Advanced Ferrocene Phosphinimine Ligands for High-Efficiency Asymmetric Allylic Substitution Manufacturing
Advanced Ferrocene Phosphinimine Ligands for High-Efficiency Asymmetric Allylic Substitution Manufacturing
The landscape of asymmetric synthesis is constantly evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. A significant breakthrough in this domain is documented in patent CN101712697B, which introduces a novel class of ferrocene phosphinimine ligands functionalized with imidazolium salt groups. This innovation addresses a critical bottleneck in organometallic catalysis: the recovery and recycling of expensive chiral ferrocene catalysts. Traditionally, ferrocene-based ligands, while highly effective, suffer from high solubility in organic solvents, making their separation from reaction products difficult and costly. The integration of an imidazolium moiety transforms these ligands into ionic liquid-soluble species, bridging the gap between homogeneous catalytic efficiency and heterogeneous ease of separation. This technological advancement offers a compelling value proposition for manufacturers seeking to optimize their production of complex chiral building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
In the realm of asymmetric allylic substitution, a cornerstone reaction for constructing carbon-carbon bonds, traditional ferrocene phosphinimine ligands have long been valued for their stereochemical induction capabilities. However, their industrial application has been severely hampered by downstream processing challenges. Because these organometallic complexes are typically soluble in most common organic reaction media, isolating the product often requires extensive chromatography or multiple extraction steps, leading to significant product loss and increased operational expenditure. Furthermore, attempts to immobilize these catalysts on solid supports like silica gel or polymers to facilitate recovery have frequently resulted in diminished catalytic activity due to steric hindrance and reduced mass transfer rates. This trade-off between recoverability and reactivity has forced process chemists to accept lower overall yields or higher costs when scaling up these critical transformations.
The Novel Approach
The methodology outlined in the patent presents a sophisticated solution by modifying the ligand architecture rather than the support matrix. By incorporating an imidazolium salt group onto the ferrocene backbone, the resulting ligand exhibits unique solubility profiles akin to ionic liquids. This allows the catalytic system to operate homogeneously, ensuring rapid reaction kinetics and high selectivity, while enabling separation through simple solvent extraction or phase separation techniques post-reaction. The catalyst can theoretically be retained in the ionic phase and reused, drastically improving atom economy. The core reaction facilitated by this system is the asymmetric allylic substitution, where the ligand coordinates with palladium to direct the nucleophilic attack with high precision. This approach not only simplifies the workflow but also enhances the sustainability profile of the synthesis by reducing solvent waste and metal contamination in the final API intermediate.
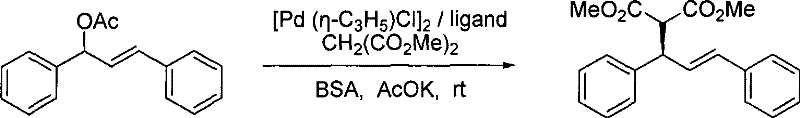
Mechanistic Insights into Ferrocene-Catalyzed Asymmetric Allylic Substitution
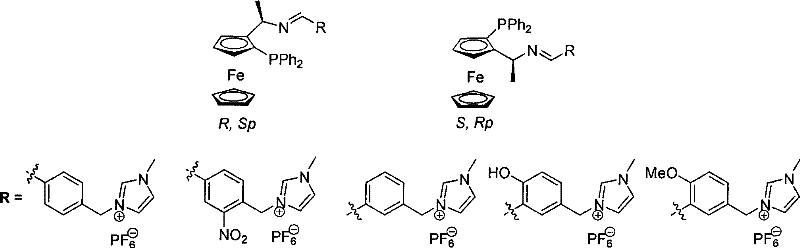
The efficacy of these imidazolium-functionalized ligands stems from the intricate interplay between the planar chirality of the ferrocene backbone and the electronic properties of the phosphinimine-imidazolium framework. The ferrocene unit provides a rigid, chiral environment that dictates the spatial orientation of the substrate within the palladium coordination sphere. Specifically, the (R, Sp) and (S, Rp) configurations create distinct chiral pockets that favor the formation of one enantiomer over the other. The phosphinimine nitrogen and the phosphorus atom act as bidentate donors, stabilizing the cationic pi-allyl palladium intermediate. Simultaneously, the imidazolium group, while primarily serving a solubility function, may also exert secondary electronic effects that stabilize the transition state through weak interactions or by modulating the electron density at the metal center. This dual functionality ensures that the catalytic cycle proceeds with minimal energy barriers, leading to the high turnover frequencies observed in the experimental data.
From an impurity control perspective, the high stereoselectivity of this system is paramount for pharmaceutical manufacturing. The patent data demonstrates that specific ligand variants can achieve enantiomeric excess (ee) values as high as 95.5%, significantly reducing the burden on downstream purification processes such as chiral resolution or recrystallization. The robustness of the ligand under ambient conditions also minimizes the formation of decomposition byproducts that often plague sensitive organometallic reactions. By operating at room temperature and avoiding harsh acidic or basic conditions during the catalytic step, the integrity of sensitive functional groups on the substrate is preserved. This results in a cleaner crude reaction profile, which is a critical factor for regulatory compliance and cost-effective production of high-purity pharmaceutical intermediates.
How to Synthesize Ferrocene Phosphinimine Ligands Efficiently
The synthesis of these advanced ligands is designed to be operationally simple, avoiding the need for cryogenic conditions or exotic reagents, which facilitates easy technology transfer to pilot and production scales. The process begins with the quaternization of N-methylimidazole with substituted benzaldehyde derivatives to form the imidazolium salt precursor, followed by an anion exchange to introduce the hexafluorophosphate counterion. This salt is then condensed with 1-(2-diphenylphosphino)ferrocenylethylamine in the presence of a mild dehydrating agent. The detailed standardized synthesis steps for producing these ligands with consistent quality are provided in the guide below.
- React aromatic aldehyde compounds with N-Methylimidazole to form imidazolium halide salts, followed by anion exchange with ammonium hexafluorophosphate.
- Dissolve the resulting imidazolium salt and 1-(2-diphenylphosphino)ferrocenylethylamine in dichloromethane.
- Add anhydrous magnesium sulfate as a dehydrating agent, stir at room temperature, and purify the resulting yellow solid ligand via organic solvent washing.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ligand technology translates into tangible strategic benefits beyond mere chemical performance. The primary advantage lies in the potential for significant cost reduction in asymmetric synthesis manufacturing. By enabling catalyst recycling and simplifying product isolation, the overall consumption of expensive palladium and chiral ligands is drastically reduced. This eliminates the need for costly metal scavenging resins or complex purification trains, directly lowering the cost of goods sold (COGS). Furthermore, the use of readily available starting materials such as ferrocenyl amines and common aromatic aldehydes ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or scarce reagents.
- Cost Reduction in Manufacturing: The ability to recover and reuse the chiral ligand due to its ionic liquid-like properties represents a major shift in process economics. Traditional homogeneous catalysts are often single-use, representing a substantial portion of raw material costs. With this new system, the effective cost per kilogram of product decreases significantly as the catalyst turnover number increases. Additionally, the simplified workup procedure reduces solvent consumption and waste disposal costs, contributing to a leaner and more profitable manufacturing process without compromising on yield or purity.
- Enhanced Supply Chain Reliability: The synthetic route for these ligands relies on commodity chemicals and standard unit operations, making it highly scalable and less susceptible to supply disruptions. Unlike complex biocatalysts or highly specialized organocatalysts that may have limited global suppliers, the precursors for these ferrocene ligands are produced by multiple chemical manufacturers worldwide. This diversity in the supply base ensures continuity of supply, allowing production planners to secure long-term contracts with confidence and avoid the bottlenecks that often delay new product launches in the competitive pharmaceutical market.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are mild, typically proceeding at room temperature in common solvents like dichloromethane. This lack of extreme thermal or pressure requirements simplifies reactor design and safety protocols, facilitating seamless scale-up from laboratory grams to multi-ton commercial production. Moreover, the potential for catalyst recycling aligns with green chemistry principles by minimizing heavy metal waste. This environmental advantage is increasingly critical for meeting stringent regulatory standards and corporate sustainability goals, positioning companies that adopt this technology as leaders in responsible chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ferrocene phosphinimine ligand technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical capabilities of the system for potential partners and licensees.
Q: How does the imidazolium group improve catalyst recovery compared to traditional ferrocene ligands?
A: The imidazolium salt group imparts ionic liquid-like solubility characteristics, allowing the catalyst to remain in a specific phase or be easily separated via extraction, overcoming the difficulty of recovering soluble ferrocene derivatives.
Q: What level of enantioselectivity can be achieved with these ligands in allylic substitution?
A: Experimental data indicates that specific configurations of these ligands can achieve enantiomeric excess (ee) values exceeding 95%, demonstrating superior stereocontrol compared to many conventional systems.
Q: Are these ligands stable under standard industrial reaction conditions?
A: Yes, the patent highlights that these compounds possess high thermal stability and maintain catalytic activity under room temperature conditions, making them robust for various synthetic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ferrocene Phosphinimine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the imidazolium-functionalized ferrocene ligands described in CN101712697B. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of ligand or intermediate meets the exacting standards required for global pharmaceutical registration.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific process needs, demonstrating how this ligand system can optimize your production economics. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with superior chiral solutions.
