Advanced Linear Phenanthroline Ligands: Revolutionizing Palladium-Catalyzed Cross-Coupling Efficiency
Advanced Linear Phenanthroline Ligands: Revolutionizing Palladium-Catalyzed Cross-Coupling Efficiency
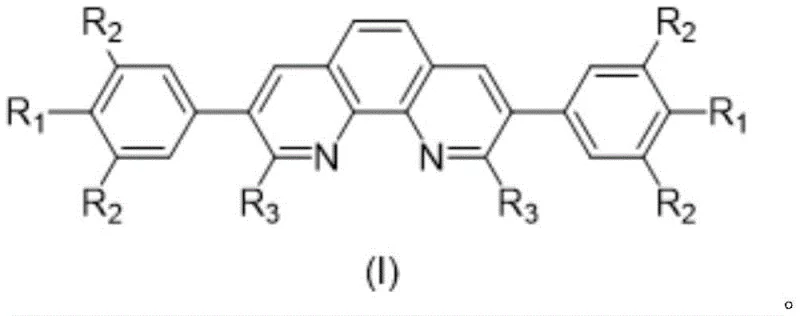
The landscape of transition metal catalysis is continuously evolving, driven by the demand for more efficient, sustainable, and cost-effective ligand systems. A significant breakthrough in this domain is documented in patent CN111100124B, which introduces a novel class of linear o-phenanthroline compounds. These compounds are not merely structural variants but represent a strategic modification of the classic 1,10-phenanthroline scaffold, specifically targeting the 2,9 and 3,8 positions to optimize electronic and steric properties. For R&D directors and process chemists, this innovation offers a compelling solution to the persistent challenge of maximizing catalyst turnover while minimizing the loading of precious metals like palladium. The patent details a robust synthetic pathway that transforms simple precursors into highly active bidentate nitrogen ligands, capable of driving complex organic transformations with exceptional efficiency.
From a commercial perspective, the implications of this technology extend far beyond the laboratory bench. As global supply chains face pressure to reduce costs and environmental footprints, the ability to utilize ligands that enhance catalyst longevity is paramount. This report analyzes the technical merits of these linear phenanthroline derivatives, evaluating their synthesis, mechanistic advantages, and potential for industrial adoption. By understanding the specific structural modifications—such as the introduction of isopropyl groups or methoxy-substituted phenyl rings—stakeholders can better appreciate how these molecules function as superior partners in catalytic cycles, ultimately leading to more economical manufacturing processes for fine chemicals and pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional 1,10-phenanthroline ligands have long served as workhorses in coordination chemistry, yet they often suffer from limitations when applied to demanding industrial catalytic cycles. Standard derivatives typically lack the necessary steric bulk to prevent catalyst deactivation pathways, such as the formation of inactive metal aggregates or the dissociation of the ligand under harsh reaction conditions. In many conventional Heck or Suzuki coupling protocols, this instability necessitates the use of excessive amounts of palladium catalysts to maintain acceptable reaction rates, directly inflating production costs. Furthermore, unmodified phenanthrolines may exhibit poor solubility in non-polar organic solvents commonly used in large-scale synthesis, leading to heterogeneous reaction mixtures that are difficult to control and scale. The background art highlights that while previous modifications have achieved Turnover Numbers (TON) around 55,000, there remains a critical gap for ligands that can push these metrics even higher while maintaining structural simplicity and ease of synthesis.
The Novel Approach
The invention disclosed in CN111100124B addresses these deficiencies through a dual-modification strategy that simultaneously functionalizes the 3,8 and 2,9 positions of the phenanthroline core. This approach creates a "pocket" around the metal center that stabilizes the active catalytic species without hindering substrate access. By introducing bulky alkyl groups, such as isopropyl, at the 2,9-positions, the ligand effectively shields the metal center from bimolecular decomposition pathways. Simultaneously, the substitution at the 3,8-positions with aryl groups allows for fine-tuning of the electronic density on the nitrogen atoms, optimizing the donation of electron density to the metal. This synergistic effect results in a ligand system that supports significantly higher turnover frequencies and numbers compared to prior art. The result is a catalytic system that operates efficiently at lower metal loadings, offering a direct route to cost reduction in fine chemical manufacturing without compromising on yield or selectivity.
Mechanistic Insights into Suzuki Coupling and Nucleophilic Substitution
The synthesis of these advanced ligands relies on a sophisticated yet practical two-step sequence that leverages well-understood organic transformations. The first step involves a Suzuki-Miyaura cross-coupling reaction, where a 3,8-dihalo-1,10-phenanthroline precursor reacts with various arylboronic acids. This reaction is catalyzed by palladium species, such as tetrakis(triphenylphosphine)palladium(0), in a mixed solvent system of toluene, ethanol, and water. The presence of a base, typically sodium carbonate, facilitates the transmetallation step essential for carbon-carbon bond formation. This step is crucial as it establishes the backbone of the ligand, installing the aryl substituents that will influence the electronic properties of the final product. The reaction proceeds smoothly at temperatures between 70°C and 100°C, demonstrating excellent functional group tolerance and yielding the intermediate bis(aryl)-1,10-phenanthroline in high purity after standard workup procedures.
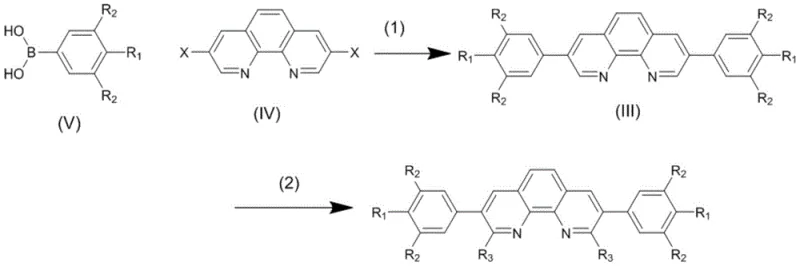
Following the initial coupling, the second critical transformation is a nucleophilic substitution at the 2,9-positions. This step involves the treatment of the intermediate with strong nucleophiles, specifically alkyllithium reagents like lithium isopropyl. The reaction is conducted under strict inert atmosphere conditions, initially at low temperatures (-5 to 0°C) to control the exotherm and prevent side reactions, before warming to ambient temperature (25°C) to drive the substitution to completion. This step installs the sterically demanding alkyl groups that are vital for the ligand's performance. The mechanism likely proceeds via an addition-elimination pathway or direct nucleophilic aromatic substitution, depending on the specific leaving groups present on the phenanthroline ring. The careful control of stoichiometry and temperature ensures that the substitution occurs selectively at the desired positions, preserving the integrity of the newly formed aryl bonds from the first step. This precise control over molecular architecture is what enables the resulting ligand to outperform conventional analogues in catalytic applications.
How to Synthesize Linear Phenanthroline Compounds Efficiently
The preparation of these high-value ligands requires strict adherence to the optimized conditions outlined in the patent to ensure maximum yield and purity. The process begins with the rigorous exclusion of oxygen and moisture, utilizing inert gas protection throughout both reaction stages to prevent the degradation of sensitive organometallic intermediates. Solvent selection is also critical; the use of a toluene/ethanol/water mixture in the first step optimizes the solubility of both organic and inorganic reagents, while anhydrous diethyl ether is preferred for the lithiation step to stabilize the organolithium species. Post-reaction processing involves careful pH control during extraction and multiple recrystallization steps to remove trace metal residues and organic byproducts.
- Perform Suzuki coupling between 3,8-dihalo-1,10-phenanthroline and arylboronic acids using a palladium catalyst in a toluene/ethanol/water system at 70-100°C.
- Isolate the intermediate bis(aryl)-1,10-phenanthroline via extraction and recrystallization.
- Conduct nucleophilic substitution by reacting the intermediate with alkyllithium reagents at low temperature (-5 to 0°C) followed by warming to 25°C to install alkyl groups at the 2,9-positions.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ligand technology presents a compelling value proposition centered on cost efficiency and supply reliability. The primary economic driver is the drastic reduction in the consumption of noble metal catalysts. Because the linear phenanthroline ligands enable significantly higher Turnover Numbers (TON), manufacturers can achieve the same output with a fraction of the palladium loading traditionally required. This directly translates to substantial cost savings, given the volatile and high price of palladium. Furthermore, the synthesis of the ligand itself relies on commodity chemicals such as phenylboronic acids and simple alkyl halides, which are readily available from a broad network of global suppliers. This reduces the risk of supply chain bottlenecks that often plague specialized reagent markets.
- Cost Reduction in Manufacturing: The implementation of this ligand system allows for a qualitative shift in process economics by minimizing the reliance on expensive precious metals. By enhancing the stability and activity of the palladium catalyst, the total amount of metal required per kilogram of product is significantly lowered. Additionally, the synthetic route avoids the use of exotic or prohibitively expensive reagents, relying instead on scalable chemistry that can be performed in standard glass-lined or stainless steel reactors. The elimination of complex purification steps often associated with removing heavy metal contaminants further streamlines the downstream processing, reducing solvent usage and waste disposal costs.
- Enhanced Supply Chain Reliability: The raw materials required for the synthesis, including 3,8-dibromo-1,10-phenanthroline and various boronic acids, are established commodities within the fine chemical industry. This ensures a stable and continuous supply flow, mitigating the risks associated with single-source dependencies. The robustness of the Suzuki coupling and nucleophilic substitution steps means that the manufacturing process is less susceptible to variations in raw material quality, providing greater consistency in batch-to-batch production. This reliability is crucial for maintaining uninterrupted production schedules in high-volume pharmaceutical and agrochemical manufacturing facilities.
- Scalability and Environmental Compliance: The process described is inherently scalable, utilizing reaction conditions that are easily transferable from pilot plant to commercial production scales. The use of common solvents like toluene, ethanol, and diethyl ether simplifies solvent recovery and recycling programs, aligning with modern green chemistry principles. Moreover, the high efficiency of the catalyst system reduces the generation of metal-containing waste streams, easing the burden on wastewater treatment facilities and ensuring compliance with increasingly stringent environmental regulations. This combination of operational simplicity and environmental stewardship makes the technology highly attractive for long-term industrial deployment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ligand technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the operational parameters and expected outcomes.
Q: What is the primary advantage of the linear phenanthroline ligand described in CN111100124B?
A: The primary advantage is the significantly enhanced Turnover Number (TON) in palladium-catalyzed reactions, specifically the Mizoroki-Heck reaction. By modifying both the 3,8 and 2,9 positions, the ligand creates a more stable and active coordination environment, allowing for reduced loading of expensive noble metal catalysts.
Q: How does the synthesis route ensure high purity for pharmaceutical applications?
A: The synthesis utilizes a robust two-step sequence starting with Suzuki coupling, which is highly selective, followed by a controlled nucleophilic substitution. The process includes rigorous post-treatment steps such as alkaline washing, solvent extraction, and recrystallization from solvents like ethanol or methanol, ensuring the removal of residual palladium and organic impurities to meet stringent purity specifications.
Q: Is this ligand suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for scalability. It employs common reagents like arylboronic acids and alkyllithiums, and uses standard solvents such as toluene, ethanol, and diethyl ether. The reaction conditions (70-100°C for coupling, ambient temperature for substitution) are manageable in large reactors, facilitating commercial scale-up without requiring exotic cryogenic or high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Linear Phenanthroline Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced ligand systems like those described in CN111100124B for the fine chemical and pharmaceutical industries. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to translate these innovative synthetic routes into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify the absence of residual metals and organic impurities, thereby guaranteeing the performance of your catalytic processes.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific applications. Whether you require custom synthesis of the ligand itself or the development of a catalytic process utilizing these compounds, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your current manufacturing challenges. We encourage you to reach out for specific COA data and route feasibility assessments to determine how our capabilities can support your supply chain goals and drive innovation in your product portfolio.
