Industrial Synthesis of Alpha,alpha-Difluoroacetaldehyde Using Advanced Ruthenium Hydrogenation Technology
Introduction to Advanced Fluorinated Aldehyde Synthesis
The industrial manufacturing of fluorinated building blocks represents a critical frontier in modern pharmaceutical chemistry, particularly for the development of metabolically stable drug candidates. Patent CN107400044B introduces a transformative methodology for the production of α,α-difluoroacetaldehyde, a highly reactive and valuable intermediate often utilized in the synthesis of complex fluorinated heterocycles and bioactive molecules. Historically, the accessibility of this compound has been hindered by the reliance on stoichiometric hydride reductions, which pose significant safety and environmental challenges. This patent disclosure outlines a sophisticated catalytic hydrogenation protocol that leverages specific ruthenium pincer complexes to achieve high chemoselectivity under mild conditions. By shifting from traditional stoichiometric reagents to a catalytic cycle driven by molecular hydrogen, the technology offers a pathway to significantly enhance the sustainability and economic viability of producing high-purity pharmaceutical intermediates. The innovation lies not only in the catalyst selection but also in the precise control of reaction parameters such as solvent composition and temperature, which are critical for suppressing over-reduction to the corresponding alcohol.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for accessing α,α-difluoroacetaldehyde have predominantly relied on the partial reduction of α,α-difluoroacetates using powerful hydride reducing agents such as lithium aluminum hydride (LiAlH4). While chemically effective on a small laboratory scale, this approach is fundamentally flawed for industrial application due to several intrinsic limitations. Firstly, the use of stoichiometric amounts of pyrophoric hydrides necessitates rigorous safety protocols, specialized handling equipment, and generates substantial quantities of aluminum-containing waste streams that require costly disposal procedures. Secondly, achieving the desired partial reduction to the aldehyde stage without progressing to the fully reduced β,β-difluoroethanol requires extremely low reaction temperatures, often cited around -78°C. Maintaining such cryogenic conditions on a manufacturing scale imposes a massive energy burden and limits the throughput of production facilities. Furthermore, the workup procedures associated with hydride reductions are notoriously complex, involving careful quenching steps that can lead to product degradation or loss of yield, thereby compromising the overall efficiency of the supply chain for these critical fine chemical intermediates.
The Novel Approach
In stark contrast to the hazardous and energy-intensive legacy methods, the novel approach detailed in the patent utilizes a homogeneous ruthenium catalyst to facilitate the hydrogenation of α,α-difluoroacetates using molecular hydrogen. This catalytic strategy operates effectively at much milder temperatures, typically ranging from -30°C to +30°C depending on the solvent system, which drastically reduces the thermal load on the manufacturing infrastructure. The core of this innovation is the ability to tune the selectivity of the reduction, favoring the formation of the aldehyde (often isolated as a stable hemiacetal) over the over-reduced alcohol byproduct. By employing specific reaction solvents, such as mixtures of alcohols and ethers, the process kinetically favors the desired transformation while thermodynamically stabilizing the product. This shift enables a cleaner reaction profile where the primary byproduct is simply the over-reduced alcohol, which can be separated via distillation due to boiling point differences, or the unreacted ester which can be recovered and recycled, thus maximizing atom economy and minimizing waste generation in the production of high-purity OLED material or pharmaceutical precursors.
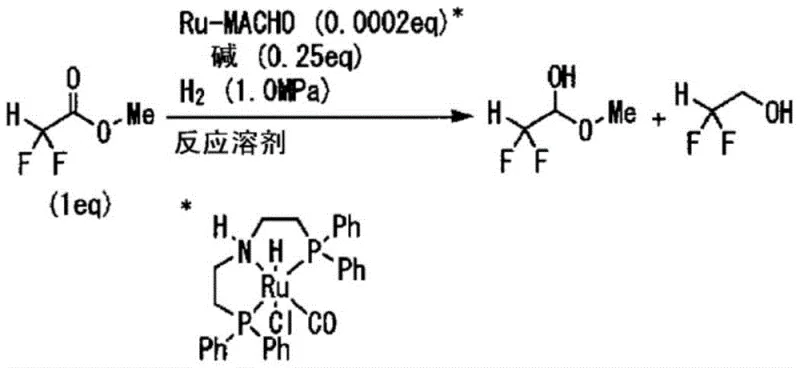
Mechanistic Insights into Ru-MACHO Catalyzed Hydrogenation
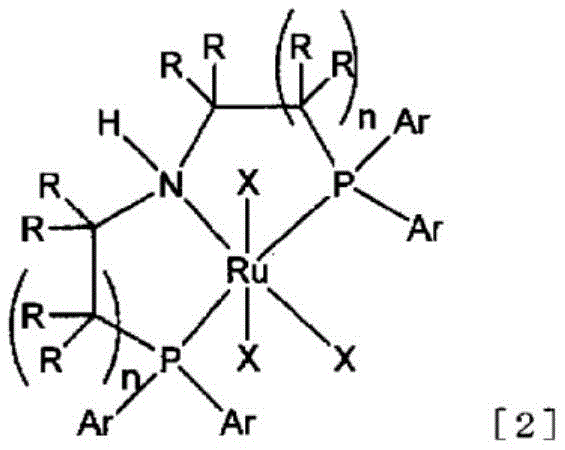
The efficacy of this synthetic route is anchored in the use of specialized ruthenium complexes, specifically those falling under the general Formula [2] and most preferably the Ru-MACHO™ type complex represented by Formula [4]. These complexes function as homogeneous catalysts featuring a PNP pincer ligand architecture that facilitates cooperative metal-ligand activation of hydrogen. In this mechanistic paradigm, the ruthenium center works in tandem with the amine moiety of the ligand to heterolytically cleave the H-H bond, generating a reactive ruthenium hydride species capable of transferring hydride to the carbonyl carbon of the ester substrate. The presence of the electron-withdrawing difluoromethyl group adjacent to the carbonyl significantly alters the electronic landscape of the substrate, making it more susceptible to nucleophilic attack but also prone to over-reduction. The steric and electronic properties of the phosphine arms in the Ru-MACHO catalyst are finely balanced to moderate this reactivity, ensuring that the reduction halts at the aldehyde oxidation state rather than proceeding to the alcohol. This level of control is essential for maintaining the integrity of the fluorinated motif, which is often the key pharmacophore in the final drug substance.
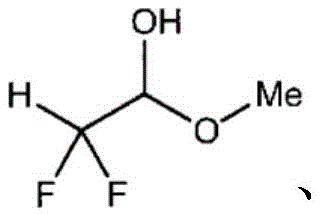
Furthermore, the mechanism accounts for the formation of stable hemiacetal derivatives rather than the free aldehyde, which is inherently unstable and prone to polymerization. In the presence of the alcohol solvent (e.g., methanol or ethanol) and the basic conditions required to activate the catalyst, the nascent α,α-difluoroacetaldehyde immediately undergoes nucleophilic addition with the solvent alcohol to form the corresponding hemiacetal (Formula [8]). This in-situ derivatization serves a dual purpose: it protects the reactive aldehyde functionality from decomposition during the reaction and workup, and it modifies the physical properties of the molecule to facilitate purification. The patent highlights that the ratio of methanol to ethanol in the solvent system can influence the distribution of methyl versus ethyl hemiacetals, providing an additional handle for process optimization. Understanding this equilibrium is vital for R&D teams aiming to replicate the high selectivity ratios (aldehyde:alcohol > 90:10) reported in the examples, as deviations in solvent purity or water content could shift the equilibrium towards unwanted byproducts or hydrate formation.
How to Synthesize Alpha,alpha-Difluoroacetaldehyde Efficiently
Implementing this catalytic hydrogenation process requires careful attention to the preparation of the reaction mixture and the control of gaseous reagents. The procedure generally involves charging a pressure-resistant vessel with the α,α-difluoroacetate substrate, the ruthenium catalyst precursor, and a suitable base such as sodium methoxide or potassium tert-butoxide. The choice of base is critical as it activates the catalyst precursor into the true active species, likely a ruthenium hydride or amido complex. Following the addition of the solvent system, which may be a single alcohol or a mixture with an ether like tert-butyl methyl ether to modulate selectivity, the reactor is pressurized with hydrogen. The reaction is then allowed to proceed with stirring at controlled temperatures, typically below 30°C for alcoholic solvents to prevent thermal degradation and maintain selectivity. Detailed standardized synthesis steps see the guide below.
- Charge a pressure-resistant reactor with alpha,alpha-difluoroacetate substrate, Ru-MACHO catalyst (0.00002-0.002 eq), and alkali metal alkoxide base.
- Add alcohol-based solvent (e.g., methanol or ethanol) and pressurize the system with hydrogen gas to 0.01-2.0 MPa.
- Maintain reaction temperature below 30°C for alcohol solvents, monitoring conversion until selectivity favors the aldehyde hemiacetal over the over-reduced alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from stoichiometric hydride reduction to catalytic hydrogenation represents a strategic opportunity to optimize the cost structure and reliability of the supply chain for fluorinated intermediates. The elimination of expensive and hazardous hydride reagents directly translates to a reduction in raw material procurement costs and a simplification of the logistics associated with handling dangerous goods. Moreover, the ability to recover and recycle unreacted starting materials, as demonstrated in the large-scale examples where recovery rates exceeded 90%, significantly improves the overall material utilization efficiency. This closed-loop capability reduces the volume of fresh feedstock required per kilogram of finished product, thereby insulating the manufacturing process from volatility in the pricing of fluorinated starting materials. The simplified workup, which primarily relies on distillation rather than complex aqueous quenches and extractions, also reduces the consumption of auxiliary solvents and utilities, contributing to a leaner and more cost-effective production model.
From a supply chain continuity perspective, the robustness of this catalytic method enhances the reliability of delivery schedules. The reaction conditions are mild enough to be performed in standard stainless steel hydrogenation reactors without the need for specialized cryogenic infrastructure, making the technology transferable across multiple manufacturing sites globally. The high turnover number of the ruthenium catalyst, evidenced by the low catalyst loading of 0.0002 equivalents relative to the substrate, ensures that the supply of the precious metal catalyst does not become a bottleneck for production scaling. Additionally, the process generates significantly less hazardous waste compared to traditional methods, aligning with increasingly stringent environmental regulations and reducing the regulatory burden associated with waste disposal permits. This environmental compliance advantage mitigates the risk of production shutdowns due to regulatory non-compliance, ensuring a steady flow of high-purity pharmaceutical intermediates to downstream customers who depend on consistent quality and availability for their own drug development timelines.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this ruthenium-catalyzed hydrogenation technology. These answers are derived directly from the experimental data and specifications provided in the patent literature to ensure accuracy and relevance for technical decision-makers. Understanding these nuances is crucial for evaluating the feasibility of adopting this method for commercial manufacturing campaigns.
Q: Why is ruthenium catalysis preferred over hydride reduction for this synthesis?
A: Ruthenium catalysis utilizes molecular hydrogen instead of stoichiometric hydride reagents like LiAlH4, eliminating hazardous waste, reducing raw material costs, and allowing for milder reaction temperatures above -78°C.
Q: How is the unstable alpha,alpha-difluoroacetaldehyde isolated?
A: The aldehyde is typically isolated as a stable hemiacetal derivative (e.g., methyl or ethyl hemiacetal) formed in situ with the alcohol solvent, which prevents polymerization and facilitates purification via distillation.
Q: What is the scalability potential of this hydrogenation process?
A: The process has been demonstrated at scales up to 40kg of substrate with high recovery rates of unreacted starting material, indicating robust potential for multi-ton commercial manufacturing without requiring extreme high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha,alpha-Difluoroacetaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the advancement of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of α,α-difluoroacetaldehyde or its stable hemiacetal derivatives meets the exacting standards required for GMP pharmaceutical synthesis. Our commitment to process excellence allows us to offer a reliable alpha,alpha-difluoroacetaldehyde supplier partnership that prioritizes both product integrity and supply security.
We invite you to engage with our technical procurement team to discuss how this advanced catalytic technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this hydrogenation route for your specific project needs. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring that your development programs proceed without interruption due to material shortages or quality inconsistencies.
