chromatography for the purification of key intermediates, specifically the hydroxyphenylpropionic acid ester derivatives. While effective on a laboratory scale, column chromatography is economically unviable for industrial production due to high solvent consumption, low throughput, and difficulties in automation. Additionally, the use of methanol as a solvent in the final salt formation step in older processes has been shown to induce decomposition of the sensitive epoxy and ester functionalities, leading to reduced overall yields and the formation of difficult-to-remove degradation products. These factors collectively contribute to a fragile supply chain and elevated production costs.
The Novel Approach
The innovative synthetic route described in the patent data fundamentally re-engineers the production workflow to eliminate these inefficiencies. A pivotal improvement is the direct utilization of S(+)-epichlorohydrin reacted with acetone in the presence of a boron trifluoride etherate catalyst. This reaction efficiently generates the chiral chloromethyl dioxolane intermediate with high stereoselectivity, bypassing the need for resolution of racemic mixtures. This step ensures that the spatial conformation required for beta-1 receptor selectivity is preserved from the outset, providing a robust foundation for the subsequent molecular assembly.

Furthermore, the novel approach replaces the hazardous phenyl chloroformate with carbonyldiimidazole (CDI) for the construction of the urea linkage. CDI is a safer, solid reagent that reacts cleanly with morpholine and ethylenediamine to form the N-(2-aminoethyl)morpholine carboxamide intermediate as an oxalate salt. This modification not only enhances operator safety but also simplifies the isolation of the amine component. Crucially, the process substitutes the impractical column chromatography purification with a scalable liquid-liquid extraction protocol using ethyl acetate and petroleum ether. This shift allows for the continuous processing of large batches, significantly enhancing the commercial scale-up of complex pharmaceutical intermediates while drastically reducing solvent waste and operational expenditure.
Mechanistic Insights into BF3-Catalyzed Chiral Protection and Coupling
The cornerstone of this synthetic strategy lies in the meticulous preservation of chirality through Lewis acid-catalyzed acetal formation. In the initial step, boron trifluoride etherate acts as a potent Lewis acid to activate the carbonyl oxygen of acetone, facilitating a nucleophilic attack by the diol functionality generated in situ or present on the epichlorohydrin derivative. This mechanism proceeds through a cyclic transition state that locks the stereochemistry of the S(+)-epichlorohydrin into the 1,3-dioxolane ring system. The choice of reaction temperature, optimized between 35°C and 45°C, is critical to balancing the reaction kinetics against the potential for acid-catalyzed ring opening or polymerization of the epoxide. Maintaining these conditions ensures that the resulting chloromethyl dioxolane retains high optical purity, which is indispensable for the biological efficacy of the final Landiolol molecule.
In the subsequent coupling phases, the mechanism shifts to nucleophilic substitution and ring-opening reactions. The phenolic hydroxyl group of the hydroxyphenylpropionic acid derivative attacks the chloromethyl group of the chiral dioxolane intermediate under basic conditions, forming the ether linkage. Later, the terminal epoxide ring is opened by the secondary amine of the morpholine-urea derivative. This ring-opening is regioselective, occurring at the less hindered carbon of the epoxide to generate the characteristic propanolamine side chain found in beta-blockers. The use of an isopropanol-water solvent system in this step is mechanistically significant; it provides sufficient solubility for both organic and salt forms of the reactants while minimizing hydrolysis of the sensitive ester and acetal groups. This careful solvent engineering prevents the formation of diol by-products that would otherwise complicate purification.
Impurity control is further enhanced by the avoidance of methanol in the final salting-out step. Methanol can act as a nucleophile towards the ester or epoxide moieties under acidic conditions, leading to transesterification or methoxy-alcohol by-products. By utilizing an ether solution containing hydrogen chloride and precipitating the product with n-hexane, the process ensures that the hydrochloride salt forms rapidly and cleanly without exposing the core structure to nucleophilic solvents. This results in a final product with a superior impurity profile, meeting the rigorous specifications required for parenteral administration. The mechanistic robustness of this route translates directly into higher batch-to-batch consistency, a key metric for high-purity pharmaceutical intermediates.
How to Synthesize Landiolol Hydrochloride Intermediates Efficiently
The execution of this synthetic pathway requires precise control over reaction parameters to maximize yield and minimize impurity formation. The process begins with the preparation of the chiral building block, followed by sequential esterification, etherification, and amine coupling. Each step has been optimized to utilize common industrial solvents and reagents, facilitating easy technology transfer from pilot plant to full-scale production. The following guide outlines the critical operational phases derived from the patent examples, emphasizing the conditions necessary to achieve the reported high yields and purity levels. For detailed standard operating procedures and safety data sheets, please refer to the technical documentation provided below.
- Synthesize (2,2-dimethyl-1,3-dioxolane-4S) methyl chloride using S(+)-epichlorohydrin and acetone with boron trifluoride etherate catalyst.
- Perform esterification with p-hydroxyphenylpropionic acid in dimethyl sulfoxide using strong base to form the protected ester intermediate.
- Execute etherification with S(+)-epichlorohydrin in acetone to introduce the epoxy group, followed by ring-opening with morpholine-derived amine.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthetic route offers transformative benefits that extend beyond simple yield improvements. The elimination of column chromatography represents a paradigm shift in downstream processing, converting a batch-limited, labor-intensive operation into a continuous, scalable extraction process. This change drastically reduces the consumption of high-purity silica gel and vast quantities of elution solvents, leading to substantial cost savings in raw materials and waste disposal. Moreover, the replacement of toxic phenyl chloroformate with carbonyldiimidazole removes a significant safety hazard from the manufacturing floor, lowering insurance premiums and reducing the need for specialized containment equipment. These operational efficiencies collectively contribute to a more resilient and cost-effective supply chain for cardiovascular drug manufacturers.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of purification steps and the use of commodity chemicals. By replacing expensive and hazardous reagents with safer alternatives like CDI and S(+)-epichlorohydrin, the direct material costs are significantly lowered. Furthermore, the ability to isolate intermediates via crystallization or extraction rather than chromatography reduces the cycle time per batch, increasing the overall throughput of the manufacturing facility. This efficiency gain allows for a more competitive pricing structure for the final API, enabling healthcare providers to access life-saving arrhythmia treatments at a reduced cost without compromising on quality or safety standards.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as acetone, ethylenediamine, and morpholine ensures that the supply chain is not vulnerable to the shortages often associated with specialized chiral reagents. S(+)-epichlorohydrin is a bulk chemical produced globally, providing a stable source of chirality that is less prone to market volatility. Additionally, the robustness of the reaction conditions, which tolerate a wider range of temperatures and concentrations compared to the sensitive prior art methods, reduces the risk of batch failures. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of major pharmaceutical companies.
- Scalability and Environmental Compliance: The environmental footprint of the manufacturing process is markedly reduced through the minimization of solvent waste and the elimination of toxic by-products. The shift to extraction-based purification aligns with green chemistry principles by reducing the E-factor (mass of waste per mass of product). This compliance with environmental regulations facilitates smoother regulatory approvals and reduces the liability associated with hazardous waste management. The process is inherently scalable, having been designed with industrial reactors in mind, allowing for seamless expansion from kilogram to multi-ton production scales to meet growing global demand for Landiolol Hydrochloride.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of Landiolol Hydrochloride intermediates. These answers are derived directly from the patented methodology and reflect the practical considerations for implementing this technology in a GMP environment. Understanding these details is vital for stakeholders evaluating the feasibility of this route for their specific production needs.
Q: How does this new synthesis method improve upon traditional Landiolol production?
A: The novel method replaces toxic phenyl chloroformate with carbonyldiimidazole and eliminates costly column chromatography purification steps, significantly reducing production costs and environmental hazards while maintaining high stereochemical purity.
Q: What are the critical quality attributes controlled in this process?
A: The process strictly controls the S-configuration at the chiral center using S(+)-epichlorohydrin throughout the synthesis, ensuring the biological activity of the final beta-blocker, while avoiding methanol-induced decomposition during salt formation.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the route is designed for industrial scalability by utilizing liquid-liquid extraction instead of chromatography for purification and employing stable, commercially available raw materials like acetone and epichlorohydrin.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Landiolol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of cardiovascular intermediates for the global pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate stereochemical requirements of molecules like Landiolol are met with precision. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to verify the identity and optical purity of every batch. Our commitment to excellence ensures that our clients receive intermediates that are fully compliant with international pharmacopoeia standards, ready for final API synthesis.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for beta-blocker production. By leveraging our expertise in chiral synthesis and process optimization, we can help you achieve significant operational efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals in the cardiovascular therapeutic sector.
