Advanced Linear Phenanthroline Ligands For High-Efficiency Catalytic Applications And Commercial Scale-Up
The landscape of modern catalytic chemistry is continuously evolving, driven by the demand for more efficient and cost-effective ligand systems that can optimize metal-catalyzed transformations. Patent CN111100124A introduces a significant breakthrough in this domain by disclosing a novel class of linear phenanthroline compounds that serve as highly efficient bidentate nitrogen ligands. These specialized structures are designed to coordinate with metals, particularly palladium, to facilitate organic reactions with exceptional turnover numbers. For research and development directors overseeing complex synthetic pathways, the introduction of such high-performance ligands represents a critical opportunity to enhance reaction kinetics and reduce overall catalyst loading. The patent details a robust preparation method that ensures the structural integrity and purity required for demanding industrial applications, positioning this technology as a cornerstone for next-generation catalytic systems in the fine chemical and pharmaceutical sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional phenanthroline derivatives, while widely used, often suffer from limitations in steric bulk and electronic tuning that restrict their catalytic efficiency in challenging transformations. Conventional 1,10-phenanthroline ligands typically exhibit lower turnover numbers, necessitating higher loadings of precious metal catalysts to achieve acceptable conversion rates, which directly impacts the cost structure of large-scale manufacturing. Furthermore, standard modification strategies often lack the precision required to create axisymmetric molecular structures that maximize orbital overlap with the metal center. This inefficiency leads to increased production costs due to the excessive consumption of noble metals and generates larger volumes of metal-contaminated waste streams that require expensive remediation. In the context of producing high-value pharmaceutical intermediates, these inefficiencies can create bottlenecks that delay project timelines and erode profit margins for procurement teams managing tight budgets.
The Novel Approach
The innovative approach detailed in the patent overcomes these historical constraints by introducing a linear phenanthroline scaffold modified specifically at the 2,9 and 3,8 positions. This strategic functionalization creates a unique steric environment that stabilizes the metal-ligand complex, thereby significantly enhancing catalytic activity and longevity. By employing a two-step synthesis involving Suzuki coupling followed by nucleophilic substitution with alkyllithium reagents, the method allows for precise control over the substituent groups, enabling the fine-tuning of electronic properties to match specific reaction requirements. This level of molecular engineering results in ligands that can drive reactions to completion with minimal catalyst loading, offering a transformative solution for cost reduction in fine chemical manufacturing. The ability to synthesize these complex structures reliably opens new avenues for developing proprietary catalytic processes that offer distinct competitive advantages in the global market.
Mechanistic Insights into Suzuki Coupling and Nucleophilic Substitution
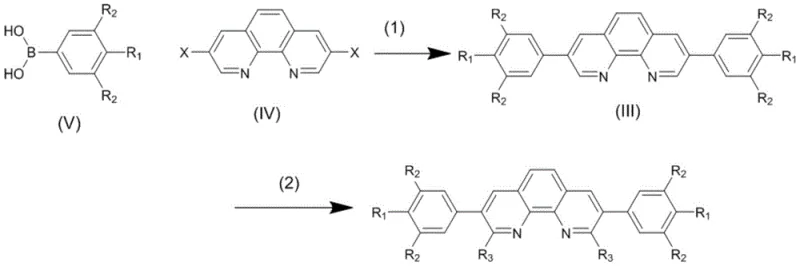
The synthesis of these advanced ligands relies on a sophisticated two-stage mechanism that begins with a palladium-catalyzed Suzuki coupling reaction. In this initial phase, a dihalo-phenanthroline precursor reacts with arylboronic acids in a mixed solvent system comprising toluene, ethanol, and water, facilitated by a base such as sodium carbonate. The choice of solvent ratio, specifically 4:3:1 for toluene, ethanol, and water, is critical for maintaining the solubility of both organic and inorganic components while ensuring optimal reaction kinetics at temperatures between 70°C and 100°C. This step constructs the core carbon-carbon bonds that define the linear architecture of the molecule, establishing the foundation for the subsequent functionalization. The precision required in this coupling step ensures that the resulting intermediate possesses the necessary symmetry and purity to support the high-performance characteristics of the final ligand product.
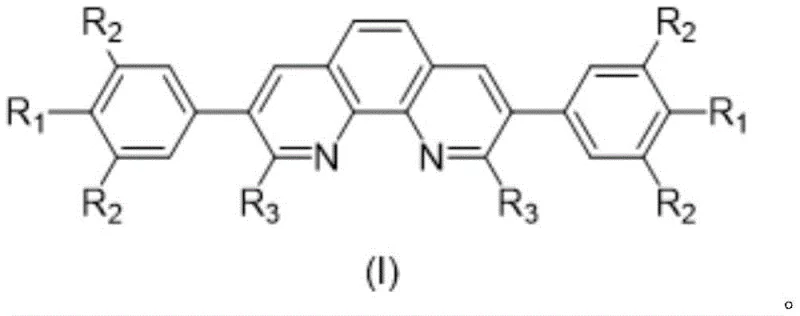
Following the initial coupling, the mechanism proceeds to a nucleophilic substitution reaction where the intermediate is treated with alkyllithium reagents under strictly anhydrous conditions. This step involves the addition of the organolithium species at low temperatures, typically between -5°C and 0°C, to prevent side reactions and ensure regioselectivity at the 2,9 positions of the phenanthroline ring. The reaction mixture is then allowed to warm to 25°C over a period of 12 to 36 hours, facilitating the complete substitution of halogen atoms with alkyl groups such as isopropyl. This transformation is crucial for introducing the steric bulk that protects the metal center during catalysis, thereby preventing catalyst deactivation and extending the operational life of the complex. The rigorous control over temperature and reaction time in this stage is essential for achieving the high yields and purity levels demanded by commercial scale-up of complex catalyst ligands.
How to Synthesize Linear Phenanthroline Efficiently
To successfully implement this synthesis route in a production environment, strict adherence to the patented reaction parameters is essential for maximizing yield and minimizing impurity formation. The process begins with the careful preparation of the Suzuki coupling mixture, ensuring that the molar ratio of the dihalo-phenanthroline to the boronic acid is maintained at approximately 1:2.5 to drive the reaction to completion without excessive reagent waste. Following the isolation of the intermediate, the nucleophilic substitution must be conducted in deoxygenated solvents like diethyl ether to prevent the quenching of the reactive alkyllithium species by moisture or oxygen. Detailed standardized synthesis steps see the guide below for specific operational protocols that ensure reproducibility and safety during the handling of pyrophoric reagents.
- Perform Suzuki coupling between dibromo-phenanthroline and arylboronic acid using a palladium catalyst in a toluene-ethanol-water system at 70-100°C.
- Isolate the intermediate product through extraction with dichloromethane and recrystallization using alcohol solvents.
- Conduct nucleophilic substitution by reacting the intermediate with alkyllithium in anhydrous ether at low temperature, followed by warming to 25°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel ligand technology offers substantial strategic benefits that extend beyond mere technical performance. The primary advantage lies in the drastic simplification of the downstream purification process, as the high selectivity of the catalyst reduces the formation of difficult-to-remove byproducts. This efficiency translates directly into significant cost savings by reducing the consumption of solvents and energy required for extensive chromatographic separations. Furthermore, the enhanced stability of the catalyst system means that smaller quantities of expensive palladium are needed to achieve the same output, effectively lowering the raw material cost per kilogram of the final active pharmaceutical ingredient. These factors combine to create a more resilient supply chain that is less vulnerable to fluctuations in the price of precious metals.
- Cost Reduction in Manufacturing: The implementation of this high-TON ligand system allows manufacturers to drastically reduce the loading of palladium catalysts required for Heck reactions, leading to substantial cost savings in raw material expenditure. By minimizing the amount of noble metal needed, companies can mitigate the financial risk associated with volatile metal markets while simultaneously reducing the cost burden of metal scavenging and removal steps. This qualitative improvement in process efficiency ensures that the overall cost of goods sold is optimized without compromising on the quality or purity of the final product, providing a clear economic advantage over conventional catalytic methods.
- Enhanced Supply Chain Reliability: The synthesis route utilizes readily available starting materials and common solvents, which ensures a stable and continuous supply of the ligand without reliance on exotic or hard-to-source reagents. This accessibility reduces the risk of supply disruptions and shortens the lead time for high-purity pharmaceutical intermediates, allowing procurement teams to maintain leaner inventory levels. The robustness of the chemical process also means that production schedules are more predictable, enabling better planning and coordination with downstream customers who depend on timely delivery of critical materials for their own manufacturing operations.
- Scalability and Environmental Compliance: The reaction conditions are well-suited for commercial scale-up, operating at moderate temperatures and pressures that do not require specialized high-pressure equipment. This ease of scalability facilitates the transition from laboratory benchtop to multi-ton production, ensuring that supply can meet growing market demand efficiently. Additionally, the reduction in catalyst loading and the use of recyclable solvent systems contribute to a greener manufacturing process, helping companies meet increasingly stringent environmental regulations and sustainability goals while maintaining high production throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this linear phenanthroline technology in industrial settings. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a reliable basis for decision-making. Understanding these aspects is crucial for stakeholders evaluating the feasibility of integrating this new ligand system into existing production workflows to achieve better economic and technical outcomes.
Q: What is the primary advantage of the linear phenanthroline structure described in CN111100124A?
A: The linear structure modified at the 2,9 and 3,8 positions significantly enhances the turnover number (TON) in Mizoroki-Heck reactions, reaching up to 100,000, which drastically reduces the required loading of expensive palladium catalysts.
Q: How does the synthesis method ensure high purity for industrial applications?
A: The process utilizes a robust two-step sequence involving Suzuki coupling and controlled nucleophilic substitution, followed by rigorous recrystallization steps using specific solvent systems like methanol or ethyl acetate to remove impurities effectively.
Q: Is this ligand suitable for large-scale pharmaceutical intermediate manufacturing?
A: Yes, the reaction conditions utilize common solvents such as toluene and ether and operate at moderate temperatures, making the process highly adaptable for commercial scale-up from kilogram to metric ton quantities without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Linear Phenanthroline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced ligand systems play in driving innovation within the pharmaceutical and fine chemical industries. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of linear phenanthroline ligand performs consistently in your catalytic processes. Our dedication to quality and technical excellence makes us the ideal partner for organizations seeking to leverage this cutting-edge technology for their manufacturing needs.
We invite you to collaborate with us to explore how this novel ligand can optimize your specific synthetic routes and deliver tangible value to your bottom line. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current production challenges. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our supply capabilities can support your long-term strategic goals and enhance your competitive position in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
