Scalable Synthesis of 2-Methoxy-4-Methylbenzylamine for Commercial Pharmaceutical Applications
The global demand for functionalized benzylamine derivatives continues to surge, driven by their critical role as building blocks in the synthesis of complex active pharmaceutical ingredients (APIs) and high-value flavor compounds. A pivotal advancement in this domain is detailed in patent CN102311352A, which discloses a robust, three-step synthetic methodology for producing 2-methoxy-4-methylbenzylamine. This technical breakthrough addresses longstanding inefficiencies in traditional amination routes by leveraging abundant commodity chemicals such as m-cresol and polyoxymethylene. By shifting away from hazardous reducing agents and expensive specialized precursors, this process offers a compelling value proposition for manufacturers seeking to optimize their supply chains. The methodology not only ensures high chemical fidelity but also aligns with modern green chemistry principles by minimizing toxic waste generation. For R&D teams and procurement strategists alike, understanding the nuances of this pathway is essential for securing a competitive edge in the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted benzylamines has often relied on pathways that introduce significant operational hazards and cost burdens. As illustrated in the prior art referenced within the patent background, traditional routes frequently employ lithium aluminum hydride (LiAlH4) for the reduction of amide or nitrile intermediates. This reagent is notoriously pyrophoric and requires strictly anhydrous conditions, necessitating expensive infrastructure and rigorous safety protocols that inflate operational expenditures. Furthermore, the quenching of excess LiAlH4 generates substantial amounts of aluminum sludge, creating a heavy burden on waste treatment facilities and complicating environmental compliance. Additionally, many conventional starting materials are not bulk commodities but rather specialized fine chemicals, leading to volatile pricing and potential supply chain disruptions. These factors collectively render traditional methods less attractive for large-scale commercial manufacturing where margin compression and safety are paramount concerns.
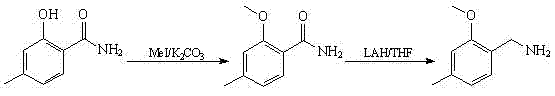
The Novel Approach
In stark contrast, the novel approach outlined in CN102311352A fundamentally reengineers the synthetic logic to prioritize safety and economic efficiency. Instead of relying on hazardous metal hydrides, the process utilizes ammonium formate as a hydrogen donor in a catalytic transfer hydrogenation or reductive amination context. This substitution eliminates the risk of fire and explosion associated with LiAlH4, allowing reactions to proceed under much milder and more manageable thermal conditions. The route begins with the carbonylation of m-cresol, a widely available industrial feedstock, ensuring that the raw material base is both secure and cost-effective. By integrating a methylation step followed by a safe reductive amination, the process achieves a streamlined workflow that reduces the number of purification stages required. This holistic redesign not only enhances operator safety but also significantly lowers the barrier to entry for scaling production to multi-ton quantities.
Mechanistic Insights into Lewis Acid Catalyzed Carbonylation and Reductive Amination
The core of this synthetic strategy lies in the precise orchestration of electrophilic aromatic substitution and subsequent functional group transformations. The initial step involves the reaction of m-cresol with polyoxymethylene in the presence of a Lewis acid catalyst, such as tin tetrachloride (SnCl4), and an organic base like tri-n-butylamine. Mechanistically, the Lewis acid activates the formaldehyde source, facilitating an electrophilic attack on the electron-rich aromatic ring of the m-cresol at the ortho position relative to the hydroxyl group. This regioselectivity is crucial, as it directs the formation of the desired 2-hydroxy-4-methyl-benzaldehyde isomer while minimizing the formation of para-substituted byproducts. The presence of the base serves to neutralize the acidic byproducts generated during the reaction, maintaining the catalytic cycle and preventing the degradation of sensitive intermediates. This careful balance of acidity and nucleophilicity ensures high conversion rates and sets the stage for the subsequent methylation step.
Following the formation of the aldehyde, the process employs a reductive amination mechanism that is distinct from direct reduction. In the final step, 2-methoxy-4-methyl-benzaldehyde reacts with ammonium formate under heating. The formate ion acts as a source of hydride equivalents, transferring hydrogen to the imine or iminium intermediate formed in situ between the aldehyde and ammonia. This transfer hydrogenation mechanism avoids the generation of gaseous hydrogen, which poses explosion risks in large reactors, and instead produces carbon dioxide and ammonia as benign byproducts. The selectivity of this reaction is high, effectively suppressing the formation of secondary or tertiary amines which are common impurities in reductive aminations. By controlling the stoichiometry and temperature, typically around 120°C, the process maximizes the yield of the primary amine while maintaining a clean impurity profile that simplifies downstream purification.
How to Synthesize 2-Methoxy-4-Methylbenzylamine Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and high yield. The process is divided into three distinct operational phases: the initial carbonylation to establish the aldehyde framework, the etherification to install the methoxy group, and the final amination to generate the target amine. Each step utilizes common solvents like toluene and acetone, which are easily recovered and recycled, further enhancing the economic viability of the route. Operators must pay close attention to the molar ratios of the Lewis acid catalyst and the base in the first step, as deviations can lead to incomplete conversion or polymerization of the formaldehyde source. The detailed standardized synthesis steps see the guide below.
- Carbonylation of m-cresol with polyoxymethylene using a Lewis acid catalyst to form 2-hydroxy-4-methyl-benzaldehyde.
- Methylation of the hydroxy-aldehyde intermediate using dimethyl sulfate in acetone to yield 2-methoxy-4-methyl-benzaldehyde.
- Reductive amination of the methoxy-aldehyde with ammonium formate under heating to produce the final amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible strategic benefits that extend beyond simple unit cost savings. The reliance on m-cresol and polyoxymethylene decouples production from the volatility of specialized precursor markets, ensuring a stable and predictable supply of raw materials even during global shortages. Furthermore, the elimination of hazardous reagents like lithium aluminum hydride drastically reduces the costs associated with safety compliance, specialized storage, and hazardous waste disposal. These operational efficiencies translate directly into improved margins and a more resilient supply chain capable of withstanding regulatory scrutiny. The simplified workup procedures also mean faster batch turnover times, allowing manufacturers to respond more agilely to market demand fluctuations without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The substitution of expensive and dangerous reducing agents with ammonium formate represents a significant decrease in direct material costs. Additionally, the use of bulk commodity chemicals as starting materials avoids the premium pricing often attached to fine chemical intermediates. The simplified purification process, driven by high reaction selectivity, reduces solvent consumption and energy usage during distillation and crystallization steps. These cumulative effects result in a substantially lower cost of goods sold (COGS), providing a competitive pricing advantage in the global marketplace for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: By utilizing raw materials that are produced on a massive industrial scale, the risk of supply disruption is minimized. Unlike specialized reagents that may have single-source suppliers or long lead times, m-cresol and polyoxymethylene are readily available from multiple vendors worldwide. This diversification of the supply base ensures continuity of operations and protects against geopolitical or logistical bottlenecks. Moreover, the stability of the intermediates allows for flexible inventory management, enabling manufacturers to stockpile key precursors without significant degradation concerns.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of pyrophoric materials make this process inherently safer to scale from pilot plant to full commercial production. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, reducing the liability and cost associated with waste treatment. The process generates minimal three wastes, facilitating easier permitting and community acceptance for manufacturing sites. This environmental stewardship not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 2-methoxy-4-methylbenzylamine. These insights are derived directly from the technical specifications and experimental data provided in the patent literature. Understanding these details is crucial for evaluating the feasibility of adopting this route for your specific production needs. The answers reflect the consensus on best practices for handling the reagents and optimizing the reaction parameters described.
Q: Why is ammonium formate preferred over lithium aluminum hydride in this synthesis?
A: Ammonium formate serves as a safer, non-hazardous hydrogen donor for reductive amination, eliminating the severe safety risks and complex quenching procedures associated with pyrophoric lithium aluminum hydride.
Q: What are the primary raw materials for this process?
A: The process utilizes m-cresol and polyoxymethylene, which are bulk industrial commodities, ensuring stable supply chains and lower input costs compared to specialized precursors.
Q: What is the expected purity of the final product?
A: The patented method demonstrates the capability to achieve GC content greater than 98% through optimized reaction conditions and standard workup procedures like extraction and distillation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methoxy-4-Methylbenzylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in delivering high-quality intermediates for the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-methoxy-4-methylbenzylamine meets the exacting standards required for API synthesis. Our infrastructure is designed to handle complex chemistries safely, leveraging the very advantages of the patented process to deliver value to our partners.
We invite you to collaborate with us to explore how this advanced synthesis can benefit your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive efficiency and reliability in your supply chain.
