Advanced Synthesis of Nicotinamide Riboside Chloride for Commercial Scale-Up
The pharmaceutical and nutraceutical industries are witnessing a surge in demand for NAD+ precursors, specifically nicotinamide riboside chloride (NR Chloride), due to its potent anti-aging properties and ability to boost cellular energy metabolism. Patent CN114369129A introduces a groundbreaking synthetic methodology that addresses critical bottlenecks in the current manufacturing landscape, offering a pathway to higher purity and significantly lower production costs. This technical insight report analyzes the novel approach which utilizes tetraacetyl ribose as a starting material, activated by a stoichiometric amount of trimethylsilyl trifluoromethanesulfonate, followed by a sophisticated ion-exchange protocol. By replacing hazardous bromination steps and excessive reagent usage with a streamlined deacetylation and tartrate exchange process, this invention represents a paradigm shift for reliable nicotinamide riboside chloride suppliers aiming to scale production. The following analysis details the mechanistic advantages, commercial viability, and operational robustness of this new route for global procurement teams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nicotinamide riboside chloride has been plagued by significant technical hurdles that compromise both yield and product safety, creating substantial risks for supply chain continuity. As illustrated in the comparative reaction schemes, Method A relies on unstable ribose chloride intermediates which decompose easily, resulting in a dismal beta-isomer yield of approximately 45 percent and generating corrosive hydrogen chloride gas that damages reactor equipment. Method B attempts to improve yield through bromination but introduces severe toxicity concerns due to the retention of harmful bromide ions within the molecular structure, which are notoriously difficult to purge completely during downstream processing. Furthermore, Method C, while improving stereoselectivity, is economically unfeasible for large-scale operations because it demands a seven-fold excess of expensive trimethylsilyl trifluoromethanesulfonate relative to nicotinamide. These legacy processes not only inflate the cost of goods sold but also leave behind toxic residues such as trifluoromethanesulfonate ions that fail to meet stringent pharmaceutical quality standards for human consumption.
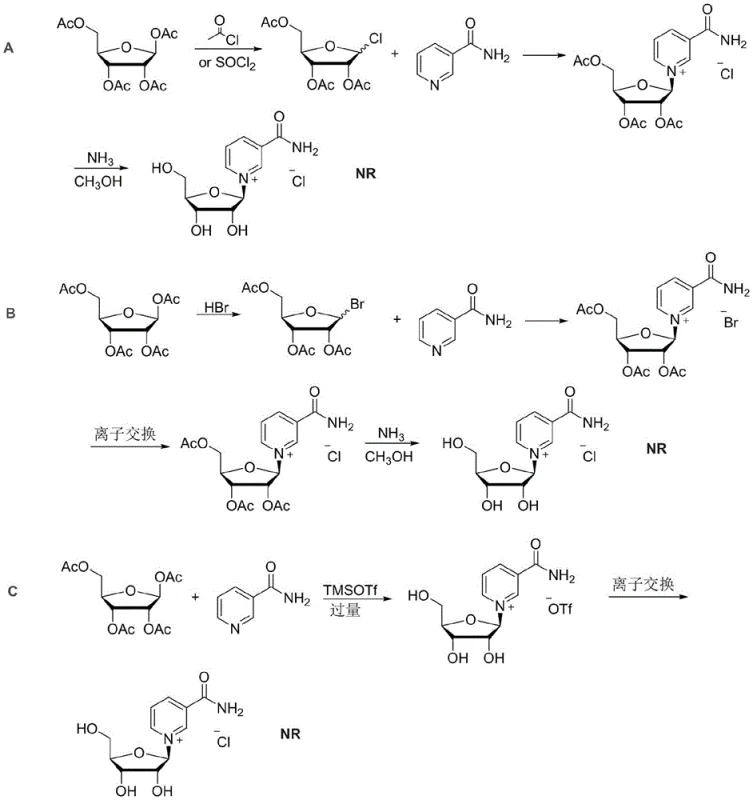
The Novel Approach
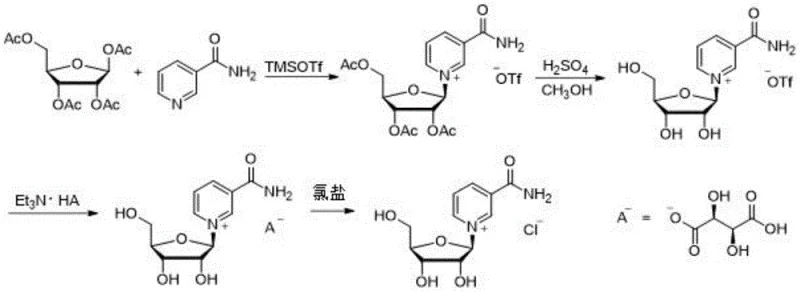
The innovative strategy disclosed in the patent overcomes these deficiencies by employing a precise 1:1 molar ratio of trimethylsilyl trifluoromethanesulfonate to activate tetraacetyl ribose, thereby eliminating the waste associated with previous excess reagent protocols. This activation step is followed by a mild deacetylation using a methanol sulfuric acid solution, which preserves the integrity of the glycosidic bond while efficiently removing protecting groups. Crucially, the process incorporates a unique ion-exchange mechanism where trifluoromethanesulfonate ions are first swapped for tartrate ions using triethylamine tartrate, and subsequently exchanged for chloride ions using soluble salts like potassium chloride. This multi-step exchange ensures that the final product is free from toxic organic acid residues and bromide contaminants, delivering a high-purity API intermediate suitable for sensitive nutraceutical applications. The visual representation of this optimized pathway highlights the streamlined nature of the reaction sequence compared to the convoluted legacy methods.
Mechanistic Insights into TMSOTf-Catalyzed Glycosylation and Ion Exchange
The core of this synthetic breakthrough lies in the controlled activation of the anomeric center of tetraacetyl ribose using trimethylsilyl trifluoromethanesulfonate (TMSOTf) as a Lewis acid catalyst. In this mechanism, the silicon atom of TMSOTf coordinates with the acetoxy group at the C1 position of the ribose ring, facilitating the formation of an oxocarbenium ion intermediate that is highly reactive towards the nucleophilic attack by the pyridine nitrogen of nicotinamide. This specific activation mode favors the formation of the beta-anomer due to the neighboring group participation of the C2-acetoxy group, which directs the incoming nucleophile to the opposite face, ensuring high stereoselectivity without the need for cryogenic temperatures or exotic catalysts. The use of acetonitrile as the solvent further stabilizes the intermediate species, allowing the reaction to proceed to completion with near-quantitative conversion, as evidenced by the 100 percent yield of the acetylated intermediate reported in the experimental examples. This level of control is essential for minimizing the formation of alpha-isomer impurities that would otherwise require costly and yield-reducing recrystallization steps later in the process.
Following the glycosylation, the deprotection and ion exchange phases are engineered to maximize purity through selective precipitation rather than chromatographic separation. The addition of sulfuric acid in methanol cleaves the acetate esters to reveal the free hydroxyl groups of the ribose moiety, generating the nicotinamide riboside trifluoromethanesulfonate salt in situ. The subsequent addition of triethylamine and L-hydrogen tartrate triggers a metathesis reaction where the bulky and potentially toxic trifluoromethanesulfonate anion is replaced by the hydrogen tartrate anion, which forms a less soluble salt that can be partially isolated. Finally, the reaction with potassium chloride or calcium chloride in cold aqueous solution exploits the low solubility of potassium hydrogen tartrate or calcium tartrate to drive the equilibrium towards the formation of the desired chloride salt. This clever use of solubility differences allows for the removal of tartrate byproducts via simple filtration, leaving the nicotinamide riboside chloride in the filtrate with exceptional purity levels exceeding 99 percent.
How to Synthesize Nicotinamide Riboside Chloride Efficiently
Implementing this synthesis route requires strict adherence to moisture-free conditions during the initial activation phase to prevent the hydrolysis of the sensitive silylating agent and the ribose intermediate. The process begins with the dissolution of nicotinamide and tetraacetyl ribose in anhydrous acetonitrile under an inert nitrogen atmosphere, followed by the dropwise addition of the activator to manage the exotherm and ensure uniform reaction kinetics. Once the acetylated intermediate is secured, the deacetylation is performed at room temperature using a standardized sulfuric acid-methanol mixture, monitored closely by HPLC to prevent over-reaction or degradation of the glycosidic linkage. The detailed standardized synthesis steps见下方的指南 outline the precise stoichiometry and workup procedures required to achieve the reported high yields and purity specifications consistently.
- Activate tetraacetyl ribose with trimethylsilyl trifluoromethanesulfonate (1: 1 molar ratio) in acetonitrile under nitrogen protection to form the trifluoromethanesulfonate intermediate.
- Perform deacetylation using a methanol sulfuric acid solution, followed by ion exchange with triethylamine tartrate to precipitate the hydrogen tartrate salt.
- Dissolve the tartrate salt in water and react with soluble chloride (potassium or calcium chloride) at 0°C to exchange the tartrate ion for chloride, yielding the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible benefits that extend beyond mere technical feasibility, directly impacting the bottom line and operational reliability. By eliminating the need for excessive amounts of expensive activating reagents and avoiding the use of hazardous brominating agents, the overall cost structure of the manufacturing process is significantly optimized without compromising on quality metrics. The reliance on simple precipitation and filtration for purification, rather than resource-intensive chromatography, drastically reduces solvent consumption and processing time, leading to substantial cost savings in utility and waste disposal. Furthermore, the use of readily available commodity chemicals such as potassium chloride and tartaric acid ensures that the supply chain remains resilient against fluctuations in the availability of specialized reagents.
- Cost Reduction in Manufacturing: The most significant economic advantage stems from the stoichiometric efficiency of the new method, which utilizes a 1:1 molar ratio of trimethylsilyl trifluoromethanesulfonate compared to the seven-fold excess required in previous iterations. This drastic reduction in reagent consumption directly lowers the raw material costs, while the elimination of toxic bromide handling reduces the capital expenditure required for corrosion-resistant equipment and specialized waste treatment facilities. Additionally, the high yield of the beta-isomer minimizes the loss of valuable starting materials, ensuring that the cost reduction in pharmaceutical intermediates manufacturing is realized through both input savings and output maximization.
- Enhanced Supply Chain Reliability: The robustness of this synthetic pathway enhances supply security by reducing dependency on hard-to-source or highly regulated reagents like thionyl chloride or large quantities of hydrobromic acid. The process operates under relatively mild conditions and utilizes common solvents like acetonitrile and methanol, which are widely available in the global chemical market, thereby mitigating the risk of production stoppages due to raw material shortages. Moreover, the simplified workup procedure involving crystallization and filtration is easier to scale and control than complex extraction or chromatographic methods, ensuring consistent batch-to-batch quality and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the avoidance of heavy metal catalysts and toxic halogenated byproducts aligns perfectly with modern green chemistry principles and stringent regulatory standards. The generation of benign byproducts such as potassium hydrogen tartrate, which can be easily filtered and potentially repurposed, simplifies the effluent treatment process and reduces the environmental footprint of the facility. This compliance advantage facilitates smoother regulatory approvals and faster time-to-market for the final nutraceutical products, making the commercial scale-up of complex pharmaceutical intermediates more feasible and sustainable in the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent specifications and experimental data. Understanding these nuances is critical for R&D teams evaluating the feasibility of technology transfer and for quality assurance personnel establishing control strategies. The answers provided reflect the specific advantages of the tartrate exchange mechanism and the safety profile improvements over legacy bromination routes.
Q: How does this synthesis method improve product safety compared to traditional bromination routes?
A: Traditional Method B utilizes hydrobromic acid for bromination, which leaves harmful bromide ion residues that are difficult to completely remove and pose toxicity risks. The novel method described in patent CN114369129A avoids bromination entirely, utilizing a trifluoromethanesulfonate activation followed by a specific tartrate exchange process that ensures the final chloride product is free from toxic bromide contaminants.
Q: What is the stereoselectivity advantage of using trimethylsilyl trifluoromethanesulfonate?
A: The use of trimethylsilyl trifluoromethanesulfonate as an activating reagent provides excellent stereocontrol during the glycosylation step. Unlike Method A which yields only about 45% of the desired beta isomer, this optimized protocol maintains a high conversion rate of tetraacetyl ribose and significantly enhances the yield of the biologically active beta isomer, reducing the burden of separating unwanted alpha isomers.
Q: How is the cost of production reduced in this new synthetic route?
A: Cost reduction is achieved primarily by optimizing reagent stoichiometry and simplifying purification. Previous Method C required a seven-fold excess of expensive trimethylsilyl trifluoromethanesulfonate, whereas this invention utilizes a precise 1:1 molar ratio. Additionally, the use of simple precipitation and filtration for ion exchange eliminates the need for complex and costly chromatographic purification steps often required to remove toxic organic acid residues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nicotinamide Riboside Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the growing global demand for high-quality NAD+ precursors. Our team of expert chemists has thoroughly evaluated the route described in CN114369129A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovation to the market. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to ensure that every batch of nicotinamide riboside chloride meets the highest international standards for safety and efficacy. Our state-of-the-art facilities are equipped to handle the specific moisture-sensitive steps and ion-exchange protocols necessary to replicate the high yields and stereoselectivity demonstrated in the patent literature.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific product pipeline. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this bromide-free process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of this superior intermediate into your supply chain.
