Revolutionizing Alpha-Thiocyanone Production: A Metal-Free Strategy for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, environmentally benign synthetic routes for high-value intermediates. A groundbreaking development in this sector is detailed in patent CN113861087A, which discloses a novel, metal-free method for the preparation of α-thiocyanone compounds. These compounds are critical building blocks in medicinal chemistry, serving as versatile precursors for synthesizing trifluoromethyl thioethers, disulfides, and pharmacologically active 2-aminothiazole scaffolds. Traditionally, accessing these motifs often required harsh conditions or toxic reagents, but this new methodology leverages elemental sulfur and trimethylsilyl cyanide (TMSCN) to directly introduce the thiocyano group onto ketone derivatives. This approach not only simplifies the synthetic workflow but also aligns perfectly with modern green chemistry principles, offering a pathway to high-purity intermediates essential for drug discovery and development.
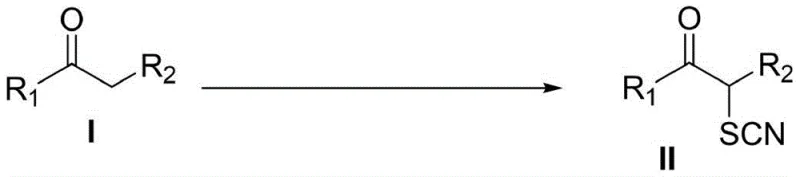
This technological leap represents a significant shift from conventional practices, addressing long-standing pain points in organic synthesis. By utilizing α-H-containing ketone derivatives as the starting material, the reaction proceeds under additive-free catalytic conditions, achieving remarkable yields without the burden of heavy metal contamination. For R&D directors and process chemists, this implies a streamlined path from bench to pilot plant, reducing the complexity of purification and ensuring that the final active pharmaceutical ingredients (APIs) derived from these intermediates maintain exceptional quality profiles. The broad substrate scope further enhances its utility, allowing for the functionalization of diverse aromatic and aliphatic ketones, thereby expanding the chemical space available for novel drug design.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of α-thiocyanoketones has been fraught with challenges that hinder efficient large-scale manufacturing. Traditional protocols frequently rely on transition metal catalysts or hazardous thiocyanating agents that pose significant safety and environmental risks. The use of heavy metals introduces a critical bottleneck: the rigorous requirement for metal removal to meet International Council for Harmonisation (ICH) guidelines, specifically ICH Q3D, regarding elemental impurities. This necessitates additional downstream processing steps, such as scavenging or complex chromatographic separations, which drastically increase production costs and extend lead times. Furthermore, many existing methods suffer from limited substrate tolerance, failing to accommodate sterically hindered or electronically diverse ketones, which restricts their applicability in the synthesis of complex drug candidates. The reliance on stoichiometric oxidants or toxic solvents in older methodologies also complicates waste management, creating substantial liabilities for chemical manufacturers aiming for sustainable operations.
The Novel Approach
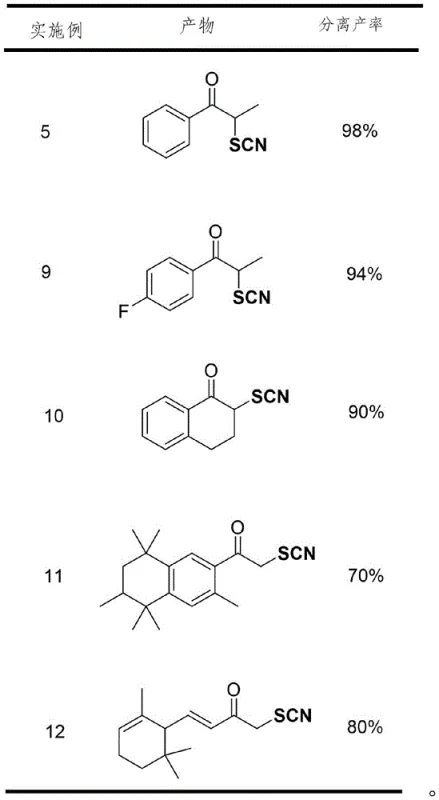
In stark contrast, the method described in patent CN113861087A offers a transformative solution by employing a metal-free, additive-free strategy. The core innovation lies in the direct utilization of elemental sulfur (S8) and TMSCN as the sulfur and cyano sources, respectively, mediated simply by heat in a polar aprotic solvent like DMSO. This eliminates the need for expensive and toxic metal catalysts entirely, thereby removing the associated purification burdens. The reaction conditions are remarkably mild yet effective, typically operating at temperatures between 60-120°C, with optimal results observed around 80°C. As demonstrated in the experimental data, this approach exhibits exceptional functional group compatibility, tolerating halogens, alkyl groups, and various ring systems without degradation. The result is a highly efficient process that delivers isolated yields often exceeding 90%, as evidenced by the comprehensive substrate screening which shows consistent performance across a wide array of structurally distinct ketones.
Mechanistic Insights into Metal-Free Thiocyanation
Understanding the mechanistic underpinnings of this transformation is crucial for process optimization and scale-up. The reaction likely proceeds through a radical or nucleophilic pathway where the acidic α-hydrogen of the ketone is activated, facilitating the insertion of elemental sulfur. The use of DMSO as a solvent plays a pivotal role, not just as a medium, but potentially as a mild oxidant or activator that assists in the generation of reactive sulfur species. Following sulfur insertion, the TMSCN acts as a safe and manageable source of the cyanide anion, which attacks the sulfur-activated intermediate to form the final C-S-CN bond. This stepwise construction avoids the formation of unstable intermediates that often plague direct thiocyanation methods. The absence of external oxidants or metal promoters suggests a self-sustaining cycle driven by the thermodynamic stability of the formed thiocyanate bond and the driving force provided by the silyl group stabilization. This mechanistic simplicity is a key factor in the reaction's robustness, minimizing side reactions such as over-oxidation or polymerization that commonly reduce yields in traditional protocols.
From an impurity control perspective, this metal-free mechanism offers distinct advantages for regulatory compliance. Since no transition metals are introduced into the reaction matrix, the risk of generating metal-complexed impurities is non-existent. This significantly simplifies the impurity profile of the crude product, making the subsequent purification via standard silica gel chromatography highly effective. The primary byproducts are likely volatile siloxanes or unreacted starting materials, which are easily removed during the aqueous workup and concentration steps. For quality control teams, this translates to a cleaner final product with fewer unidentified peaks in HPLC or NMR analysis. The high selectivity of the reaction ensures that sensitive functional groups on the aromatic rings, such as fluorine or nitro groups, remain intact, preserving the integrity of the molecular scaffold required for downstream biological activity. This level of precision is essential for producing high-purity pharmaceutical intermediates where even trace impurities can alter the efficacy or safety of the final drug product.
How to Synthesize Alpha-Thiocyanone Efficiently
Implementing this synthesis in a laboratory or pilot setting requires adherence to specific parameters to maximize yield and safety. The protocol is designed for operational simplicity, utilizing standard glassware and commercially available reagents. The reaction is typically conducted under an air atmosphere, removing the need for inert gas lines or gloveboxes, which further reduces operational complexity. The workup procedure is straightforward, involving a simple aqueous quench followed by organic extraction, making it amenable to automation or continuous flow processing. For detailed procedural specifics, including precise molar ratios and temperature ramps, operators should refer to the standardized guidelines derived from the patent examples.
- Combine ketone derivative, sulfur powder, and TMSCN in DMSO solvent within a magnetic stirring reactor.
- Heat the reaction mixture to 80-100°C and stir under air atmosphere for 5-20 hours until completion.
- Quench with water, extract with ethyl acetate, concentrate, and purify via silica gel flash chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this metal-free synthesis route presents compelling economic and logistical benefits. The elimination of precious metal catalysts directly impacts the bill of materials, removing a significant cost driver associated with palladium, copper, or rhodium complexes. Moreover, the reliance on commodity chemicals like elemental sulfur and TMSCN ensures a stable and resilient supply chain, as these reagents are produced globally in massive quantities and are not subject to the geopolitical volatility often seen with rare earth metals or specialized ligands. The simplified workup procedure also reduces the consumption of auxiliary materials such as scavenger resins or specialized filtration media, contributing to overall cost reduction in pharmaceutical intermediate manufacturing. By streamlining the production process, companies can achieve faster turnaround times from order to delivery, enhancing responsiveness to market demands.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the complete removal of transition metal catalysts. In traditional synthesis, these catalysts can account for a substantial portion of raw material costs, and their removal requires expensive scavenging agents. By switching to this metal-free protocol, manufacturers eliminate both the cost of the catalyst and the cost of its removal. Additionally, the use of DMSO, a low-cost, high-boiling solvent, allows for reactions to proceed efficiently without the need for cryogenic cooling or high-pressure equipment, further lowering energy and capital expenditure. The high yields reported, often approaching quantitative levels, minimize waste and maximize the throughput of the reactor, effectively lowering the cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available reagents. Elemental sulfur is a byproduct of the petroleum industry and is abundant, while TMSCN is a standard reagent in organic synthesis with multiple global suppliers. This diversity of supply sources mitigates the risk of single-source bottlenecks that can halt production lines. Furthermore, the reaction's tolerance to air and moisture simplifies storage and handling requirements, reducing the need for specialized infrastructure. This robustness ensures that production schedules can be maintained consistently, reducing lead time for high-purity pharmaceutical intermediates and allowing for more accurate forecasting and inventory management.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by its operational simplicity. The absence of exothermic metal-catalyzed steps reduces safety risks associated with thermal runaways, making it safer to operate in large reactors. From an environmental standpoint, the process generates less hazardous waste, as there are no heavy metal residues to dispose of as special waste. The solvent system is compatible with standard recovery and recycling protocols, supporting sustainability goals. This alignment with green chemistry principles not only reduces disposal costs but also enhances the corporate social responsibility profile of the manufacturing entity, which is increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. They are derived from the specific experimental conditions and beneficial effects outlined in the patent documentation, providing clarity for potential partners and technical stakeholders. Understanding these details is vital for assessing the feasibility of integrating this technology into existing production workflows.
Q: What are the primary advantages of this metal-free synthesis method?
A: The primary advantage is the elimination of transition metal catalysts, which removes the need for expensive metal scavenging steps and ensures the final product meets stringent heavy metal limits required for pharmaceutical applications.
Q: What is the typical yield range for this reaction?
A: According to the patent data, the reaction demonstrates excellent efficiency with isolated yields ranging from 70% to 98%, depending on the specific substrate structure and electronic properties.
Q: Is this process scalable for industrial production?
A: Yes, the process utilizes readily available reagents like elemental sulfur and TMSCN in common solvents like DMSO, and involves a simple workup procedure, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Thiocyanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthetic route for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of alpha-thiocyanone meets the highest international standards. We understand that consistency and quality are paramount in the pharmaceutical supply chain, and our commitment to process excellence guarantees reliable delivery of complex intermediates.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By partnering with us, you gain access to our deep expertise in process optimization and regulatory compliance. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your drug development timeline with superior chemical solutions.
