Advanced Trospium Chloride Synthesis via Pd/C Catalysis for Commercial Scale-Up
Introduction to Advanced Trospium Chloride Manufacturing
The pharmaceutical industry constantly seeks robust synthetic pathways that balance high purity with environmental sustainability, particularly for complex antimuscarinic agents like Trospium Chloride. Patent CN102718760B introduces a transformative synthesis technique that addresses critical bottlenecks in traditional manufacturing, offering a streamlined four-step process that begins with the preparation of benziloyl chloride and culminates in the final condensation reaction. This methodology represents a significant departure from legacy routes by integrating catalytic hydrogenation for key intermediate transformations, thereby enhancing atom economy and reducing the ecological footprint of production. For R&D directors and procurement specialists, understanding this patented approach is essential for securing a reliable trospium chloride supplier capable of meeting stringent quality standards while optimizing operational expenditures. The strategic implementation of palladium-catalyzed demethylation not only improves yield but also simplifies the purification landscape, ensuring a consistent supply of high-purity trospium chloride for global markets.
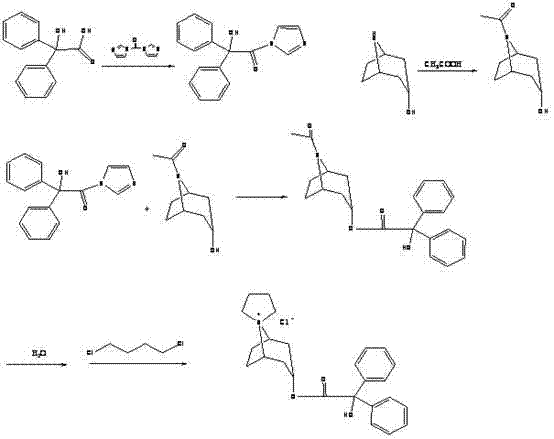
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Trospium Chloride has been plagued by inefficient methodologies that rely on harsh chemical oxidants or expensive starting materials, creating substantial barriers to cost-effective manufacturing. Traditional routes often necessitate the direct purchase of nortropine, a precursor that is notoriously difficult to source domestically and commands a premium price due to its complex supply chain. Furthermore, alternative demethylation strategies employing potassium permanganate oxidation generate vast quantities of manganese sludge, posing severe environmental hazards and requiring elaborate waste treatment protocols that inflate production costs. Other methods utilizing triphenylphosphine photo-oxidation suffer from extended reaction cycles and low throughput, rendering them unsuitable for the high-volume demands of modern pharmaceutical supply chains. These legacy processes are characterized by poor atom economy and cumbersome post-reaction workups, which collectively erode profit margins and compromise the reliability of supply for critical urological medications.
The Novel Approach
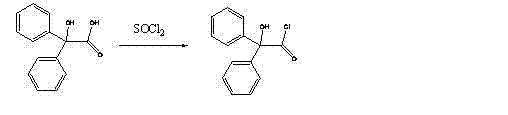
In stark contrast, the novel approach detailed in the patent leverages a concise synthetic strategy that circumvents these historical inefficiencies by generating nortropine in-situ from the more accessible alpha-tropanol. This paradigm shift allows manufacturers to bypass the procurement of costly nortropine entirely, instead utilizing a catalytic hydrogenation step that is both economically viable and environmentally benign. The process initiates with the efficient conversion of benzilic acid to benziloyl chloride using thionyl chloride, establishing a reactive acylating agent under controlled conditions. Subsequently, the core innovation lies in the Pd/C catalyzed demethylation, which operates under mild pressure and temperature to selectively remove the N-methyl group without degrading the sensitive tropane skeleton. This streamlined workflow minimizes unit operations, reduces solvent consumption, and facilitates a smoother transition from laboratory scale to commercial production, positioning it as a superior choice for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Pd/C Catalytic Hydrogenation Demethylation
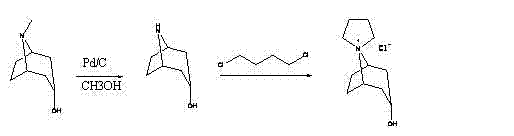
The cornerstone of this advanced synthesis is the mechanistic elegance of the palladium-on-carbon (Pd/C) catalyzed hydrogenolysis, which facilitates the selective cleavage of the N-methyl bond in alpha-tropanol. Unlike oxidative methods that risk over-oxidation of the tropane ring or the hydroxyl group, catalytic hydrogenation offers exceptional chemoselectivity, preserving the structural integrity of the bicyclic amine while efficiently liberating methane or methyl species as benign byproducts. The mechanism involves the adsorption of the tertiary amine onto the palladium surface, where activated hydrogen species attack the steric environment around the nitrogen atom, promoting bond scission with high fidelity. This precision is critical for maintaining the stereochemical purity of the resulting nortropine, which is a prerequisite for the biological activity of the final Trospium Chloride API. By operating at moderate temperatures ranging from 45°C to 60°C and under controlled hydrogen pressure, the reaction kinetics are optimized to maximize conversion rates while minimizing the formation of side products that could complicate downstream purification.
Furthermore, the impurity profile generated by this catalytic route is significantly cleaner compared to oxidative alternatives, directly impacting the ease of isolation and the final quality of the drug substance. The absence of heavy metal oxidants like manganese eliminates the risk of metal contamination, a critical parameter for regulatory compliance in pharmaceutical manufacturing. The catalyst itself can be recovered and reused multiple times through simple filtration, contributing to a circular economy model within the plant and reducing the consumption of precious metals. This mechanistic advantage translates into a robust process window where variations in raw material quality can be tolerated without compromising the yield or purity of the chlorinated quaternary ammonium salt intermediate. Consequently, the overall process stability is enhanced, ensuring that the commercial scale-up of complex antimuscarinic agents remains predictable and reproducible across different production batches.
How to Synthesize Trospium Chloride Efficiently
Executing this synthesis requires precise control over reaction parameters to leverage the full benefits of the patented methodology, starting with the activation of benzilic acid and proceeding through the catalytic transformation of the tropane scaffold. The protocol dictates a specific molar ratio of reagents, such as maintaining a benzilic acid to thionyl chloride weight ratio of approximately 1:2.6 to 3, to ensure complete conversion to the acid chloride without excessive reagent waste. Following this, the hydrogenation step utilizes a catalyst loading of 1% to 1.5% relative to the substrate weight, balanced with hydrochloric acid to facilitate the demethylation under a hydrogen atmosphere. The subsequent quaternization with 1,4-dichlorobutane must be carefully managed at temperatures between 45°C and 50°C to promote the formation of the spiro-ammonium salt while preventing polymerization or degradation. Detailed standard operating procedures for each unit operation are essential for maintaining consistency, and the comprehensive synthesis steps are outlined below for technical reference.
- Preparation of benziloyl chloride by reacting benzilic acid with thionyl chloride.
- Synthesis of nortropine via Pd/C catalytic hydrogenation demethylation of alpha-tropanol.
- Formation of the quaternary ammonium salt using nortropine and 1,4-dichlorobutane.
- Final condensation of benziloyl chloride with the quaternary salt to yield Trospium Chloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers profound strategic advantages that extend beyond mere technical feasibility, fundamentally altering the cost structure and reliability of the supply base. By shifting the starting material from expensive nortropine to the more commoditized alpha-tropanol, the process decouples production costs from the volatile pricing of specialized intermediates, leading to substantial cost savings in the long term. The elimination of hazardous oxidants not only reduces waste disposal fees but also mitigates regulatory risks associated with environmental compliance, ensuring uninterrupted production schedules even in regions with strict ecological mandates. Additionally, the ability to recover and reuse the Pd/C catalyst multiple times drastically lowers the consumption of precious metals, further enhancing the economic viability of the process without sacrificing reaction efficiency. These factors combine to create a resilient supply chain capable of withstanding market fluctuations and delivering high-purity trospium chloride with reduced lead time for high-purity trospium chloride.
- Cost Reduction in Manufacturing: The primary driver of cost efficiency in this process is the substitution of costly nortropine with alpha-tropanol, coupled with the reusable nature of the heterogeneous Pd/C catalyst. By avoiding the purchase of premium-priced intermediates and minimizing catalyst turnover, manufacturers can achieve a significantly lower cost of goods sold (COGS). The simplified workup procedures, which omit complex extraction and heavy metal removal steps, further reduce labor and utility expenses, allowing for competitive pricing strategies in the global marketplace.
- Enhanced Supply Chain Reliability: Sourcing alpha-tropanol is generally more stable and less prone to disruption than sourcing nortropine, which is often a bottleneck in traditional supply chains. The robustness of the catalytic hydrogenation step ensures high yields and consistent quality, reducing the incidence of batch failures that can delay shipments. This reliability is crucial for maintaining continuous inventory levels for downstream API manufacturers, ensuring that patient demand for urological treatments is met without interruption.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are easily transferable from pilot plants to multi-ton reactors. The absence of toxic manganese sludge and the reduction in solvent usage align with green chemistry principles, facilitating easier permitting and operation in environmentally sensitive jurisdictions. This compliance capability future-proofs the manufacturing asset against tightening global regulations on chemical waste and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity for stakeholders evaluating its potential for integration into their existing portfolios. These insights are derived directly from the patent specifications and practical considerations for industrial application, ensuring that decision-makers have accurate information regarding process capabilities and limitations. Understanding these nuances is vital for assessing the feasibility of adopting this route for commercial production and for engaging in informed discussions with technical partners.
Q: What are the advantages of using Pd/C catalytic hydrogenation over traditional oxidation methods?
A: The Pd/C catalytic hydrogenation method eliminates the use of toxic oxidants like potassium permanganate, thereby avoiding heavy metal pollution and simplifying post-reaction treatment significantly.
Q: How does this process improve cost efficiency for Trospium Chloride manufacturing?
A: By synthesizing nortropine in-situ from the more readily available alpha-tropanol, the process bypasses the need to purchase expensive nortropine directly, substantially lowering raw material costs.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process features short reaction steps, high atom economy, and reusable catalysts, making it highly scalable and compliant with modern environmental regulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trospium Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable manufacturing processes in the modern pharmaceutical landscape, and we are uniquely positioned to leverage this advanced Trospium Chloride synthesis technology for our global clients. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and speed. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Trospium Chloride meets the highest international standards for safety and efficacy. Our commitment to excellence extends beyond mere compliance; we actively optimize our processes to deliver superior value through enhanced yield and reduced environmental impact.
We invite you to collaborate with us to explore how this innovative synthesis route can benefit your specific supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project requirements, demonstrating the tangible economic advantages of switching to this catalytic method. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to secure a reliable and cost-effective supply of this vital urological intermediate.
