Scalable Synthesis of Diquafosol Tetrasodium: A Novel Metal-Catalyzed Route for High-Purity Ophthalmic APIs
Scalable Synthesis of Diquafosol Tetrasodium: A Novel Metal-Catalyzed Route for High-Purity Ophthalmic APIs
The pharmaceutical landscape for ophthalmic treatments has seen significant advancements with the introduction of P1,P4-di(uridine-5'-) tetraphosphate, commonly known as Diquafosol Tetrasodium. This compound, marketed under trade names such as Diquas, serves as a potent P2Y2 receptor agonist for the treatment of dry eye disease. The synthesis of this complex nucleotide derivative presents substantial challenges, particularly regarding purity profiles and residual metal content, which are critical for ocular safety. Patent CN111116694B discloses a groundbreaking process that addresses these historical bottlenecks by utilizing a specific metal-catalyzed coupling strategy. This technical insight report analyzes the proprietary methodology, highlighting its potential to redefine supply chain standards for high-value ophthalmic active pharmaceutical ingredients.
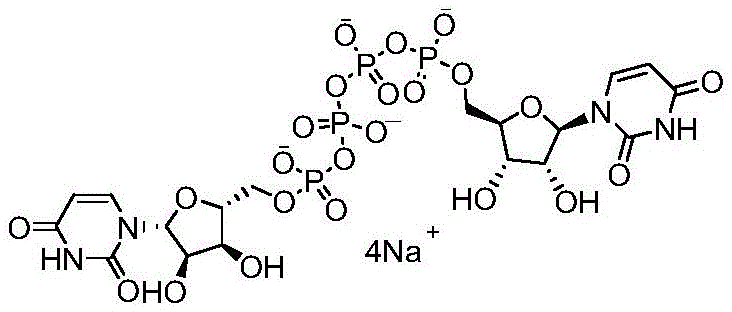
The structural complexity of Diquafosol Tetrasodium necessitates a synthetic route that can precisely construct the tetraphosphate bridge between two uridine moieties without generating excessive polyphosphate byproducts. Traditional methods often struggle with the formation of difficult-to-remove impurities such as P1,P5-bis(uridine-5'-) pentaphosphate and P1,P6-bis(uridine-5'-) hexaphosphate. The disclosed invention offers a robust solution by optimizing the reaction environment through the use of triethylamine salts and specific metal catalysts, ensuring that the final product meets the stringent purity requirements demanded by global pharmacopoeias for ophthalmic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
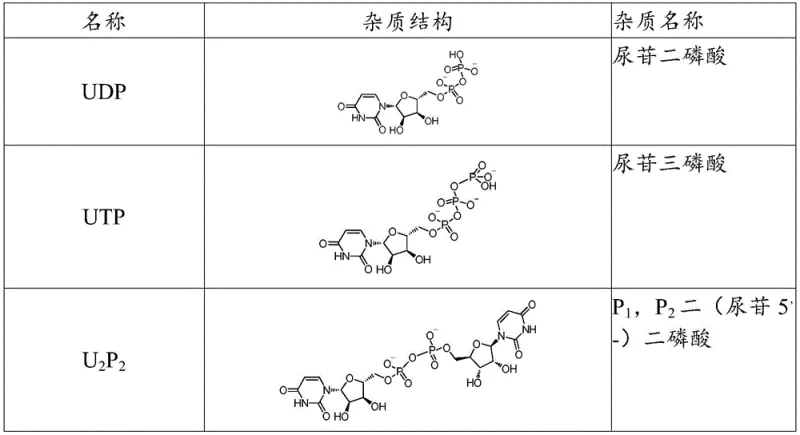
Historical synthetic routes for P1,P4-di(uridine-5'-) tetraphosphate have been plagued by significant environmental and safety concerns, primarily due to the reliance on highly toxic tributylamine reagents. In prior art methodologies, tributylamine salts of uridine diphosphate or uridine monophosphate were condensed using various activators, leading to reaction mixtures that were not only hazardous to handle but also difficult to purify. A major drawback of these conventional approaches is the lack of control over specific impurity profiles; while they might achieve moderate yields, they frequently fail to suppress the formation of higher polyphosphate impurities like U2P5 and U2P6. Furthermore, existing purification techniques, such as standard anion exchange chromatography, often prove insufficient for removing these structurally similar byproducts, resulting in final products that may not meet the rigorous <1% total impurity threshold required for pharmaceutical-grade materials.
The Novel Approach
The innovative process described in the patent data fundamentally shifts the paradigm by replacing toxic tributylamine with triethylamine salts and introducing a controlled metal salt catalysis system. This method involves the reaction of imidazole pyrophosphate triethylamine salt with uridine monophosphate triethylamine salt in N,N-dimethylformamide (DMF). The strategic selection of manganese chloride or zinc chloride as the catalyst, specifically at a molar ratio of 5-10:1 relative to the imidazole pyrophosphate, drives the coupling efficiency to unprecedented levels. This approach not only mitigates the environmental hazards associated with tributylamine but also inherently suppresses the formation of critical impurities at the source, simplifying the downstream purification burden significantly.
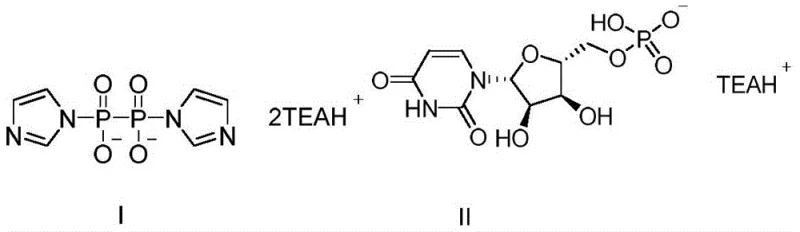
Mechanistic Insights into Metal-Catalyzed Nucleotide Coupling
The efficacy of this synthesis relies heavily on the precise mechanistic interplay between the activated pyrophosphate species and the metal catalyst. The imidazole pyrophosphate triethylamine salt acts as a highly reactive electrophile, activated by the imidazole leaving group. When introduced to the uridine monophosphate triethylamine salt in the presence of anhydrous manganese chloride or zinc chloride, the metal cation likely coordinates with the phosphate oxygens, stabilizing the transition state and facilitating the nucleophilic attack by the phosphate group of the uridine monophosphate. This coordination chemistry is crucial; without the specific metal catalyst, the reaction proceeds sluggishly or yields a complex mixture of products. The optimization of the catalyst loading to a 5-10:1 molar ratio ensures that the activation energy is sufficiently lowered to drive the reaction to completion within a practical timeframe of 2-4 hours at mild temperatures of 25-35°C.
Controlling the impurity profile is perhaps the most critical aspect of this mechanism, particularly regarding the suppression of U2P5 and U2P6. These higher polyphosphates typically arise from over-phosphorylation or side reactions involving excess activating agents. By fine-tuning the stoichiometry of the triethylamine salts and the specific activation of the pyrophosphate intermediate, the process minimizes the availability of reactive species that could lead to chain elongation beyond the desired tetraphosphate structure. Additionally, the subsequent purification protocol is designed to target these specific contaminants. The use of a chlorine-type anion exchange resin, combined with a specific elution gradient, allows for the separation of the target tetraphosphate from the pentaphosphate and hexaphosphate analogues, which possess different charge densities and binding affinities.
How to Synthesize P1,P4-di(uridine-5'-) tetraphosphate Efficiently
The synthesis of this complex ophthalmic intermediate requires strict adherence to the optimized parameters regarding solvent quality, reagent stoichiometry, and temperature control to ensure reproducibility and high yield. The process begins with the preparation of high-purity triethylamine salts of the starting materials, followed by the critical coupling step in DMF. Detailed operational procedures, including specific quenching methods and workup sequences, are essential for achieving the low metal ion residuals required for ocular safety. For a comprehensive, step-by-step technical guide on executing this synthesis, please refer to the standardized protocol outlined below.
- Preparation of imidazole pyrophosphate triethylamine salt and uridine monophosphate triethylamine salt precursors.
- Coupling reaction in DMF using manganese chloride or zinc chloride catalyst at 25-35°C.
- Purification via ethyl acetate precipitation, bicarbonate/carbonate treatment, anion exchange, and chelating resin filtration.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this novel synthetic route offers transformative advantages for procurement managers and supply chain directors seeking reliable sources of high-quality ophthalmic intermediates. The elimination of tributylamine not only aligns with increasingly stringent global environmental regulations but also drastically reduces the costs associated with hazardous waste disposal and worker safety protocols. By switching to triethylamine, manufacturers can streamline their EHS (Environment, Health, and Safety) compliance processes, leading to a more sustainable and cost-effective production lifecycle. Furthermore, the improved yield and purity directly translate to better material throughput, reducing the overall cost of goods sold (COGS) for the final API.
- Cost Reduction in Manufacturing: The substitution of expensive and toxic reagents with more economical and safer alternatives like triethylamine significantly lowers raw material costs. Moreover, the high selectivity of the metal-catalyzed reaction reduces the burden on downstream purification, meaning less solvent and resin consumption per kilogram of product. This efficiency gain allows for substantial cost reduction in API manufacturing, making the final therapeutic more accessible while maintaining healthy margins for suppliers.
- Enhanced Supply Chain Reliability: The robustness of this chemical process ensures consistent batch-to-batch quality, which is paramount for maintaining uninterrupted drug supply. The ability to consistently achieve purities exceeding 99% with metal residues below 10ppm reduces the risk of batch rejection during QC testing. This reliability fosters stronger partnerships between API manufacturers and pharmaceutical companies, ensuring that clinical and commercial timelines are met without delay due to quality failures.
- Scalability and Environmental Compliance: The reaction conditions, operating at mild temperatures (25-35°C) and atmospheric pressure, are inherently scalable from laboratory to industrial production scales. The simplified workup procedure, involving precipitation and filtration rather than complex chromatographic separations for every batch, facilitates large-scale manufacturing. Additionally, the reduced toxicity of the reagents simplifies the environmental permitting process for new manufacturing facilities, accelerating the time-to-market for new generic or branded versions of Diquafosol.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating the feasibility of adopting this new supply source. The following questions address common concerns regarding impurity control, metal residues, and process scalability, drawing directly from the experimental data and technical disclosures found in the patent literature. These insights are intended to provide clarity on how this method outperforms legacy technologies in terms of both quality and operational efficiency.
Q: How does this process control toxic metal residues?
A: The process utilizes a specific combination of sodium bicarbonate and sodium carbonate during workup, followed by chelating ion exchange resin (e.g., IRC-748), to reduce metal ion residues to below 10ppm, meeting ophthalmic standards.
Q: What are the critical impurities monitored in this synthesis?
A: Key impurities include UDP, UTP, and higher polyphosphates like U2P5 and U2P6. The novel purification strategy effectively removes U2P5 and U2P6, which are difficult to eliminate in conventional methods.
Q: Why is triethylamine preferred over tributylamine in this route?
A: Triethylamine is significantly less toxic and environmentally hazardous than tributylamine. Furthermore, the specific triethylamine salt intermediates allow for better solubility control and easier removal during the aqueous workup phases.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diquafosol Tetrasodium Supplier
The technological advancements detailed in this report underscore the complexity and precision required to manufacture high-purity P1,P4-di(uridine-5'-) tetraphosphate. As a leader in the fine chemical industry, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to translate such sophisticated laboratory processes into commercial reality. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are equipped with rigorous QC labs capable of detecting impurities at trace levels, guaranteeing that every batch meets stringent purity specifications and regulatory requirements for ophthalmic use.
We invite pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology for your pipeline. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to discuss route feasibility assessments and to obtain specific COA data that demonstrates our capability to deliver this critical intermediate with the highest standards of quality and safety.
