Advanced T3P-Mediated Synthesis of Citicoline Sodium: Enhancing Purity and Supply Chain Safety for Global Pharma
Introduction to Next-Generation Citicoline Sodium Manufacturing
The pharmaceutical landscape for neuroprotective agents is undergoing a significant transformation driven by the demand for higher purity and greener synthesis routes. Patent CN110684066A introduces a groundbreaking methodology for the preparation of Citicoline Sodium, a critical active pharmaceutical ingredient (API) used extensively in the treatment of acute cerebral infarction and cognitive disorders. This innovation addresses the longstanding limitations of conventional chemical synthesis by replacing hazardous reagents with a mild, efficient condensing agent known as 1-propylphosphoric cyclic anhydride (T3P). The result is a robust process capable of delivering Citicoline Sodium with a mass content exceeding 99.0%, while strictly controlling critical impurities to levels below 0.2%. For global procurement teams and R&D directors, this patent represents a pivotal shift towards sustainable manufacturing that aligns with modern regulatory standards for residual solvents and environmental safety.
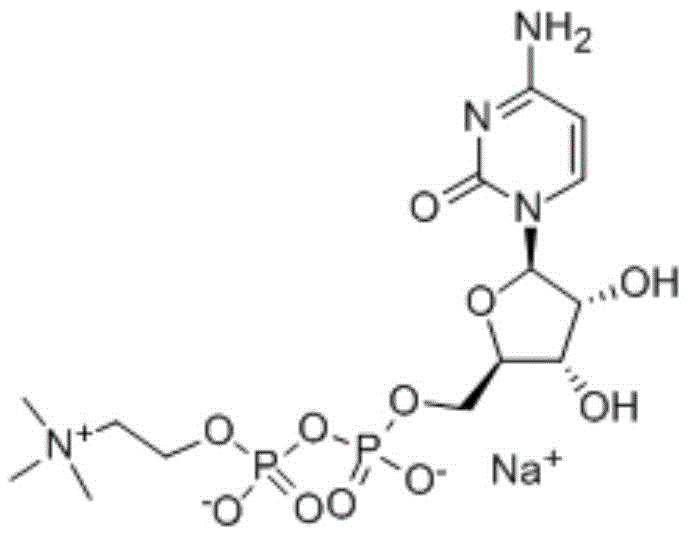
By leveraging this advanced synthetic pathway, manufacturers can overcome the complex purification challenges associated with biological fermentation and older chemical methods. The technology ensures a consistent supply of high-quality intermediates, reducing the risk of batch failures due to solvent residues or unexpected byproduct formation. As the industry moves towards stricter ICH guidelines, adopting such forward-thinking synthetic strategies becomes not just an option, but a necessity for maintaining competitiveness in the reliable pharmaceutical intermediates supplier market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
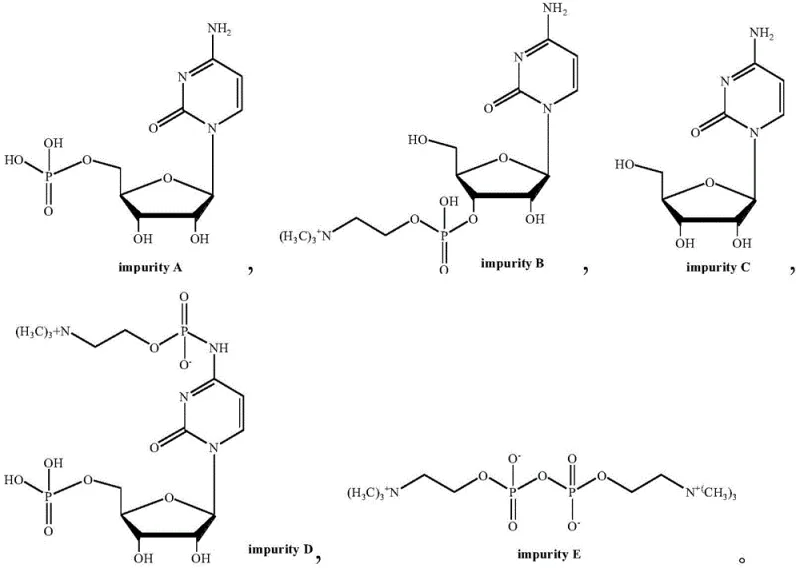
Historically, the industrial production of Citicoline Sodium has relied heavily on a cumbersome chemical synthesis route that poses significant safety and environmental risks. The traditional process typically begins with phosphorylcholine chloride calcium salt, requiring a dehydration step using benzene, a known Class 1 carcinogen, under reflux conditions. This is followed by activation using highly corrosive and toxic reagents such as oxalyl chloride, acetyl chloride, or solid phosgene in organic amine solvents. These harsh conditions inevitably lead to the formation of multiple side products, including the problematic Impurities D and E, which are difficult to remove and compromise the final API quality. Furthermore, the generation of large volumes of acidic wastewater and solid waste salts like calcium carbonate creates a substantial burden on waste treatment facilities, driving up operational costs and complicating environmental compliance.
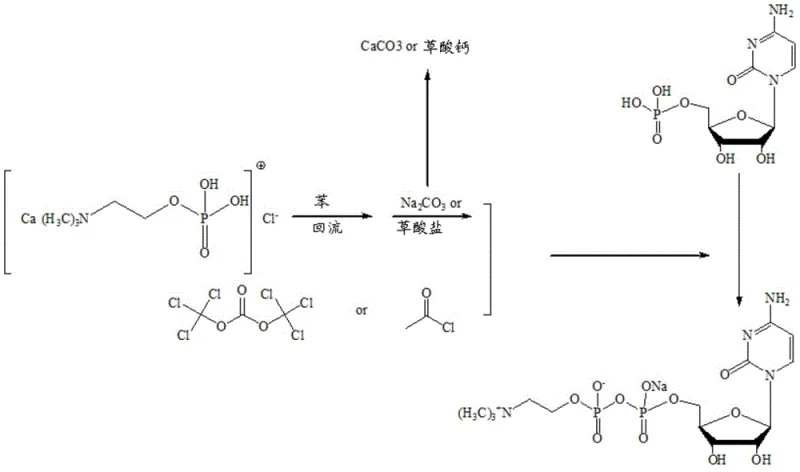
The reliance on such aggressive chemistry also necessitates specialized equipment with high corrosion resistance, increasing capital expenditure for manufacturing plants. The complexity of the post-processing steps, which often involve vacuum distillation and column chromatography to achieve acceptable purity, further reduces overall yield and extends production cycles. For supply chain managers, these inefficiencies translate into longer lead times and higher vulnerability to raw material shortages, making the traditional route increasingly unsustainable in a cost-sensitive global market.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a direct condensation strategy mediated by T3P, fundamentally simplifying the reaction architecture. By dispersing 5'-cytidylic acid and phosphorylcholine chloride directly in a safe aprotic solvent like acetonitrile, the process eliminates the need for preliminary dehydration with benzene and the subsequent removal of calcium ions. The use of T3P as a coupling agent allows the reaction to proceed under mild thermal conditions, avoiding the extreme activation energies that drive side reactions in the traditional method. This streamlined workflow not only shortens the synthetic route but also drastically reduces the generation of hazardous waste, offering a clear pathway for cost reduction in pharmaceutical intermediates manufacturing through simplified downstream processing and solvent recovery.
Mechanistic Insights into T3P-Mediated Phosphorylation
The core of this technological breakthrough lies in the unique reactivity profile of 1-propylphosphoric cyclic anhydride (T3P). Unlike traditional activators such as acid chlorides or phosgene, which generate highly reactive acyl intermediates prone to non-selective attacks, T3P operates through a synergistic condensation mechanism that favors the target phosphodiester bond formation. The cyclic structure of T3P provides excellent activation capability for the phosphate group without excessively activating the amino groups on the cytidine ring. This selectivity is crucial because it prevents the formation of Impurity D, a common byproduct in traditional synthesis where the amino group is inadvertently acylated. Additionally, the absence of a discrete, highly unstable intermediate transition state minimizes the occurrence of Impurity E, ensuring that the reaction proceeds cleanly towards the desired Citicoline acid precursor.
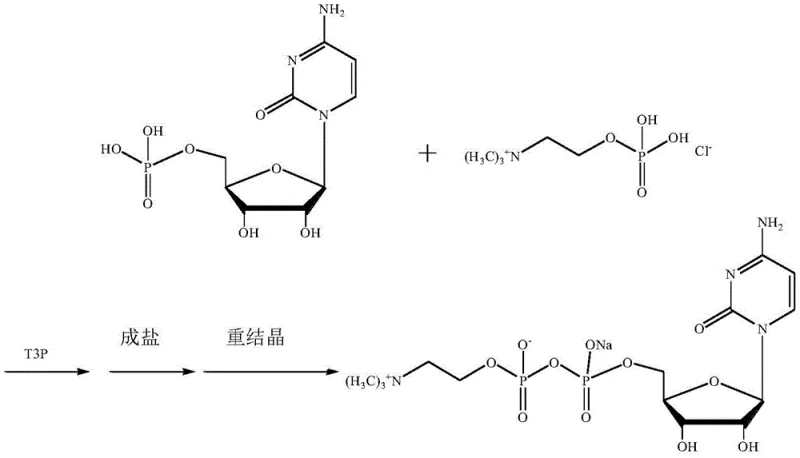
Furthermore, the steric hindrance inherent in the T3P molecule plays a protective role during the reaction. It preferentially activates the phosphate hydrogen over the sterically hindered hydroxyl groups at the 3' and 4' positions of the ribose ring. This kinetic control ensures that the primary condensation occurs at the correct site, significantly reducing the formation of Impurity B. The mild nature of T3P also means it does not react violently with trace moisture, removing the strict requirement for anhydrous conditions and Class 1 solvents. This mechanistic advantage translates directly into a more robust process window, allowing for scalable production with consistent quality outcomes that meet the rigorous demands of high-purity pharmaceutical intermediates.
Impurity control is further enhanced by the specific crystallization protocols employed in the later stages. By carefully managing cooling rates and solvent compositions during the precipitation of Citicoline acid and its subsequent sodium salt, the process effectively excludes remaining trace impurities like A, B, and C into the mother liquor. The result is a final product with exceptional stability, as evidenced by long-term testing where impurity levels remain negligible even after extended storage periods. This level of control is essential for ensuring the safety and efficacy of the final neurological therapeutic formulations.
How to Synthesize Citicoline Sodium Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize yield and purity. The process is designed to be scalable, moving seamlessly from laboratory optimization to commercial production without the need for exotic equipment. Key to success is the management of the T3P addition rate and temperature to maintain kinetic control, followed by a strategic crystallization sequence that leverages solubility differences to purge impurities. Detailed standard operating procedures for this methodology are critical for reproducibility.
- Disperse 5'-cytidylic acid and phosphorylcholine chloride in an aprotic organic solvent like acetonitrile, then add 1-propylphosphoric cyclic anhydride (T3P) at controlled temperatures to initiate condensation.
- Cool the reaction filtrate to precipitate citicoline acid crystals, filtering out insolubles to separate the intermediate from the reaction matrix.
- React the isolated citicoline acid with a sodium base such as sodium bicarbonate in water, followed by recrystallization using ethanol/water mixtures to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this T3P-based synthesis offers profound strategic advantages beyond mere technical superiority. The elimination of benzene and phosgene derivatives removes significant regulatory hurdles and safety liabilities from the manufacturing process. This shift not only streamlines the approval process for new drug filings but also reduces the insurance and compliance costs associated with handling hazardous materials. By simplifying the waste stream to mostly recyclable organic solvents and benign salts, the process aligns perfectly with green chemistry initiatives, enhancing the corporate sustainability profile of the supplier.
- Cost Reduction in Manufacturing: The streamlined nature of the T3P route eliminates multiple unit operations such as calcium removal and extensive solvent exchanges. This reduction in process steps directly lowers energy consumption and labor costs. Furthermore, the ability to recycle reaction solvents like acetonitrile after simple distillation significantly reduces raw material expenses. The avoidance of expensive corrosion-resistant equipment required for handling strong acids and phosgene also translates into lower capital depreciation costs, providing a substantial economic edge in competitive bidding scenarios.
- Enhanced Supply Chain Reliability: Traditional methods are often bottlenecked by the availability and strict handling regulations of Class 1 solvents and toxic activators. By switching to widely available and safer reagents, the supply chain becomes more resilient to regulatory shocks and logistical disruptions. The shorter reaction times and simplified workup procedures mean faster batch turnover, allowing suppliers to respond more agilely to fluctuations in market demand. This reliability is crucial for securing long-term contracts with major pharmaceutical companies that prioritize uninterrupted supply continuity.
- Scalability and Environmental Compliance: The mild reaction conditions and reduced waste generation make this process inherently easier to scale from pilot plant to multi-ton production. The absence of highly toxic byproducts simplifies effluent treatment, reducing the environmental footprint and ensuring compliance with increasingly stringent global environmental regulations. This scalability ensures that the supplier can meet growing global demand for neuroprotective agents without compromising on quality or facing production caps due to environmental permits.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis method is vital for stakeholders evaluating its potential impact on their supply chains. The following questions address common concerns regarding purity, stability, and regulatory alignment. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a transparent view of the technology's capabilities.
Q: How does the T3P method improve impurity profiles compared to traditional phosgene routes?
A: The T3P method avoids the formation of highly reactive acyl chloride intermediates found in traditional phosgene routes. This kinetic control prevents the activation of amino groups on the cytidine ring, effectively eliminating Impurity D and significantly reducing Impurity E, resulting in a cleaner product profile.
Q: Why is the elimination of benzene critical for regulatory compliance in API manufacturing?
A: Benzene is classified as a Class 1 solvent with unacceptable toxicity and carcinogenic potential. By replacing benzene dehydration steps with mild T3P condensation in safer solvents like acetonitrile, manufacturers avoid stringent residual solvent limits and reduce the environmental burden of hazardous waste disposal.
Q: What are the stability advantages of Citicoline Sodium produced via this new route?
A: Long-term stability studies indicate that Citicoline Sodium produced via this method maintains content above 99.0% even after 18 months under accelerated conditions. The controlled crystallization process minimizes the entrapment of hydrolytic impurities like Impurity C, ensuring superior shelf-life for finished dosage forms.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Citicoline Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthesis technologies like the T3P-mediated route is key to securing the future of pharmaceutical supply chains. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing settings. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Citicoline Sodium meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage these technological advancements for your product portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and product quality.
