Advanced Near-Infrared Aza-BODIPY Dyes: Technical Breakthroughs and Commercial Scalability
Advanced Near-Infrared Aza-BODIPY Dyes: Technical Breakthroughs and Commercial Scalability
The landscape of optical functional materials is undergoing a significant transformation driven by the demand for deeper tissue penetration and higher stability in imaging applications. Patent CN102286213A introduces a groundbreaking class of near-infrared aza-BODIPY dyes that address the critical limitations of existing fluorophores. Unlike traditional visible-range BODIPY dyes which are restricted to the 400-650 nm range, this innovation successfully extends the maximum absorption wavelength beyond 730 nm and fluorescence emission over 740 nm. This spectral shift is achieved through a novel structural modification that incorporates conjugated ethylenic bonds directly into the aza-BODIPY core, effectively expanding the pi-electron system. For R&D directors and procurement specialists in the electronic chemical and pharmaceutical sectors, this represents a pivotal opportunity to access high-performance materials that combine the brightness of quantum dots with the safety and stability of organic small molecules.
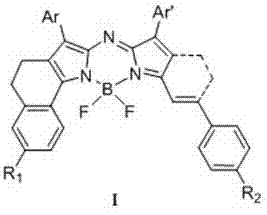
The general formula presented in the patent highlights the versatility of the molecular scaffold, allowing for extensive functionalization to tune solubility and targeting capabilities. The core structure features a boron-dipyrromethene framework where the nitrogen atoms are bridged, creating a rigid planar system that minimizes non-radiative energy loss. This structural rigidity is paramount for achieving the reported fluorescence quantum yields of up to 0.28, a figure that rivals many commercial cyanine dyes while offering superior resistance to photobleaching. For supply chain managers, the ability to source a dye that maintains integrity under prolonged illumination translates directly to reduced waste and more reliable assay results in downstream applications such as cell imaging and photodynamic therapy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of near-infrared fluorescence has been dominated by cyanine dyes and semiconductor quantum dots, each carrying significant drawbacks that hinder their widespread adoption in sensitive biological and electronic applications. Cyanine dyes, while widely available, are notoriously prone to aggregation in aqueous environments, which leads to self-quenching and a drastic reduction in fluorescence efficiency. Furthermore, their polymethine chains are chemically unstable, susceptible to oxidation and photodegradation, resulting in short shelf-lives and inconsistent performance during long-term imaging experiments. On the other hand, quantum dots offer excellent brightness but introduce severe toxicity concerns due to the potential leakage of heavy metal ions like cadmium or lead, necessitating complex and costly encapsulation strategies to ensure biocompatibility. These inherent instability and safety issues create substantial bottlenecks in the manufacturing of reliable optical probes, driving up the cost of quality control and limiting the scope of in vivo applications.
The Novel Approach
The methodology outlined in CN102286213A circumvents these challenges by leveraging the inherent stability of the aza-BODIPY scaffold and enhancing it through strategic conjugation extension. By introducing conjugated double bonds via a vinyl linkage between the pyrrole ring and the peripheral aromatic systems, the inventors have successfully red-shifted the absorption and emission profiles into the near-infrared window without compromising the molecular integrity. This approach eliminates the need for unstable polymethine chains found in cyanines and avoids the use of toxic heavy metals entirely. The synthesis utilizes robust organic reactions such as nitrosation and Lewis acid-mediated complexation, which are highly reproducible and scalable. For manufacturers, this means a transition from fragile, temperamental dyes to robust chemical entities that can withstand the rigors of industrial processing and storage, thereby ensuring a consistent supply of high-purity electronic chemicals and biological probes.
Mechanistic Insights into Conjugation-Extended Aza-BODIPY Formation
The core of this technological advancement lies in the precise construction of the conjugated system prior to the formation of the boron complex. The mechanism begins with the preparation of a pyrrole intermediate bearing a conjugated ethylenic bond, typically synthesized via the reaction of an unsaturated ketone with phenylazirine under strong basic conditions using LDA (Lithium Diisopropylamide). This step is critical as it installs the necessary pi-system extension that will later dictate the optical properties of the final dye. The unsaturated ketone itself is derived from accessible precursors like aromatic aldehydes and methyl ketones through aldol condensation, ensuring that the raw material costs remain low and the supply chain is resilient. The subsequent reaction with phenylazirine requires careful temperature control, typically at -78°C, to manage the reactivity of the lithiated species and ensure regioselective formation of the desired pyrrole isomer.
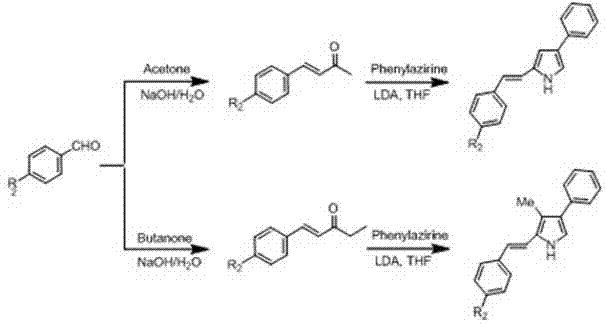
Following the preparation of the conjugated pyrrole, the process moves to the formation of the aza-dipyrromethene backbone. This involves the nitrosation of a second, non-conjugated pyrrole component, such as 4,5-dihydro-7-methoxy-3-phenylbenzo[g]indole, using sodium nitrite in acetic acid. The resulting nitroso-pyrrole acts as an electrophile that condenses with the conjugated pyrrole nucleophile in the presence of acetic anhydride. This condensation step creates the extended dipyrromethene ligand which is then immediately complexed with boron trifluoride etherate in the presence of a base like triethylamine. The coordination of the boron atom locks the conformation of the ligand, rigidifying the structure and preventing rotational relaxation that would otherwise dissipate energy as heat. This mechanistic sequence ensures that the final product possesses the narrow half-peak width and high molar extinction coefficient required for high-resolution imaging and sensing applications.
How to Synthesize Near-Infrared Aza-BODIPY Dyes Efficiently
The synthesis of these advanced dyes follows a logical three-stage progression that balances chemical complexity with operational feasibility. The process initiates with the construction of the functionalized pyrrole building blocks, followed by their assembly into the chromophore, and concludes with the stabilization via boron chelation. Each stage has been optimized in the patent examples to maximize yield and purity, utilizing standard laboratory equipment and reagents that are readily available on the global chemical market. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of reaction conditions and workup procedures.
- Synthesize conjugated pyrrole intermediates by reacting aromatic aldehydes with methyl ketones or aryl lithium with beta-methoxycyclohexenone, followed by reaction with phenylazirine under LDA conditions.
- Perform nitrosation on the non-conjugated pyrrole component (e.g., 4,5-dihydro-7-methoxy-3-phenylbenzo[g]indole) using sodium nitrite in acetic acid.
- Condense the nitrosated pyrrole with the conjugated pyrrole intermediate in acetic anhydride, followed by complexation with boron trifluoride etherate to form the final aza-BODIPY core.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aza-BODIPY technology offers distinct strategic advantages over legacy dye systems. The primary value driver is the significant reduction in risk associated with material stability and regulatory compliance. Unlike quantum dots which face increasing scrutiny regarding heavy metal content, these organic dyes are composed of carbon, hydrogen, nitrogen, oxygen, and boron, simplifying the regulatory approval process for medical and consumer electronic applications. Furthermore, the enhanced photostability means that end-users require less frequent replacement of reagents, fostering stronger customer loyalty and reducing the frequency of urgent replenishment orders that strain logistics networks. The synthetic route relies on commodity chemicals rather than exotic catalysts, insulating the production cost from volatile precious metal markets.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the avoidance of complex encapsulation processes required for quantum dots leads to substantial cost savings in the overall manufacturing budget. The synthesis utilizes straightforward condensation and complexation reactions that do not require specialized high-pressure or high-temperature equipment, allowing for production in standard glass-lined reactors. Additionally, the high purity achievable through simple recrystallization and column chromatography reduces the need for expensive preparative HPLC purification steps, further lowering the cost of goods sold while maintaining high-purity electronic chemical standards.
- Enhanced Supply Chain Reliability: The starting materials, such as aromatic aldehydes, acetone, and pyrrole derivatives, are bulk commodities produced by numerous global suppliers, ensuring a diversified and secure supply base. This reduces the risk of single-source bottlenecks that often plague the supply of specialized fluorophores. The robustness of the chemical intermediates allows for stockpiling without significant degradation, enabling manufacturers to maintain safety stocks and respond rapidly to fluctuations in market demand without compromising product quality or extending lead times for high-purity optical materials.
- Scalability and Environmental Compliance: The reaction conditions are mild and generate waste streams that are easier to treat compared to heavy metal-containing processes. The use of common organic solvents like dichloromethane and ethanol facilitates solvent recovery and recycling, aligning with green chemistry principles and reducing environmental disposal costs. The process is inherently scalable from gram to kilogram quantities without significant re-optimization, supporting the commercial scale-up of complex optical functional materials needed for emerging technologies in bio-imaging and optoelectronics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this near-infrared dye technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and application suitability. Understanding these details is crucial for integrating these materials into existing product lines or developing new diagnostic tools.
Q: What are the key optical properties of the aza-BODIPY dyes described in CN102286213A?
A: The patented dyes exhibit absorption wavelengths exceeding 730 nm and fluorescence emission over 740 nm, placing them firmly in the near-infrared region. They demonstrate a fluorescence quantum yield reaching 0.28, which is comparable to cyanine dyes but with significantly improved photostability and narrower half-peak widths.
Q: How does this synthesis route improve upon traditional cyanine dye manufacturing?
A: Traditional cyanine dyes often suffer from low quantum yields and poor photostability due to aggregation and photobleaching. This novel aza-BODIPY route introduces conjugated ethylenic bonds into the matrix structure, expanding conjugation and reducing non-radiative deactivation, thereby enhancing stability and efficiency without the toxicity concerns associated with quantum dots.
Q: Is this process suitable for large-scale production of optical materials?
A: Yes, the synthesis relies on standard organic transformations such as aldol condensation, nitrosation, and Lewis acid complexation, which are well-established in industrial chemistry. The use of readily available starting materials like aromatic aldehydes and ketones supports scalable manufacturing for high-purity electronic chemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Near-Infrared Aza-BODIPY Dye Supplier
As the demand for advanced optical materials grows, partnering with an experienced CDMO becomes essential for navigating the complexities of scale-up and quality assurance. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our rigorous QC labs and stringent purity specifications guarantee that every batch of aza-BODIPY dye meets the exacting standards required for sensitive applications like cell imaging and photodynamic therapy, minimizing the risk of batch-to-batch variability.
We invite you to collaborate with our technical team to explore how this patented technology can enhance your product portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your R&D and sourcing decisions, ensuring a seamless transition from laboratory discovery to commercial success.
