Revolutionizing Dry Eye Treatment: Scalable Synthesis of Diquafosol Tetrasodium via Inorganic Ammonia Catalysis
Revolutionizing Dry Eye Treatment: Scalable Synthesis of Diquafosol Tetrasodium via Inorganic Ammonia Catalysis
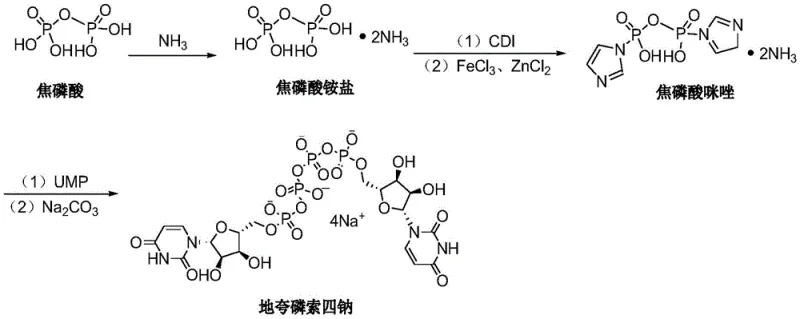
The global ophthalmic pharmaceutical market is witnessing a surge in demand for effective treatments for dry eye disease, with Diquafosol Tetrasodium emerging as a critical P2Y2 receptor agonist. Recent technological advancements disclosed in patent CN113651864A introduce a groundbreaking preparation method that fundamentally alters the safety and efficiency profile of this key active pharmaceutical ingredient (API). Unlike traditional synthetic routes that rely heavily on hazardous organic amines, this novel methodology leverages inorganic ammonia for salification, coupled with a sophisticated Lewis acid catalytic system. For R&D directors and procurement strategists, this patent represents a pivotal shift towards greener, safer, and more cost-effective manufacturing protocols. The transition from toxic organic bases to inorganic reagents not only mitigates regulatory hurdles regarding residual solvents but also streamlines the downstream purification processes, ensuring a higher purity profile essential for ophthalmic applications where safety margins are exceptionally narrow.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex nucleotide analogues like Diquafosol has been plagued by the reliance on volatile and toxic organic bases such as n-tributylamine and triethylamine during the salification and activation stages. These conventional methods present severe challenges for large-scale manufacturing, primarily due to the difficulty in completely removing these high-boiling organic amines from the final product matrix. Residual organic bases pose significant toxicity risks, necessitating extensive and costly purification steps that often involve multiple recrystallizations or chromatographic separations, thereby driving up the overall cost of goods sold (COGS). Furthermore, the handling of large quantities of toxic organic amines imposes stringent safety protocols on the workforce, increasing operational overheads and environmental compliance burdens related to volatile organic compound (VOC) emissions. The inherent instability of some organic amine salts can also lead to inconsistent reaction kinetics, resulting in variable yields and impurity profiles that complicate quality control assurance.
The Novel Approach
The innovative route described in the patent data circumvents these legacy issues by utilizing inorganic ammonia gas or aqueous ammonia for the initial salification of pyrophosphoric acid. This substitution effectively eliminates the introduction of toxic organic nitrogenous residues at the source, drastically simplifying the impurity landscape of the crude product. The process integrates nanofiltration technology to concentrate the ammonium pyrophosphate intermediate, a technique that offers superior control over molecular weight cut-offs and solvent exchange compared to traditional evaporation methods. Following salification, the activation step employs carbonyldiimidazole (CDI) in a controlled manner to generate a highly reactive imidazolide intermediate, which is then subjected to a catalytic condensation with Uridine 5'-monophosphate (UMP). This streamlined approach not only enhances the safety profile for production personnel but also aligns with modern green chemistry principles by reducing the ecological footprint of the manufacturing process.
Mechanistic Insights into FeCl3-ZnCl2 Catalyzed Phosphate Coupling
The core chemical transformation in this synthesis relies on a meticulously optimized Lewis acid catalytic system comprising anhydrous ferric trichloride (FeCl3) and anhydrous zinc chloride (ZnCl2). The mechanistic rationale behind using a mixed metal halide catalyst lies in the synergistic activation of the phosphate electrophile. FeCl3 acts as a potent Lewis acid to coordinate with the oxygen atoms of the activated pyrophosphate intermediate, increasing the electrophilicity of the phosphorus center and facilitating the nucleophilic attack by the phosphate group of UMP. The addition of ZnCl2 modulates the acidity and solubility profile of the catalyst mixture, preventing excessive degradation of the sensitive nucleoside substrate while maintaining high catalytic turnover. The patent specifies a weight ratio of FeCl3 to ZnCl2 between 1:1 and 1:1.5, a narrow window that suggests a delicate balance is required to maximize coupling efficiency without promoting hydrolysis of the pyrophosphate bond. This dual-catalyst strategy ensures that the reaction proceeds smoothly at low temperatures (0-15°C), preserving the stereochemical integrity of the ribose sugar and minimizing the formation of regioisomeric byproducts.
Impurity control is further reinforced through the strategic use of nanofiltration and precise pH regulation during the final salt formation. By concentrating the reaction mixture via nanofiltration prior to activation, the process removes excess water that could otherwise hydrolyze the CDI-activated intermediate, a common side reaction that leads to reduced yields. In the final step, the conversion of the crude acid form to the tetrasodium salt is achieved by adjusting the pH to 8-10 using sodium carbonate. This specific pH range is critical; it ensures complete deprotonation of the phosphate groups to form the stable tetrasodium salt while avoiding alkaline conditions that could degrade the uracil base or the glycosidic bond. The subsequent precipitation with 95% ethanol serves as a final polishing step, leveraging solubility differences to exclude inorganic salts and unreacted starting materials, thereby delivering a product with the high purity specifications required for ophthalmic drug registration.
How to Synthesize Diquafosol Tetrasodium Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing Diquafosol Tetrasodium with high consistency and safety. The process begins with the careful introduction of inorganic ammonia into an aqueous pyrophosphoric acid solution, followed by nanofiltration concentration to remove excess water and isolate the ammonium pyrophosphate species. This intermediate is then activated in dimethylformamide (DMF) using carbonyldiimidazole (CDI) under strictly anhydrous conditions to prevent premature hydrolysis. The subsequent coupling with Uridine 5'-monophosphate requires precise temperature control and the incremental addition of the FeCl3/ZnCl2 catalyst to manage the exotherm and ensure uniform reaction progress. For a comprehensive understanding of the specific operational parameters, stoichiometry, and workup procedures necessary for successful replication, please refer to the standardized synthesis guide below.
- Salification: Introduce inorganic ammonia into pyrophosphoric acid aqueous solution, perform nanofiltration, concentrate under reduced pressure at 45-50°C, and add DMF to obtain Solution A.
- Activation: Dilute Solution A with DMF, add carbonyldiimidazole (CDI) in multiple portions, and stir to form the activated intermediate Solution B.
- Catalytic Condensation: Add Uridine 5'-monophosphate (UMP) and a mixed catalyst of FeCl3/ZnCl2 to Solution B, react at 0-15°C, precipitate with ethyl acetate, and convert to tetrasodium salt using sodium carbonate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers compelling economic and logistical advantages that extend beyond simple yield improvements. The elimination of toxic organic amines like n-tributylamine removes a significant cost center associated with hazardous material handling, storage, and specialized waste disposal. By switching to inorganic ammonia, a commodity chemical with a stable and abundant global supply, manufacturers can insulate their production costs from the volatility often seen in the specialty organic amine market. Furthermore, the simplified purification train, enabled by the cleaner reaction profile and nanofiltration steps, reduces the consumption of expensive chromatography resins and organic solvents, leading to substantial cost savings in raw material utilization. The enhanced safety profile also translates to lower insurance premiums and reduced regulatory compliance costs, making the overall manufacturing operation more lean and resilient against supply chain disruptions.
- Cost Reduction in Manufacturing: The replacement of expensive and toxic organic bases with inexpensive inorganic ammonia drastically lowers the raw material costs associated with the salification step. Additionally, the removal of complex amine stripping and recovery units simplifies the plant infrastructure, reducing both capital expenditure (CAPEX) and operational expenditure (OPEX). The high selectivity of the FeCl3/ZnCl2 catalyst system minimizes the formation of difficult-to-remove impurities, thereby reducing the number of recrystallization cycles needed to meet pharmacopeial standards. This efficiency gain directly impacts the bottom line by shortening the batch cycle time and increasing the throughput of existing manufacturing facilities without the need for major equipment upgrades.
- Enhanced Supply Chain Reliability: Relying on inorganic ammonia and standard metal halides like ferric chloride and zinc chloride ensures a stable supply of critical reagents, as these are produced on a massive industrial scale globally. Unlike specialized organic amines which may have limited suppliers and long lead times, these inorganic inputs are readily available from multiple sources, mitigating the risk of production stoppages due to raw material shortages. The robustness of the nanofiltration process also allows for greater flexibility in sourcing solvent grades, as the filtration step can effectively purify the intermediate regardless of minor variations in input quality. This supply chain resilience is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with nanofiltration and controlled precipitation being unit operations that translate seamlessly from pilot plant to commercial tonnage scales. The significant reduction in VOC emissions, achieved by eliminating volatile organic amines, ensures strict adherence to increasingly stringent environmental regulations across major manufacturing hubs. This eco-friendly profile not only future-proofs the manufacturing site against tightening emission caps but also enhances the brand reputation of the supplier as a sustainable partner. The simplified waste stream, predominantly consisting of inorganic salts and recoverable solvents, facilitates easier treatment and disposal, further reducing the environmental liability and associated costs of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the patented synthesis of Diquafosol Tetrasodium. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the process's viability for commercial adoption. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios. The answers highlight the specific advantages of the inorganic ammonia route and the catalytic system, offering a clear perspective on why this method represents a superior alternative to legacy technologies.
Q: Why is inorganic ammonia preferred over organic amines in this synthesis?
A: Inorganic ammonia eliminates the toxicity risks associated with organic bases like n-tributylamine and triethylamine. It ensures zero toxic residue in the final API, enhancing patient safety and simplifying waste treatment.
Q: What is the role of the FeCl3 and ZnCl2 catalyst system?
A: The mixed Lewis acid catalyst system facilitates the efficient coupling of the activated pyrophosphate intermediate with Uridine 5'-monophosphate, improving reaction selectivity and yield while minimizing side reactions.
Q: How does nanofiltration improve the process scalability?
A: Nanofiltration allows for the efficient concentration and purification of the ammonium pyrophosphate intermediate without thermal degradation, making the process more robust and easier to scale for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diquafosol Tetrasodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthesis route in meeting the growing global demand for high-quality dry eye therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of nucleotide chemistry, including rigorous moisture control and specialized corrosion-resistant reactors for Lewis acid catalysis. We are committed to delivering Diquafosol Tetrasodium with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify the absence of toxic residues and ensure batch-to-batch consistency.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge technology for their supply chains. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this greener synthesis route can optimize your COGS. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and security in your ophthalmic drug portfolio.
