Advanced Synthesis of 2-Methoxy-4-Methylbenzylamine for Commercial Scale-Up and Cost Reduction
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic pathways that balance high purity with operational safety and economic viability. A pivotal advancement in this domain is detailed in Chinese Patent CN102311352B, which discloses a novel, three-step synthesis method for 2-methoxy-4-methylbenzylamine, a critical intermediate widely utilized in the formulation of active pharmaceutical ingredients and specialty fragrances. Unlike traditional routes that rely on hazardous reagents and expensive precursors, this patented methodology leverages a Lewis acid-catalyzed carbonylation followed by a safe reductive amination strategy. For R&D directors and procurement specialists evaluating supply chain resilience, this technology represents a significant shift towards greener, more cost-effective manufacturing protocols that do not compromise on the stringent quality standards required for global regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-methoxy-4-methylbenzylamine has been hindered by reliance on inefficient and dangerous chemical transformations, as exemplified by prior art such as WO2007/124152. The conventional pathway typically initiates with 2-hydroxy-4-methylbenzamide, a specialized starting material that is not readily available as a bulk commodity, leading to inflated raw material costs and potential supply bottlenecks. Furthermore, the critical reduction step in these legacy processes necessitates the use of lithium aluminum hydride (LiAlH4), a highly reactive and pyrophoric reducing agent that demands rigorous anhydrous conditions and specialized handling infrastructure. The inherent risks associated with LiAlH4 not only escalate safety protocols and insurance premiums but also complicate waste disposal due to the generation of aluminum salts, creating a substantial environmental burden for large-scale facilities.
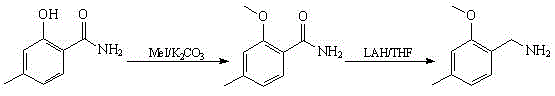
The Novel Approach
In stark contrast, the innovative process outlined in CN102311352B fundamentally reengineers the synthetic logic by utilizing m-cresol and polyoxymethylene as the foundational building blocks, both of which are abundant industrial chemicals with stable pricing and reliable availability. This new route bypasses the need for hazardous metal hydrides entirely by employing a reductive amination technique using ammonium formate, which operates under much milder thermal conditions and generates benign byproducts like carbon dioxide and water. By shifting from an amide reduction strategy to an aldehyde-based reductive amination, the process achieves a total yield of approximately 61% while drastically simplifying the downstream purification workup, thereby offering a compelling value proposition for manufacturers aiming to optimize their cost of goods sold without sacrificing product integrity.
Mechanistic Insights into Lewis Acid-Catalyzed Carbonylation and Reductive Amination
The core of this synthetic breakthrough lies in the precise orchestration of electrophilic aromatic substitution and subsequent functional group interconversions. The initial step involves the carbonylation of m-cresol with polyoxymethylene, catalyzed by a Lewis acid such as tin tetrachloride (SnCl4) in the presence of an organic base like tributylamine. Mechanistically, the Lewis acid activates the formaldehyde source, facilitating the formation of a hydroxymethyl intermediate which subsequently undergoes dehydration and rearrangement to yield 2-hydroxy-4-methyl-benzaldehyde with high regioselectivity. The choice of SnCl4 is critical, as its strong coordination ability ensures efficient activation of the carbonyl species while the bulky organic base neutralizes generated protons, preventing polymerization side reactions and ensuring a clean conversion profile that minimizes the formation of difficult-to-remove impurities.
Following the methylation of the phenolic hydroxyl group using dimethyl sulfate, the final transformation employs a Leuckart-Wallach type reductive amination mechanism. In this step, 2-methoxy-4-methyl-benzaldehyde reacts with ammonium formate at elevated temperatures (approximately 120°C) to form an imine intermediate in situ, which is immediately reduced by the formate ion acting as a hydride donor. This intramolecular redox process is exceptionally elegant because it avoids the introduction of external hydrogen gas or reactive metal hydrides, thereby eliminating the risk of over-reduction or explosive hazards. The result is a highly selective formation of the primary amine functionality, with the patent data indicating a GC purity exceeding 98% after simple distillation, demonstrating the robustness of this mechanism for producing high-specification intermediates.
How to Synthesize 2-Methoxy-4-Methylbenzylamine Efficiently
Implementing this synthesis requires strict adherence to the optimized reaction parameters defined in the patent to ensure maximum yield and safety. The process is divided into three distinct operational stages: the Lewis acid-catalyzed formylation, the O-methylation, and the final reductive amination, each requiring specific solvent systems and thermal controls to drive the equilibrium towards the desired product. Operators must pay particular attention to the stoichiometric ratios of the Lewis acid catalyst and the base in the first step, as well as the temperature ramping profile during the ammonium formate reduction, to prevent thermal runaways. The following guide outlines the standardized procedural framework derived directly from the experimental examples provided in the intellectual property documentation.
- Perform carbonylation of m-cresol with polyoxymethylene using SnCl4 catalyst in toluene at 100°C to yield 2-hydroxy-4-methyl-benzaldehyde.
- Execute methylation of the aldehyde intermediate using dimethyl sulfate in acetone solvent under reflux conditions.
- Conduct reductive amination with ammonium formate at 120°C to safely generate the final amine product without hazardous hydride reagents.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers transformative benefits that extend far beyond simple chemical yield improvements. By transitioning away from specialized amide precursors to commodity chemicals like m-cresol, organizations can insulate themselves from the volatility of niche chemical markets and secure a more predictable cost structure for long-term production planning. Furthermore, the elimination of hazardous reagents like lithium aluminum hydride significantly lowers the barrier to entry for contract manufacturing organizations, as it removes the need for expensive explosion-proof facilities and specialized waste treatment protocols, thereby unlocking a broader network of potential suppliers and reducing overall lead times for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the substitution of high-cost starting materials with low-cost bulk commodities, which creates a substantial margin improvement at the raw material level. Additionally, the avoidance of expensive metal hydride reagents and the associated quenching agents reduces the consumption of auxiliary chemicals, while the simplified workup procedures minimize solvent usage and energy consumption during distillation. These cumulative efficiencies translate into a significantly lower cost of production per kilogram, allowing for more competitive pricing strategies in the global marketplace without eroding profit margins.
- Enhanced Supply Chain Reliability: Reliance on m-cresol and polyoxymethylene ensures a stable supply chain, as these materials are produced on a massive scale for diverse industrial applications, mitigating the risk of shortages that often plague specialized fine chemical intermediates. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent batch-to-batch reproducibility which is critical for maintaining uninterrupted production schedules. This reliability allows supply chain heads to reduce safety stock levels and optimize inventory turnover, knowing that the synthesis is resilient to common logistical disruptions.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is exceptionally well-suited for commercial scale-up of complex pharmaceutical intermediates due to its mild operating conditions and benign waste profile. The absence of heavy metal waste streams and the generation of mostly organic and aqueous waste simplify effluent treatment, ensuring compliance with increasingly stringent environmental regulations across different jurisdictions. The process can be seamlessly scaled from pilot plant quantities to multi-ton annual production capacities using standard stainless steel reactors, facilitating rapid technology transfer and minimizing capital expenditure requirements for facility upgrades.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived from a detailed analysis of the patent's experimental data and comparative advantages. These insights are intended to assist technical teams in evaluating the feasibility of adopting this route for their specific manufacturing needs, covering aspects of safety, purity, and operational complexity. Understanding these nuances is essential for making informed decisions about process validation and supplier qualification.
Q: Why is the ammonium formate reduction method preferred over lithium aluminum hydride?
A: The use of ammonium formate eliminates the severe safety hazards associated with lithium aluminum hydride, such as pyrophoric risks and strict anhydrous requirements, thereby drastically reducing operational costs and insurance liabilities while maintaining high yields.
Q: What are the primary cost drivers reduced in this new synthetic route?
A: This route utilizes m-cresol and polyoxymethylene, which are bulk commodity chemicals, replacing expensive specialized amide starting materials, resulting in substantial raw material cost savings and improved supply chain stability.
Q: Is this process suitable for multi-ton commercial production?
A: Yes, the process operates at moderate temperatures (60-120°C) using common industrial solvents like toluene and acetone, making it highly scalable with minimal requirement for specialized cryogenic or high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methoxy-4-Methylbenzylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a superior synthetic route requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN102311352B are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal requirements of this Lewis acid-catalyzed process, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for pharmaceutical and fragrance applications.
We invite forward-thinking organizations to collaborate with us to leverage this advanced technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this safer, more efficient route for your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make a data-driven decision that enhances both your product quality and your bottom line.
