Revolutionizing 2,6-Dichloropurine Production: A Deep Dive into Ionic Liquid Catalysis and Commercial Scalability
Revolutionizing 2,6-Dichloropurine Production: A Deep Dive into Ionic Liquid Catalysis and Commercial Scalability
The pharmaceutical industry constantly seeks robust, scalable, and environmentally sustainable pathways for critical antiviral intermediates. Patent CN114349754B, published in May 2023, introduces a transformative synthesis method for 2,6-dichloropurine, a pivotal building block for nucleoside analogs such as fludarabine. This technology leverages the unique solvation and catalytic properties of chloride ionic liquids to overcome the longstanding limitations of traditional diazotization processes. By replacing hazardous heavy metal catalysts with tunable ionic media, this approach not only enhances reaction selectivity but also aligns with modern green chemistry principles essential for GMP manufacturing. The structural integrity of the target molecule, shown below, is preserved with exceptional fidelity, minimizing degradation pathways that typically plague acidic chlorination reactions.
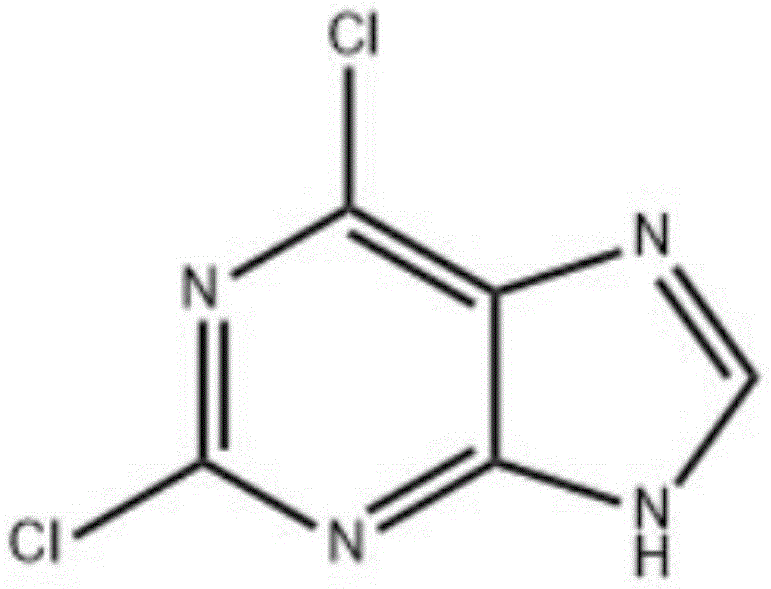
For R&D directors and process chemists, the implications of this patent extend beyond mere yield improvements; it represents a fundamental shift in how heterocyclic amines are functionalized. The ability to achieve content levels exceeding 99 percent without extensive chromatographic purification suggests a streamlined downstream processing workflow. Furthermore, the protocol's compatibility with mother liquor recycling introduces a circular economy aspect to the synthesis, addressing both cost pressures and environmental compliance mandates simultaneously. As we analyze the technical nuances, it becomes evident that this methodology offers a reliable pharmaceutical intermediate supplier with a distinct competitive advantage in the antiviral market sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 2,6-dichloropurine has been fraught with significant technical and regulatory hurdles that compromise both economic viability and product quality. Traditional routes often involve the direct chlorination of hypoxanthine 1-N-oxide using phosphorus oxychloride, a process hampered by the scarcity and high cost of the starting material. Alternative pathways utilizing 2,6-dichloro-4,5-diaminopyrimidine suffer from poor thermal stability, where chlorine atoms are prone to hydrolysis under the requisite acidic and high-temperature conditions, leading to diminished yields and complex impurity profiles. Perhaps most critically for pharmaceutical applications, established diazotization methods frequently employ cuprous chloride or zinc chloride as catalysts. While these transition metals facilitate the reaction, they introduce severe contamination risks; removing trace heavy metals to meet ICH Q3D guidelines requires additional, costly purification steps involving chelating resins or repeated recrystallizations, which further erodes overall process efficiency and throughput.
The Novel Approach
In stark contrast to these legacy technologies, the method disclosed in CN114349754B utilizes optimized chloride ionic liquids, such as 1-butyl-3-methylimidazolium chloride ([BMIm]Cl) or 1-ethyl-3-methylimidazolium chloride ([EMIm]Cl), to create a highly efficient reaction environment. These ionic liquids serve a dual function as both the reaction medium and the catalyst, eliminating the need for exogenous heavy metal additives entirely. The structural mimicry between the imidazole cation of the ionic liquid and the purine substrate facilitates superior solubility and molecular activation, driving the diazotization of 2-amino-6-chloropurine with sodium nitrite to near-quantitative conversion. As illustrated by the catalyst structure below, the nitrogen-containing heterocyclic framework provides a stabilizing interaction that suppresses common side reactions such as azo coupling, ring opening, and hydrolytic dechlorination.
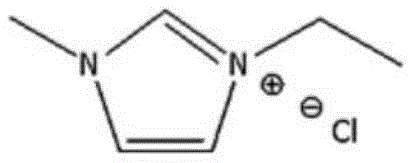
This novel approach fundamentally alters the thermodynamic and kinetic landscape of the synthesis, allowing the reaction to proceed smoothly at mild temperatures ranging from 0°C to 20°C. The result is a dramatic improvement in selectivity, where the desired 2,6-dichloropurine is obtained with yields consistently surpassing 98 percent and purity levels exceeding 99 percent. Moreover, the process incorporates an innovative post-treatment strategy where the extraction mother liquor is cooled to minus 10°C to precipitate impurities and salts, enabling the filtrate to be directly recycled for subsequent batches. This closed-loop capability not only minimizes solvent consumption and waste generation but also significantly reduces the variable costs associated with raw material procurement, presenting a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Ionic Liquid-Promoted Diazotization
To fully appreciate the technical superiority of this synthesis, one must examine the mechanistic role played by the chloride ionic liquid in facilitating the diazotization of the amino-purine substrate. In conventional aqueous acidic media, the protonation of the purine nitrogen atoms can compete with the desired diazotization of the exocyclic amine, often leading to incomplete conversion or decomposition of the sensitive diazonium intermediate. However, the ionic liquid environment modifies the acidity function of the system, creating a microenvironment that stabilizes the nitrosonium ion (NO+) generated from sodium nitrite and hydrochloric acid. The chloride anions within the ionic lattice likely participate in hydrogen bonding networks with the substrate, orienting the 2-amino-6-chloropurine molecule in a conformation that favors nucleophilic attack by the nitrosonium species. This precise molecular organization lowers the activation energy barrier for the formation of the diazonium salt, ensuring rapid and complete conversion even at the lower end of the temperature spectrum (0°C), thereby preserving the integrity of the chlorine substituents against hydrolytic displacement.
Furthermore, the suppression of side reactions is a critical mechanistic advantage that directly impacts the impurity profile of the final API intermediate. In the absence of the ionic liquid promoter, the reactive diazonium intermediate is susceptible to coupling with unreacted amine to form azo dyes, or it may undergo hydrolysis to revert to the hydroxy-purine or suffer ring degradation under harsh acidic conditions. The bulky organic cations of the ionic liquid, such as the butyl or ethyl groups on the imidazolium ring, provide a steric shield around the reactive centers, effectively isolating the diazonium species from unwanted bimolecular collisions. This "cage effect" ensures that the subsequent replacement of the diazo group by a chloride ion (from the abundant chloride source in the medium) proceeds with high fidelity. Consequently, the resulting crude product requires minimal purification, typically just a single recrystallization from methanol, to achieve the stringent purity specifications demanded by downstream nucleoside synthesis, validating the process as a high-purity pharmaceutical intermediate solution.
How to Synthesize 2,6-Dichloropurine Efficiently
Implementing this advanced synthesis protocol requires careful attention to reagent ratios and temperature control to maximize the benefits of the ionic liquid system. The general procedure involves charging a reaction vessel with the selected chloride ionic liquid, followed by the addition of concentrated hydrochloric acid and the substrate, 2-amino-6-chloropurine. Once a homogeneous mixture is achieved, sodium nitrite is introduced slowly to manage the exotherm and gas evolution, maintaining the internal temperature strictly between 0°C and 20°C for a duration of 0.5 to 4 hours depending on the specific ionic liquid variant used. Following the reaction completion, the product is isolated via ethyl acetate extraction, and the organic phase is concentrated to yield the crude solid, which is then purified by methanol recrystallization. For detailed operational parameters, stoichiometry, and safety considerations, please refer to the standardized synthesis steps outlined below.
- Charge the reactor with optimized chloride ionic liquid (e.g., [BMIm]Cl) and concentrated hydrochloric acid, then add 2-amino-6-chloropurine under stirring.
- Maintain temperature between 0-20°C and slowly add sodium nitrite to initiate the diazotization reaction, stirring for 0.5 to 4 hours.
- Extract the crude product with ethyl acetate, recrystallize from methanol, and recycle the cooled mother liquor for subsequent batches.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, the adoption of this ionic liquid-based synthesis offers profound advantages that extend well beyond the laboratory bench. For procurement managers tasked with optimizing the cost of goods sold (COGS), the elimination of expensive and regulated heavy metal catalysts represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. Traditional processes relying on copper or zinc salts incur hidden costs related to metal scavenging agents, specialized waste disposal for heavy metal sludge, and extended quality control testing to verify residual metal limits. By transitioning to a metal-free ionic liquid system, manufacturers can streamline their vendor qualification processes and reduce the complexity of their supply chain, ensuring a more consistent and reliable supply of critical raw materials without the volatility associated with transition metal markets.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the drastic simplification of the downstream processing workflow. Since the ionic liquid promotes such high selectivity, the formation of difficult-to-remove impurities is minimized, reducing the number of recrystallization cycles required to meet purity specs. Additionally, the ability to recycle the mother liquor after a simple cooling and filtration step means that a significant portion of the solvent and ionic liquid inventory is retained within the process loop rather than being discharged as waste. This circular utilization of materials leads to substantial cost savings over time, as the consumption of fresh ionic liquid and hydrochloric acid is markedly decreased, directly improving the gross margin profile for large-scale production campaigns.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on specialized reagents that have limited global suppliers. The reagents utilized in this novel process—concentrated hydrochloric acid, sodium nitrite, and commodity imidazole derivatives—are widely available from multiple chemical distributors, mitigating the risk of single-source bottlenecks. Furthermore, the robustness of the reaction conditions, which tolerate a relatively wide temperature window (0-20°C) and do not require cryogenic cooling or anhydrous environments, enhances the operational reliability of the manufacturing plant. This resilience ensures that production schedules can be maintained even during periods of utility fluctuation or minor equipment deviations, providing downstream API manufacturers with the dependable delivery timelines necessary for their own clinical or commercial planning.
- Scalability and Environmental Compliance: As regulatory bodies worldwide tighten restrictions on industrial effluent, the "green" credentials of this synthesis become a critical asset for supply chain heads. The process generates significantly less hazardous waste compared to chlorination methods using phosphorus oxychloride or thionyl chloride, and the absence of heavy metals simplifies wastewater treatment protocols. The scalability of the reaction is inherently supported by the liquid nature of the ionic medium, which facilitates efficient heat transfer and mixing in large-scale reactors, allowing for seamless commercial scale-up of complex pharmaceutical intermediates from pilot kilogram quantities to multi-ton annual production capacities without the need for major process re-engineering.
Frequently Asked Questions (FAQ)
Understanding the technical specifics of this synthesis is crucial for stakeholders evaluating its integration into existing supply chains. The following questions address common inquiries regarding the practical implementation, purity profiles, and regulatory advantages of the ionic liquid method described in patent CN114349754B. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a transparent view of the technology's capabilities and limitations in an industrial setting.
Q: Why are chloride ionic liquids preferred over traditional solvents for 2,6-dichloropurine synthesis?
A: Chloride ionic liquids act as both solvent and catalyst, offering superior activation of the 2-amino-6-chloropurine substrate due to structural similarity (imidazole vs. purine). This significantly suppresses side reactions like azo coupling and hydrolysis, leading to yields exceeding 98% compared to roughly 60% in traditional zinc chloride methods.
Q: How does this process address heavy metal contamination concerns in pharmaceutical intermediates?
A: Conventional methods often rely on cuprous chloride or zinc chloride catalysts, which leave difficult-to-remove heavy metal residues. This novel ionic liquid process eliminates the need for transition metal catalysts entirely, ensuring the final product meets stringent purity specifications (>99%) without complex metal scavenging steps.
Q: Can the reaction mother liquor be recycled to reduce waste?
A: Yes, the process includes a specific post-treatment where the ethyl acetate extraction mother liquor is cooled to -10°C for 24 hours. This precipitates impurities and salts, allowing the filtrate to be directly reused in the next batch, drastically reducing three-waste discharge and raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Dichloropurine Supplier
The technological advancements detailed in this report underscore the immense potential of ionic liquid catalysis in modernizing the production of vital antiviral intermediates. At NINGBO INNO PHARMCHEM, we recognize that translating such innovative patent literature into commercial reality requires not just chemical expertise, but deep engineering proficiency. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of high yield and purity are realized in every batch we deliver. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace organic impurities and residual solvents, guaranteeing that our 2,6-dichloropurine meets the exacting standards required for global pharmaceutical registration.
We invite procurement leaders and R&D teams to collaborate with us to leverage this next-generation synthesis technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this metal-free route for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data from our recent pilot runs and to discuss route feasibility assessments tailored to your project timelines, ensuring a secure and cost-effective supply of this critical intermediate for your antiviral drug development programs.
