Advanced Synthetic Route for Landiolol Hydrochloride Intermediates and Commercial Scale-Up
Introduction to Patent CN110483470B and Landiolol Hydrochloride
The pharmaceutical landscape for short-acting beta-blockers has been significantly advanced by the development of Landiolol Hydrochloride, a potent agent primarily utilized for the emergency treatment of tachycardia arrhythmias during surgical procedures. As detailed in patent CN110483470B, published in late September 2022, there exists a critical need for improved synthetic methodologies that can deliver this active pharmaceutical ingredient (API) with higher efficiency and reduced environmental impact. Landiolol is distinguished by its ultra-short half-life and rapid onset of action, properties that make it indispensable in critical care settings where precise hemodynamic control is required. However, the commercial viability of such a sophisticated molecule often hinges on the robustness of its supply chain and the elegance of its chemical synthesis. This patent introduces a transformative approach to constructing the Landiolol backbone, specifically focusing on the preparation of key intermediates designated as Formula 8 and their subsequent conversion to the hydrochloride salt. By leveraging a novel isocyanate-based coupling strategy, the inventors have addressed longstanding bottlenecks associated with traditional amide bond formation in this chemical space.
The Limitations of Conventional Methods vs. The Novel Approach
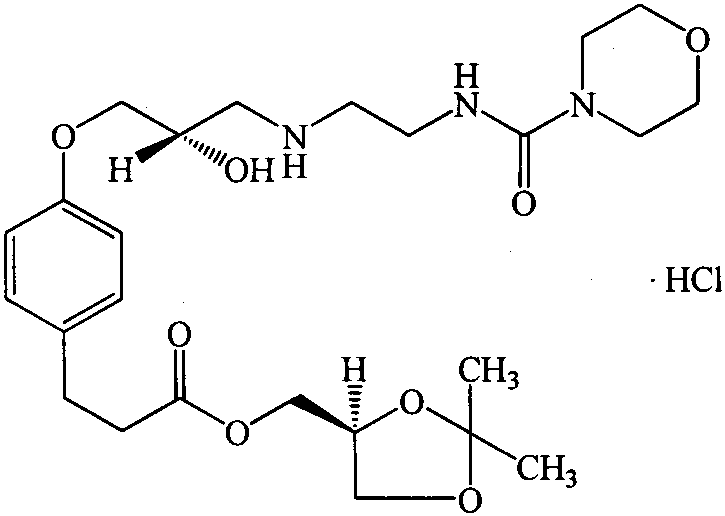
The Limitations of Conventional Methods
Historically, the synthesis of Landiolol Hydrochloride and its analogues has relied heavily on the condensation of an epoxy-containing precursor (Compound 6a) with a pre-activated amine derivative (Compound 10). As illustrated in the prior art reaction schemes, this conventional pathway necessitates the use of aggressive activating agents such as phenyl chloroformate or carbonyldiimidazole (CDI) to facilitate the formation of the urea linkage. These reagents are not only expensive but also introduce significant toxicity hazards and complicate the downstream purification processes. The generation of stoichiometric amounts of toxic byproducts, such as phenol or imidazole derivatives, imposes a heavy burden on waste management systems and increases the overall cost of goods sold (COGS). Furthermore, the preparation of Compound 10 itself is often multi-step and low-yielding, creating a fragile supply chain that is susceptible to disruptions. The reliance on these harsh activation methods also poses risks to the stereochemical integrity of the chiral centers within the molecule, potentially leading to difficult-to-remove impurities that compromise the safety profile of the final drug product.
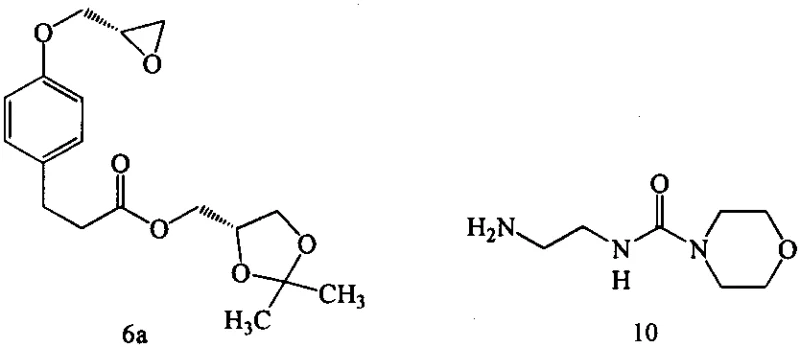
The Novel Approach
In stark contrast to the cumbersome legacy methods, the technology disclosed in CN110483470B proposes a streamlined and atom-economical route centered around the direct reaction of an epoxy-amine intermediate with an isocyanate species. The core innovation lies in the generation and utilization of Compound 7, a versatile intermediate formed by the ring-opening of the epoxide (Compound 6) by an amino-isocyanate (Compound 3). This strategic shift eliminates the need for external coupling reagents entirely. Instead of activating the amine post-synthesis, the isocyanate functionality is introduced early in the sequence, allowing for a highly efficient nucleophilic attack by various organic amines (AH) in the subsequent step. This approach not only simplifies the operational workflow by reducing the number of unit operations but also dramatically improves the impurity profile of the crude reaction mixture. The versatility of this method is underscored by its compatibility with a wide range of amines, including morpholine and isopropylamine, enabling the rapid synthesis of diverse Landiolol analogues without re-optimizing the entire synthetic tree.
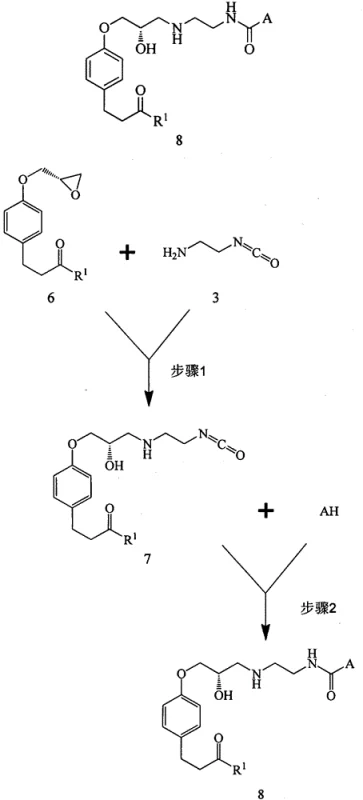
Mechanistic Insights into Isocyanate-Mediated Urea Formation
The mechanistic elegance of this new process is rooted in the high reactivity of the isocyanate group towards nucleophiles. In the first critical step, the primary amine of Compound 3 acts as a nucleophile, attacking the less sterically hindered carbon of the oxirane ring in Compound 6. This ring-opening reaction is typically catalyzed by mild organic bases such as triethylamine or inorganic bases like sodium bicarbonate, proceeding smoothly at temperatures ranging from 10°C to 30°C. The result is the formation of Compound 7, which retains the reactive isocyanate moiety intact while establishing the crucial ether linkage to the aromatic core. This intermediate is remarkably stable under the reaction conditions yet sufficiently reactive to undergo the subsequent transformation. In the second step, the isocyanate group of Compound 7 serves as an electrophile, readily accepting a nucleophilic attack from the secondary amine (AH), such as morpholine. This addition reaction proceeds without the need for additional activation energy or coupling catalysts, directly yielding the urea linkage found in the final Landiolol structure. The absence of leaving groups in this step means that the atom economy is near perfect, with virtually all reactant mass incorporated into the desired product.
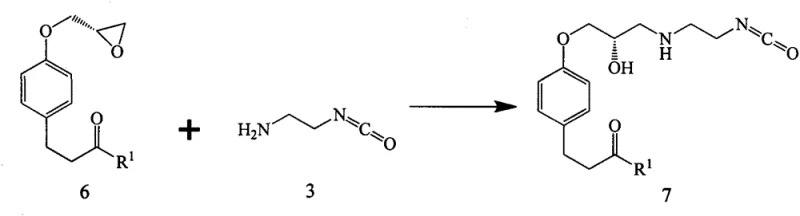
From an impurity control perspective, this mechanism offers distinct advantages over carbodiimide-mediated couplings. Traditional methods often suffer from the formation of N-acylurea byproducts or racemization at the chiral center adjacent to the carbonyl group. The direct isocyanate-amine addition described here bypasses these pathways entirely. The reaction is clean, producing minimal side products, which facilitates easier purification via simple aqueous workups and crystallization. The patent data highlights that the intermediate Compound 7a can be isolated with an HPLC purity of 99.4%, and the final coupled product Compound 8a achieves purities exceeding 99.6%. This high level of chemical fidelity is critical for meeting the stringent regulatory requirements for injectable pharmaceuticals, where impurity thresholds are exceptionally low. Furthermore, the mild reaction conditions preserve the acid-sensitive acetal protecting groups present in the side chain, preventing premature deprotection that could lead to complex mixtures of degradation products.
How to Synthesize Landiolol Hydrochloride Efficiently
The practical implementation of this synthetic route involves a carefully controlled sequence of reactions designed to maximize yield and minimize operational complexity. The process begins with the preparation of the key isocyanate intermediate, followed by its coupling with the chosen amine and final salt formation. Each step has been optimized for scalability, utilizing solvents and reagents that are readily available in bulk quantities. The following guide outlines the standardized protocol derived from the patent examples, providing a roadmap for R&D teams looking to adopt this superior methodology. For detailed operational parameters, stoichiometry, and specific workup procedures, please refer to the structured synthesis guide below.
- React the epoxy compound (Formula 6) with the amino-isocyanate derivative (Formula 3) in a solvent like acetonitrile or THF with a base to form the key intermediate (Formula 7).
- Couple the intermediate (Formula 7) with a specific organic amine (AH), such as morpholine, to generate the free base compound (Formula 8).
- Convert the free base (Formula 8) into the final hydrochloride salt using an HCl solution in a suitable solvent like ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route represents a significant opportunity to optimize the cost structure and reliability of the Landiolol supply chain. The elimination of expensive and hazardous coupling reagents translates directly into reduced raw material costs and lower expenditures on waste disposal. By simplifying the synthesis to fewer steps with higher overall yields, manufacturers can achieve substantial cost savings in production overheads. The robustness of the process also implies a more resilient supply chain, as it reduces dependency on niche reagents that may be subject to market volatility. Additionally, the high purity of the intermediates reduces the need for extensive chromatographic purification, which is often a bottleneck in large-scale manufacturing. This efficiency gain allows for faster batch turnover times and increased production capacity without the need for capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The primary driver for cost reduction in this process is the complete removal of activating agents like phenyl chloroformate and CDI. These reagents are not only costly to purchase but also require careful handling and disposal due to their toxicity. By replacing them with a direct isocyanate coupling strategy, the process eliminates an entire class of expensive consumables. Furthermore, the high yields reported in the patent examples (often exceeding 85% for intermediates and 95% for final coupling) mean that less starting material is wasted. This improved material efficiency lowers the effective cost per kilogram of the API. The simplified workup procedures, which rely on standard aqueous washes and crystallization rather than complex chromatography, further reduce labor and solvent costs, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: Supply chain reliability is significantly bolstered by the use of commodity chemicals and robust reaction conditions. The solvents employed, such as acetonitrile, dichloromethane, and ethyl acetate, are widely produced and easily sourced from multiple suppliers, mitigating the risk of single-source dependency. The reaction tolerates a broad range of temperatures and does not require specialized cryogenic or high-pressure equipment, making it adaptable to various manufacturing sites globally. This flexibility ensures that production can be maintained even if one facility faces operational challenges. Moreover, the stability of the key intermediate (Compound 7) allows for potential stockpiling or campaign manufacturing, providing a buffer against demand fluctuations. The consistent quality of the output, as evidenced by the high HPLC purity data, reduces the risk of batch failures and recalls, ensuring a steady flow of product to the market.
- Scalability and Environmental Compliance: Scalability is inherent in the design of this process, which avoids exothermic hazards associated with large-scale additions of activating agents. The mild reaction temperatures (10-30°C) are easily maintained in large reactors using standard cooling systems. From an environmental compliance standpoint, the process is markedly greener. The avoidance of toxic reagents reduces the load of hazardous waste generated per kilogram of product. The atom economy of the urea formation step is excellent, as no small molecule byproducts are released. This aligns with modern green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations. The reduced solvent usage and simplified purification steps also lower the overall E-factor (mass of waste per mass of product), making the process more sustainable and environmentally friendly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Landiolol synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a reliable basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of technology transfer and the potential return on investment for adopting this new route.
Q: What are the safety advantages of this new Landiolol synthesis route?
A: The novel process eliminates the use of highly toxic reagents such as phenyl chloroformate or carbonyldiimidazole, which were required in conventional methods to activate the amine. By utilizing a direct isocyanate coupling strategy, the process significantly reduces hazardous waste generation and improves operator safety profiles.
Q: What purity levels can be achieved with this manufacturing method?
A: According to the patent data, the process consistently yields intermediates and final products with HPLC purity exceeding 99%. For instance, the key intermediate (Formula 7a) was isolated with 99.4% purity, and the final Landiolol Hydrochloride achieved 99.85% purity after crystallization.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the methodology is designed for scalability. It utilizes common industrial solvents like acetonitrile, dichloromethane, and ethyl acetate, and operates under mild temperature conditions (10-30°C), making it highly adaptable for multi-kilogram to ton-scale manufacturing without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Landiolol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a robust and scalable supply chain for life-saving medications like Landiolol Hydrochloride. Our team of expert chemists has thoroughly analyzed the innovative route described in CN110483470B and is fully prepared to execute this chemistry at a commercial scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of API intermediate or finished product meets the highest global standards. We understand that in the pharmaceutical industry, quality cannot be compromised, and our commitment to excellence is reflected in our comprehensive quality assurance protocols.
We invite you to collaborate with us to leverage this advanced synthetic technology for your Landiolol projects. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. Our technical procurement team is ready to provide you with specific COA data from pilot batches and detailed route feasibility assessments to demonstrate the tangible benefits of this process. Contact us today to discuss how we can support your supply chain with high-quality, cost-effective Landiolol Hydrochloride intermediates and drive your project forward with confidence.
