Scaling High-Efficiency Ferrocene Catalysts for Commercial Pharmaceutical Intermediate Production
Scaling High-Efficiency Ferrocene Catalysts for Commercial Pharmaceutical Intermediate Production
The landscape of asymmetric synthesis is continuously evolving, driven by the urgent need for more efficient and recoverable chiral catalysts in the production of high-value pharmaceutical intermediates. A significant technological breakthrough in this domain is documented in Chinese Patent CN101486737A, which introduces a novel ferrocene phosphinimine ligand containing a quaternary ammonium salt group. This innovation addresses one of the most persistent challenges in industrial homogeneous catalysis: the difficulty of separating expensive chiral ligands from the reaction mixture after the process is complete. By integrating a quaternary ammonium moiety into the ferrocene backbone, the inventors have created a system that combines high catalytic activity with unique solubility characteristics, enabling potential recycling strategies that were previously unattainable with standard ferrocene derivatives. For global procurement and R&D teams, understanding the implications of this technology is critical for optimizing supply chains and reducing the overall cost of goods sold for complex chiral molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, ferrocene-based chiral ligands have been highly prized for their ability to induce planar chirality and facilitate asymmetric transformations with high stereoselectivity. However, their widespread industrial adoption has been severely hindered by their high solubility in most common organic solvents, which makes post-reaction recovery nearly impossible without destructive or excessively costly purification steps. Conventional methods often rely on immobilizing these catalysts on solid supports such as polymers, silica gel, or dendrimers to facilitate filtration, but this approach frequently compromises the catalytic activity due to steric hindrance and reduced solubility of the active sites. Furthermore, the loss of precious metal complexes and chiral ligands in the mother liquor represents a significant economic drain, particularly when scaling up processes for commercial manufacturing where raw material costs are a primary concern. The inability to efficiently recycle these high-value components creates a bottleneck in the sustainable production of fine chemicals and active pharmaceutical ingredients.
The Novel Approach
The technology disclosed in patent CN101486737A offers a transformative solution by modifying the ligand structure to include a quaternary ammonium salt group, which fundamentally alters its physicochemical properties without sacrificing catalytic performance. This novel design ensures that the ligand remains soluble in polar organic solvents like dichloromethane during the reaction but becomes insoluble in non-polar solvents such as diethyl ether or n-hexane, allowing for straightforward precipitation and recovery. 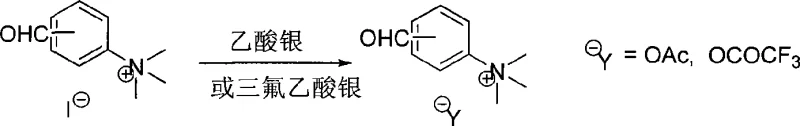 The synthesis of these advanced ligands is remarkably straightforward, involving the condensation of specific aromatic aldehyde quaternary ammonium salts with ferrocene amines under mild conditions. This simplicity in preparation translates directly to manufacturing advantages, as it eliminates the need for complex multi-step syntheses or harsh reaction conditions that often plague traditional catalyst production. By enabling a liquid-phase reaction with solid-phase recovery potential, this approach bridges the gap between the high activity of homogeneous catalysis and the ease of separation associated with heterogeneous systems, offering a compelling value proposition for cost-sensitive chemical manufacturing.
The synthesis of these advanced ligands is remarkably straightforward, involving the condensation of specific aromatic aldehyde quaternary ammonium salts with ferrocene amines under mild conditions. This simplicity in preparation translates directly to manufacturing advantages, as it eliminates the need for complex multi-step syntheses or harsh reaction conditions that often plague traditional catalyst production. By enabling a liquid-phase reaction with solid-phase recovery potential, this approach bridges the gap between the high activity of homogeneous catalysis and the ease of separation associated with heterogeneous systems, offering a compelling value proposition for cost-sensitive chemical manufacturing.
Mechanistic Insights into Ferrocene-Catalyzed Asymmetric Allylic Substitution
At the heart of this technology lies the unique structural architecture of the ferrocene phosphinimine ligand, which leverages the rigid planar chirality of the ferrocene skeleton to control the stereochemical outcome of the reaction. 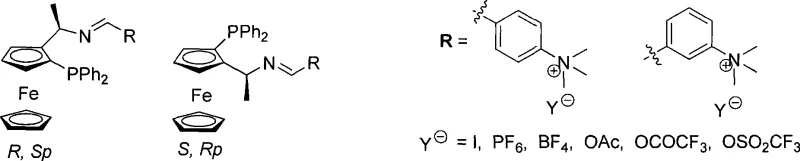 The presence of the quaternary ammonium group not only aids in solubility tuning but also influences the electronic environment around the phosphorus and nitrogen donor atoms, potentially enhancing the coordination stability with palladium centers. In the catalytic cycle, the ligand forms a chiral complex with allylpalladium chloride dimers, creating a sterically defined pocket that directs the nucleophilic attack of substrates like dimethyl malonate to one specific face of the allyl intermediate. This precise spatial arrangement is crucial for achieving high enantiomeric excess (ee) values, which are paramount for meeting the stringent regulatory requirements of the pharmaceutical industry regarding impurity profiles and optical purity. The robustness of the ferrocene backbone ensures thermal stability, allowing the catalyst to maintain its integrity throughout the reaction duration without significant decomposition.
The presence of the quaternary ammonium group not only aids in solubility tuning but also influences the electronic environment around the phosphorus and nitrogen donor atoms, potentially enhancing the coordination stability with palladium centers. In the catalytic cycle, the ligand forms a chiral complex with allylpalladium chloride dimers, creating a sterically defined pocket that directs the nucleophilic attack of substrates like dimethyl malonate to one specific face of the allyl intermediate. This precise spatial arrangement is crucial for achieving high enantiomeric excess (ee) values, which are paramount for meeting the stringent regulatory requirements of the pharmaceutical industry regarding impurity profiles and optical purity. The robustness of the ferrocene backbone ensures thermal stability, allowing the catalyst to maintain its integrity throughout the reaction duration without significant decomposition.
The application of this ligand system in asymmetric allylic substitution reactions demonstrates exceptional versatility and efficiency under mild operational parameters. 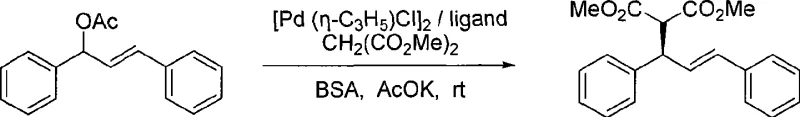 As detailed in the patent examples, the reaction proceeds smoothly at room temperature under an argon atmosphere, utilizing dichloromethane as the solvent and requiring only catalytic amounts of the chiral ligand relative to the substrate. The system effectively converts allylic acetates into substituted products with yields consistently exceeding 90% and enantiomeric excess values reaching up to 95.5% depending on the specific anion configuration. Such high levels of stereocontrol are achieved without the need for cryogenic conditions or excessive pressure, which significantly reduces the energy footprint of the process. For R&D directors, this mechanistic reliability means that method development timelines can be shortened, as the catalyst provides predictable and reproducible results across different batches of raw materials, thereby de-risking the scale-up phase for new drug candidates.
As detailed in the patent examples, the reaction proceeds smoothly at room temperature under an argon atmosphere, utilizing dichloromethane as the solvent and requiring only catalytic amounts of the chiral ligand relative to the substrate. The system effectively converts allylic acetates into substituted products with yields consistently exceeding 90% and enantiomeric excess values reaching up to 95.5% depending on the specific anion configuration. Such high levels of stereocontrol are achieved without the need for cryogenic conditions or excessive pressure, which significantly reduces the energy footprint of the process. For R&D directors, this mechanistic reliability means that method development timelines can be shortened, as the catalyst provides predictable and reproducible results across different batches of raw materials, thereby de-risking the scale-up phase for new drug candidates.
How to Synthesize Ferrocene Phosphinimine Ligand Efficiently
Implementing this catalytic technology in a production environment requires a clear understanding of the standardized synthesis protocol to ensure consistent quality and performance of the ligand. The process begins with the preparation of the key aromatic aldehyde quaternary ammonium salt intermediates, which serve as the foundation for the final ligand structure. Detailed standardized synthesis steps see the guide below, outlining the precise molar ratios, solvent choices, and workup procedures necessary to achieve the high purity levels required for effective catalysis. Adhering to these protocols ensures that the resulting ligand possesses the correct stereochemistry and anion composition to deliver the reported high yields and ee values in downstream applications.
- Prepare the aromatic aldehyde quaternary ammonium salt by reacting formyl-trimethylaniline iodide with anion exchange agents like ammonium hexafluorophosphate in distilled water at room temperature.
- Condense the prepared quaternary ammonium salt with 1-(2-diphenylphosphino)ferroceneethylamine in dichloromethane using a dehydrating agent such as anhydrous magnesium sulfate.
- Isolate the final yellow solid ligand by filtration, concentration, and washing with organic solvents like anhydrous ether to ensure high purity for catalytic use.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this quaternary ammonium ferrocene ligand technology presents a strategic opportunity to optimize operational expenditures and enhance supply chain resilience. The primary economic driver is the potential for catalyst recovery and reuse, which directly mitigates the cost burden associated with purchasing expensive chiral ligands for every production batch. By simplifying the separation process through solubility switching, manufacturers can reduce the consumption of raw materials and minimize waste generation, leading to substantial cost savings in the overall manufacturing budget. Furthermore, the operational simplicity of the reaction conditions reduces the dependency on specialized equipment capable of handling extreme temperatures or pressures, thereby lowering capital expenditure requirements for facility upgrades.
- Cost Reduction in Manufacturing: The elimination of complex immobilization supports and the potential for ligand recycling drastically simplify the production workflow, removing the need for expensive filtration media or chromatography steps often required to remove metal residues. This streamlining of the downstream processing phase translates into reduced labor hours and lower utility consumption, contributing to a more competitive cost structure for the final pharmaceutical intermediate. Additionally, the high catalytic activity means that lower loadings of the palladium precursor are required, further decreasing the input cost of precious metals which are subject to volatile market pricing. These cumulative efficiencies allow for a more robust margin profile even when facing fluctuations in raw material availability.
- Enhanced Supply Chain Reliability: The synthesis of the ligand relies on readily available starting materials such as ferrocene derivatives and common aromatic aldehydes, which reduces the risk of supply disruptions associated with exotic or single-source reagents. The robustness of the reaction conditions, which tolerate room temperature operations, ensures that production schedules are not vulnerable to delays caused by equipment failures related to heating or cooling systems. This reliability is critical for maintaining continuous supply to downstream API manufacturers, ensuring that project timelines are met without unexpected interruptions. Moreover, the stability of the ligand during storage simplifies logistics, as it does not require stringent temperature-controlled shipping conditions that often complicate international transport.
- Scalability and Environmental Compliance: The use of dichloromethane and standard workup procedures aligns well with existing infrastructure in most fine chemical plants, facilitating a seamless transition from laboratory scale to commercial tonnage production. The ability to precipitate the catalyst or product using non-polar solvents reduces the volume of aqueous waste streams, simplifying wastewater treatment and ensuring compliance with increasingly strict environmental regulations. This environmental advantage is becoming a key differentiator in vendor selection processes, as multinational corporations prioritize suppliers who demonstrate a commitment to green chemistry principles. The high atom economy of the substitution reaction further supports sustainability goals by maximizing the conversion of raw materials into the desired product with minimal byproduct formation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ferrocene-based catalytic system in industrial settings. These insights are derived directly from the experimental data and beneficial effects described in the underlying patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: How does the quaternary ammonium group improve catalyst recovery?
A: The quaternary ammonium group imparts specific solubility properties, making the ligand soluble in dichloromethane but insoluble in ethers or hexanes, allowing for easy precipitation and recycling without complex immobilization on solid supports.
Q: What enantiomeric excess (ee) values are achievable with this ligand?
A: According to patent data, the ligand achieves ee values ranging from 60.5% to 95.5% in asymmetric allylic substitution reactions, depending on the specific anion and stereochemistry employed.
Q: Is this catalytic system suitable for large-scale manufacturing?
A: Yes, the process operates at room temperature under argon protection with simple workup procedures, eliminating the need for extreme conditions and facilitating scalable production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ferrocene Phosphinimine Ligand Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercially viable chemical solutions, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses the expertise to adapt the synthesis of complex ferrocene ligands to meet stringent purity specifications, ensuring that every batch delivered meets the rigorous quality standards required by the global pharmaceutical industry. We operate state-of-the-art rigorous QC labs equipped to verify enantiomeric excess and metal residue levels, providing our partners with the confidence that their supply chain is secure and compliant. Our commitment to quality assurance extends beyond mere specification testing, encompassing a holistic approach to process safety and consistency that safeguards your production timelines.
We invite you to engage with our technical procurement team to discuss how this innovative catalytic technology can be tailored to your specific process requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the potential economic benefits of switching to this recoverable ligand system for your allylic substitution reactions. We encourage you to contact us to obtain specific COA data and route feasibility assessments that will demonstrate the practical viability of this approach for your projects. Partnering with us ensures access to cutting-edge chemical technologies backed by reliable manufacturing capabilities and a dedication to long-term collaborative success.
