Advanced Synthesis of Citicoline Sodium: Scalable Route for Pharmaceutical Manufacturing
Introduction to Next-Generation Citicoline Sodium Manufacturing
The pharmaceutical industry constantly seeks more efficient pathways for producing critical neuroprotective agents, and the synthesis of Citicoline Sodium (CAS 33818-15-4) is a prime example of where process innovation drives commercial viability. As detailed in the recent patent CN114057813B, a novel method has been disclosed that fundamentally restructures the synthetic approach to this vital nucleoside derivative. Unlike legacy processes that rely on cumbersome activation steps or biological fermentation which struggles with scale, this new methodology leverages a direct phosphorylation strategy using phosphorus oxychloride. This technical breakthrough allows for a streamlined three-step reaction sequence that bypasses the need for pre-activating cytidine into morpholine derivatives or utilizing expensive carbodiimide coupling agents. For R&D directors and procurement managers alike, this represents a significant shift towards a more robust, cost-effective, and environmentally sustainable manufacturing protocol that ensures a reliable supply of high-purity pharmaceutical intermediates.
The strategic importance of this patent lies in its ability to reconcile high yield with operational simplicity. Historically, the production of cytidine diphosphate choline has been plagued by low overall yields and complex purification requirements due to the formation of stubborn byproducts. The disclosed invention addresses these pain points by optimizing the reaction conditions to favor the formation of the desired phosphodiester bond while minimizing hydrolysis of the sensitive nucleoside backbone. By maintaining strict temperature controls between 0°C and 25°C and utilizing specific phosphate solvents, the process achieves a total yield exceeding 80% with HPLC purity greater than 99.5%. This level of efficiency is not merely a laboratory curiosity but a tangible asset for supply chain heads looking to secure long-term contracts for active pharmaceutical ingredients (APIs) and their precursors without the risk of batch-to-batch variability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
To fully appreciate the value of this new synthesis route, one must first understand the significant bottlenecks inherent in the prior art. Early methods, such as those reported by Kiyomi Kikugawa in 1971, relied heavily on p-toluenesulfonyl chloride for activation. While chemically feasible, this approach generates substantial amounts of sulfonate waste, creating a heavy environmental burden and complicating the waste disposal logistics for large-scale facilities. Furthermore, the use of sulfonyl chlorides often requires stringent anhydrous conditions and excess reagents, driving up the raw material costs and increasing the hazard profile of the manufacturing site. Later iterations attempted to improve selectivity by using cytidylic acid mono-morpholine, but this introduced a new set of problems, specifically the need for additional synthetic steps to prepare the morpholine intermediate, thereby lengthening the production cycle and accumulating yield losses at each stage.
Another prevalent conventional method involves the use of dicyclohexylcarbodiimide (DCC) or similar condensing agents to couple cytidylic acid derivatives with phosphorylcholine. While DCC is a powerful dehydrating agent, its usage in GMP manufacturing is increasingly disfavored due to the formation of dicyclohexylurea (DCU), a byproduct that is notoriously difficult to remove completely from the final product matrix. The presence of DCU residues can compromise the safety profile of the pharmaceutical intermediate, necessitating extensive and costly purification steps such as repeated chromatography or recrystallization. Additionally, the biological synthesis routes, while green in theory, currently suffer from low titers and the high cost of enzyme preparation, rendering them unsuitable for the ton-scale production required by the global market. These cumulative inefficiencies result in higher COGS (Cost of Goods Sold) and longer lead times, which are critical disadvantages in a competitive supply chain.
The Novel Approach
The methodology presented in CN114057813B offers a decisive break from these traditional constraints by employing a direct phosphorylation-condensation strategy. The core innovation lies in the generation of a reactive cytidine-5'-phosphorodichloridate intermediate in situ, which is then immediately condensed with phosphorylcholine chloride without an intervening hydrolysis step. This "one-pot" philosophy drastically reduces the number of unit operations, solvent swaps, and isolation steps required. By eliminating the need for pre-synthesized activated nucleotides or expensive coupling reagents like DCC, the process inherently lowers the material cost and simplifies the reactor setup. The use of triethyl phosphate or trimethyl phosphate as both solvent and reactant further enhances the atom economy of the reaction, ensuring that the phosphorylation proceeds with high efficiency.
Moreover, the novel approach incorporates a sophisticated purification protocol that replaces the traditional activated carbon treatment with ion exchange resin technology. This shift is crucial for maintaining high recovery rates while achieving exceptional purity levels. Activated carbon, while effective at removing colored impurities, often non-specifically adsorbs the target nucleoside product, leading to significant yield erosion. In contrast, ion exchange resins can be tuned to selectively retain ionic impurities while allowing the zwitterionic citicoline sodium to pass through or be eluted under specific pH conditions. This results in a finished product with HPLC purity consistently above 99.5%, meeting the rigorous standards of international pharmacopeias. The combination of a shortened synthetic route and advanced purification makes this method uniquely positioned for commercial scale-up, offering a compelling value proposition for manufacturers seeking to optimize their production lines.
Mechanistic Insights into Direct Phosphorylation and Condensation
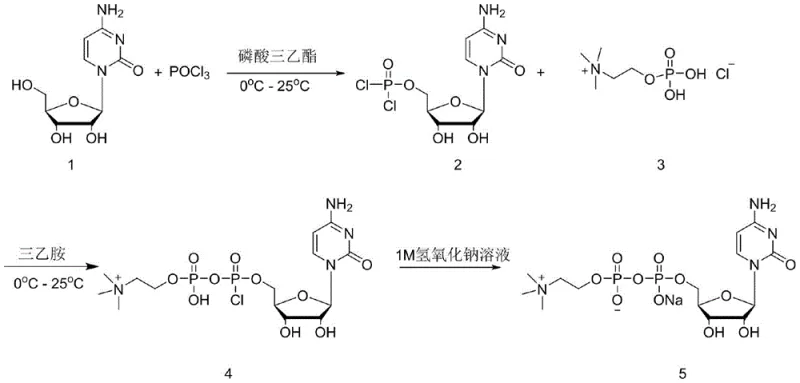
The chemical elegance of this synthesis rests on the precise control of phosphorus chemistry to activate the 5'-hydroxyl group of cytidine. In the initial step, cytidine reacts with phosphorus oxychloride (POCl3) in a phosphate ester solvent at low temperatures (0°C). Under these conditions, the primary alcohol at the 5' position of the ribose sugar acts as a nucleophile, attacking the electrophilic phosphorus center of POCl3. This results in the displacement of one chloride ion and the formation of a cytidine-5'-phosphorodichloridate species. The choice of solvent is critical here; trialkyl phosphates not only dissolve the polar nucleoside but also stabilize the reactive intermediate, preventing premature hydrolysis which would lead to the formation of inactive cytidine-5'-monophosphate (CMP). The reaction is typically monitored via liquid chromatography to ensure complete consumption of the starting cytidine before proceeding to the next stage.
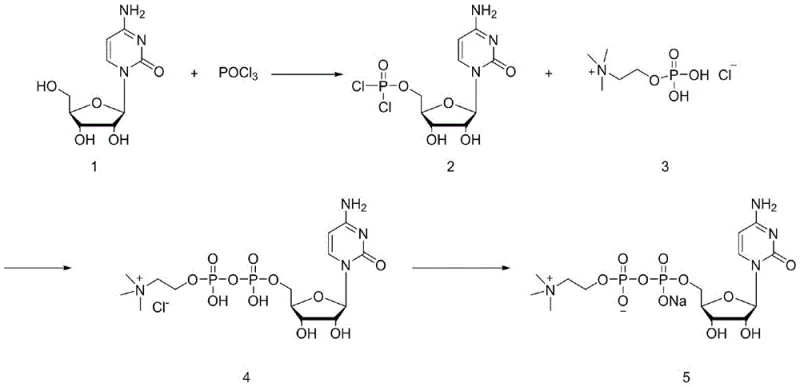
Following the formation of the dichlorophosphate intermediate, the reaction mixture is treated with a base, such as triethylamine or diisopropylethylamine, to scavenge the hydrogen chloride generated during the subsequent condensation. Phosphorylcholine chloride is then introduced, acting as a nucleophile to attack the remaining electrophilic sites on the phosphorus atom of the cytidine intermediate. This step forms the pyrophosphate linkage characteristic of citicoline. A key mechanistic advantage of this route is that the condensation occurs directly on the dichlorophosphate intermediate without hydrolyzing it to the monophosphate first. This preserves the high-energy phosphate bond potential and drives the equilibrium towards the formation of the diphosphate product. Finally, the reaction is quenched with aqueous sodium hydroxide, which hydrolyzes any remaining P-Cl bonds to P-OH and converts the acidic protons to sodium salts, yielding the final citicoline sodium product ready for purification.
Impurity control is embedded within the reaction mechanism itself. One of the major degradation pathways for cytidine derivatives is the deamination of the cytosine ring to form uridine derivatives, which are structurally similar and difficult to separate. The patented process mitigates this risk by maintaining mild reaction temperatures (0°C - 25°C) and avoiding strongly acidic or basic conditions during the critical bond-forming steps. Furthermore, the use of ion exchange resin in the workup phase provides a secondary barrier against impurities. The resin effectively separates the target citicoline sodium from unreacted phosphorylcholine, inorganic phosphates, and any trace uridine byproducts based on differences in charge density and pKa values. This dual strategy of kinetic control during synthesis and thermodynamic separation during purification ensures that the final impurity profile is exceptionally clean, satisfying the stringent requirements for neuroactive pharmaceutical ingredients.
How to Synthesize Citicoline Sodium Efficiently
Implementing this synthesis route in a production environment requires adherence to specific operational parameters to maximize yield and safety. The process is designed to be robust, tolerating slight variations in reagent addition rates while maintaining high selectivity. The following guide outlines the critical phases of the manufacturing protocol, emphasizing the conditions necessary to achieve the reported >80% yield and >99.5% purity. Operators should note that temperature control during the phosphorylation step is paramount to prevent exothermic runaways and side reactions.
- Disperse cytidine in a phosphate solvent (e.g., triethyl phosphate) at 0°C and add phosphorus oxychloride dropwise, followed by stirring at room temperature to form the dichlorophosphate intermediate.
- Cool the reaction mixture to 0°C, add a base such as triethylamine, and introduce phosphorylcholine chloride in batches, heating to 25°C to facilitate condensation without prior hydrolysis.
- Hydrolyze the condensation intermediate with aqueous sodium hydroxide, concentrate the aqueous phase, and purify the crude product using anion exchange resin followed by ethanol recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis method offers tangible benefits that extend beyond simple chemistry. The most immediate impact is seen in the reduction of raw material costs and the simplification of the supply base. By eliminating the need for specialized coupling agents like DCC or DCI, which are not only expensive but also subject to market volatility, manufacturers can stabilize their input costs. Furthermore, the reagents used in this process—cytidine, phosphorus oxychloride, and triethylamine—are commodity chemicals available from multiple global suppliers. This diversification of the supply base reduces the risk of single-source dependency and ensures continuity of supply even during market disruptions. The simplified workflow also translates to reduced labor hours and lower utility consumption per kilogram of product, contributing to a leaner cost structure.
Scalability is another critical advantage that addresses the concerns of supply chain heads planning for long-term growth. The patent data includes successful execution of the process at a 2-kilogram scale (Example 8), demonstrating that the reaction kinetics and heat transfer characteristics remain favorable as the batch size increases. Unlike enzymatic processes that require complex bioreactor setups and sterile conditions, this chemical synthesis can be performed in standard glass-lined or stainless steel reactors found in most fine chemical plants. The absence of filtration steps to remove solid byproducts like DCU (dicyclohexylurea) further streamlines the operation, reducing the downtime associated with equipment cleaning and maintenance. This operational efficiency allows for faster turnaround times between batches, effectively increasing the annual production capacity of existing facilities without the need for significant capital expenditure on new infrastructure.
From an environmental and regulatory perspective, this method aligns well with modern sustainability goals and waste reduction mandates. Traditional methods utilizing p-toluenesulfonyl chloride generate stoichiometric amounts of sulfonate waste, which requires costly treatment before disposal. In contrast, the byproducts of the POCl3 method are primarily inorganic salts and amines, which are easier to manage and treat in standard wastewater facilities. The high purity of the crude product obtained after crystallization also reduces the load on downstream purification units, minimizing the volume of solvent waste generated during recrystallization. For companies aiming to reduce their carbon footprint and meet ISO 14001 standards, adopting this cleaner synthesis route provides a clear pathway to compliance. The combination of cost efficiency, supply security, and environmental stewardship makes this technology a strategically sound choice for securing a reliable citicoline sodium supplier partnership.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of citicoline sodium, derived from the specific advantages and operational details outlined in the patent literature. These insights are intended to clarify the feasibility of adoption and the quality expectations associated with this manufacturing route.
Q: Why is this synthesis method superior to traditional DCC-mediated coupling?
A: Traditional methods often utilize dicyclohexylcarbodiimide (DCC) as a condensing agent, which generates dicyclohexylurea (DCU) as a byproduct that is difficult to remove and increases waste. The novel route described in CN114057813B eliminates the need for DCC entirely by using phosphorus oxychloride for direct activation, significantly simplifying post-treatment and reducing hazardous waste generation.
Q: How does the process ensure high purity (>99.5%) without activated carbon?
A: The process achieves high purity through a combination of selective reaction conditions that minimize side products like uridine, followed by a robust purification strategy. Instead of using activated carbon, which can lead to product adsorption losses, the method employs ion exchange resin treatment and controlled recrystallization in an alcohol/water system to effectively remove ionic impurities and residual solvents.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the route is specifically designed for scalability. It utilizes common, commercially available reagents like cytidine and phosphorus oxychloride, avoids complex biological enzymes or expensive coupling agents, and has been demonstrated successfully at multi-kilogram scales (e.g., 2kg batch size) with stable yields exceeding 80%, making it highly viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Citicoline Sodium Supplier
The technical advancements detailed in patent CN114057813B underscore the potential for high-efficiency manufacturing of neuroprotective intermediates, yet translating such protocols into consistent commercial supply requires deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this capability, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reaction vessels capable of handling sensitive phosphorylation chemistries under strict temperature control, ensuring that the delicate balance between yield and purity is maintained at every scale. We understand that for pharmaceutical applications, consistency is key; therefore, our stringent purity specifications and rigorous QC labs employ advanced HPLC and NMR techniques to verify that every batch meets or exceeds the 99.5% purity benchmark established by the latest research.
We invite global partners to collaborate with us to optimize their supply chains for citicoline sodium and related nucleoside derivatives. By partnering with NINGBO INNO PHARMCHEM, you gain access not just to a product, but to a comprehensive technical service that includes a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential clients to contact our technical procurement team to request specific COA data from our recent pilot runs and to discuss route feasibility assessments for your broader portfolio. Let us help you secure a stable, high-quality supply of critical pharmaceutical intermediates that drives your product development forward without compromising on cost or compliance.
