Advanced Metal-Free Synthesis of Alpha-Thiocyanones for Commercial Scale-Up
Advanced Metal-Free Synthesis of Alpha-Thiocyanones for Commercial Scale-Up
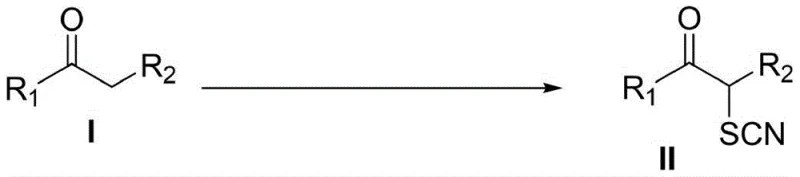
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes that balance efficiency with regulatory compliance. A significant breakthrough in this domain is detailed in patent CN113861087A, which discloses a novel method for preparing alpha-thiocyanatone compounds under metal-free and additive-free catalytic reaction conditions. This technology utilizes alpha-H-containing ketone derivatives, elemental sulfur, and trimethylsilyl cyanide (TMSCN) as cyano-group sources to directly introduce thiocyano groups. The significance of this development cannot be overstated for a reliable pharmaceutical intermediate supplier, as it offers a pathway to obtain good yields with a wider substrate range while adhering to green chemistry principles. By eliminating the need for transition metal catalysts, this process addresses critical pain points regarding metal residue limits in final drug substances, thereby streamlining the purification workflow and enhancing the overall economic viability of producing these valuable scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-thiocyanoketones has relied heavily on the use of toxic thiocyanating reagents such as thiocyanogen or metal-catalyzed systems involving copper or iron salts. These conventional approaches often suffer from significant drawbacks, including the generation of hazardous waste streams and the stringent requirement for removing trace metal impurities to meet International Council for Harmonisation (ICH) guidelines. Furthermore, many traditional methods exhibit poor functional group tolerance, leading to side reactions that compromise the purity profile of the final high-purity alpha-thiocyanone product. The reliance on expensive catalysts and harsh reaction conditions also inflates the cost of goods sold (COGS), creating a bottleneck for cost reduction in API manufacturing. Additionally, the handling of unstable thiocyanating agents poses safety risks during large-scale operations, limiting the ability of supply chain managers to ensure consistent continuity of supply for downstream drug development projects.
The Novel Approach
In stark contrast, the methodology described in CN113861087A leverages elemental sulfur and TMSCN in a dimethyl sulfoxide (DMSO) solvent system to achieve direct thiocyanation. This innovative strategy operates under air atmosphere at moderate temperatures, typically between 60-120°C, with an optimal range of 80-100°C. As illustrated in the specific optimization studies, this approach achieves exceptional conversion rates, with model substrates like propiophenone yielding up to 98% of the desired product. The absence of metal catalysts not only simplifies the downstream processing by removing the need for scavenger resins but also significantly reduces the environmental footprint of the synthesis. This metal-free protocol represents a paradigm shift for the commercial scale-up of complex pharmaceutical intermediates, offering a safer, more economical, and environmentally benign alternative that aligns perfectly with modern sustainable manufacturing goals.

Mechanistic Insights into Metal-Free Thiocyanation
The mechanistic pathway of this transformation involves the activation of elemental sulfur by the ketone substrate in the presence of TMSCN, facilitating the formation of the carbon-sulfur bond without external metal promotion. While the exact radical or ionic nature may vary depending on the specific substrate electronics, the process effectively generates the thiocyano moiety at the alpha-position of the carbonyl group. This direct functionalization avoids the need for pre-functionalized starting materials, such as alpha-halo ketones, which are often corrosive and difficult to handle on a multi-ton scale. The reaction proceeds smoothly in polar aprotic solvents like DMSO, which stabilizes the transition states and enhances the solubility of the inorganic sulfur species. For R&D directors, understanding this mechanism is crucial as it highlights the robustness of the reaction against varying electronic environments, ensuring consistent performance across a diverse library of ketone derivatives used in medicinal chemistry campaigns.
From an impurity control perspective, the metal-free nature of this catalysis is a decisive advantage. In traditional metal-catalyzed C-H functionalization, residual metals like palladium, copper, or iron can coordinate with the product or persist through crystallization steps, necessitating additional purification stages that erode yield. By utilizing organic-soluble reagents like TMSCN and elemental sulfur, the impurity profile is dominated by organic by-products that are generally easier to separate via standard extraction or chromatography. This results in a cleaner crude reaction mixture, allowing for higher recovery rates during isolation. Consequently, the process supports the production of high-purity OLED material precursors or pharmaceutical intermediates with minimal risk of metal contamination, thereby reducing the analytical burden on quality control laboratories and accelerating the release of batches for clinical trial material production.
How to Synthesize Alpha-Thiocyanone Efficiently
To implement this synthesis effectively, one must adhere to the optimized parameters established in the patent data to maximize yield and minimize side products. The process begins by charging a reactor with the specific ketone derivative, finely powdered sulfur, and TMSCN in a molar ratio that ensures complete consumption of the limiting reagent, typically favoring an excess of sulfur and cyanide source. The reaction mixture is then heated to the optimal temperature of 80°C under an air atmosphere, where it is stirred for approximately 12 hours to ensure full conversion. Following the reaction, a straightforward aqueous workup involving quenching with water and extraction with ethyl acetate allows for the separation of the organic product. The detailed standardized synthetic steps, including specific stoichiometric ratios and purification protocols for various substrates, are outlined below to guide process chemists in replicating these results.
- Mix ketone substrate, sulfur powder, and TMSCN in DMSO solvent within a reactor equipped with magnetic stirring.
- Heat the reaction mixture to 80-100°C under air atmosphere and stir for approximately 12 hours until completion.
- Quench with water, extract with ethyl acetate, concentrate, and purify via silica gel chromatography to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route offers tangible strategic benefits that extend beyond simple chemical transformation. The primary advantage lies in the drastic simplification of the raw material supply chain; elemental sulfur is a commodity chemical available in vast quantities globally, and TMSCN is a standard reagent in fine chemical inventories. This contrasts sharply with proprietary metal catalysts that may have long lead times or single-source dependencies. By shifting to this open innovation model, companies can mitigate supply risk and negotiate better pricing due to the commoditization of inputs. Furthermore, the elimination of expensive catalyst recovery systems and metal scavengers translates directly into lower operational expenditures, supporting broader initiatives for cost reduction in electronic chemical manufacturing and pharma sectors alike.
- Cost Reduction in Manufacturing: The economic impact of removing transition metal catalysts from the process cannot be understated, as it eliminates the capital expenditure associated with specialized filtration equipment and the recurring cost of scavenger resins. Without the need to drive metal levels down to parts-per-million specifications, the number of processing steps is reduced, leading to significant savings in labor, energy, and solvent consumption. This streamlined workflow enhances the overall throughput of the manufacturing facility, allowing for faster turnover of batches and improved asset utilization. Consequently, the total cost of ownership for producing these intermediates is substantially lowered, providing a competitive edge in price-sensitive markets where margin compression is a constant challenge for generic drug manufacturers.
- Enhanced Supply Chain Reliability: Utilizing stable, shelf-stable reagents like sulfur powder and TMSCN ensures that production schedules are not disrupted by the degradation of sensitive catalytic species. The reaction's tolerance to air atmosphere further simplifies the engineering requirements, as there is no need for rigorous inert gas blanketing or glovebox conditions, which are prone to failure and maintenance issues. This robustness allows for greater flexibility in manufacturing site selection, enabling production in facilities that may lack advanced anaerobic capabilities. For supply chain planners, this means reducing lead time for high-purity intermediates and ensuring a steady flow of materials to downstream formulation units, thereby safeguarding against stockouts that could delay critical drug launches or agrochemical seasons.
- Scalability and Environmental Compliance: The green chemistry credentials of this process align perfectly with increasingly stringent environmental regulations regarding heavy metal discharge and solvent waste. By avoiding toxic metal salts, the wastewater treatment burden is significantly lightened, reducing the cost and complexity of effluent management. The high atom economy and excellent yields observed in the substrate scope, often exceeding 90% for aryl ketones, mean that less raw material is wasted as by-product, contributing to a more sustainable manufacturing lifecycle. This environmental stewardship not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the organization, making it a more attractive partner for multinational corporations committed to sustainability goals in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alpha-thiocyanation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear picture of what process teams can expect during technology transfer. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: Does this synthesis method involve heavy metal catalysts?
A: No, the patented process (CN113861087A) operates under strictly metal-free and additive-free conditions, utilizing elemental sulfur and TMSCN, which eliminates the need for costly heavy metal removal steps required by ICH Q3D guidelines.
Q: What is the substrate scope for this alpha-thiocyanation reaction?
A: The method demonstrates broad compatibility with various ketone derivatives containing alpha-hydrogens, including aryl ketones, heteroaryl ketones, cyclic ketones, and alkenyl ketones, maintaining yields above 70% even with steric hindrance.
Q: Is the process scalable for industrial production?
A: Yes, the reaction uses inexpensive reagents like sulfur powder and runs under air atmosphere at moderate temperatures (80°C), making it highly suitable for commercial scale-up without requiring specialized inert gas equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Thiocyanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthetic methodologies in modernizing the production of critical chemical building blocks. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the CN113861087A process are translated into reliable industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of alpha-thiocyanone delivered meets the highest standards required for pharmaceutical and agrochemical applications. We are committed to leveraging our technical expertise to optimize these green routes further, ensuring maximum efficiency and minimal environmental impact for our global clientele.
We invite you to collaborate with us to explore how this advanced synthesis method can enhance your product portfolio and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your long-term supply chain stability and growth objectives in the competitive fine chemicals market.
