Revolutionizing Photoinitiator 819 Production: A Safe, High-Yield Hydrolysis Strategy for Industrial Scale-Up
Introduction to Advanced Photoinitiator Synthesis
The global demand for high-performance UV curing systems has necessitated a critical re-evaluation of how key photoinitiators are manufactured, particularly regarding safety and efficiency. Patent CN102942593A introduces a groundbreaking synthetic methodology for producing Bis(2,4,6-trimethylbenzoyl)phenylphosphine oxide, widely known in the industry as Photoinitiator 819. This document details a transformative shift away from hazardous alkali metal reductions toward a controlled hydrolysis pathway that utilizes water as a benign reagent. For R&D directors and supply chain leaders, this patent represents a pivotal opportunity to secure a more stable and cost-effective source of this essential polymer additive. By fundamentally altering the reaction mechanism, the inventors have addressed the chronic issues of low yield and operational danger that have plagued the traditional manufacturing of this compound for decades. The following analysis dissects the technical merits of this innovation and its profound implications for commercial procurement strategies in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
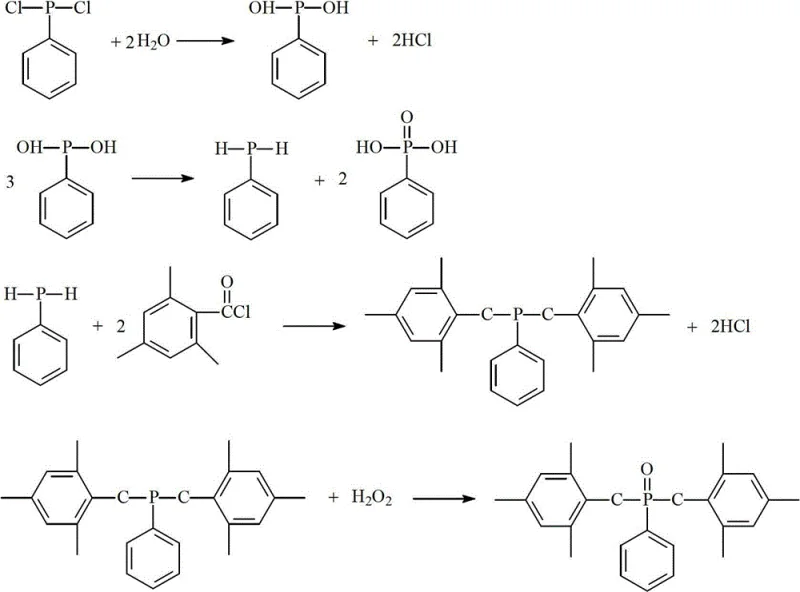
Historically, the industrial synthesis of Photoinitiator 819 has relied heavily on the reduction of phenylphosphine dichloride using highly reactive alkali metals such as sodium or potassium. This traditional approach presents severe inherent risks, primarily due to the pyrophoric nature of these metals, which can spontaneously ignite upon exposure to air or moisture, leading to catastrophic fire hazards during large-scale production. Furthermore, the quenching of excess metal and the subsequent hydrolysis or alcoholysis steps are notoriously difficult to control, often resulting in violent exothermic events that compromise operator safety and equipment integrity. From a yield perspective, these legacy processes are inefficient, typically capping at conversion rates below 60% due to the formation of complex side products and the difficulty in isolating the intermediate phenylphosphine without degradation. The waste streams generated are also problematic, containing heavy metal residues and requiring expensive treatment protocols before disposal, thereby inflating the overall environmental compliance costs for manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in CN102942593A circumvents these dangers by employing a controlled hydrolysis strategy that completely eliminates the need for dangerous metallic reducing agents. By reacting phenylphosphine dichloride directly with water under a nitrogen atmosphere, the process generates the necessary phenylphosphine intermediate in situ alongside phenylphosphonic acid as a separable byproduct. This shift not only removes the fire risk associated with alkali metals but also simplifies the reaction engineering, allowing for operation within a moderate temperature range of -10 to 100°C without the need for cryogenic cooling or specialized inert metal handling facilities. The subsequent acylation and oxidation steps are equally streamlined, utilizing common reagents like 2,4,6-trimethylbenzoyl chloride and hydrogen peroxide, which are readily available and easy to handle. This holistic redesign of the synthetic route ensures a much smoother workflow, significantly reducing the complexity of the unit operations required to bring this high-value photoinitiator to market.
Mechanistic Insights into Hydrolysis-Driven Phosphine Generation
The core innovation of this patent lies in the clever manipulation of phosphorus chemistry to generate the reactive phenylphosphine species without external reduction. As illustrated in the reaction scheme, the initial step involves the hydrolysis of phenylphosphine dichloride, which undergoes a disproportionation-like behavior in the presence of water to yield both phenylphosphine and phenylphosphonic acid. This mechanism is critical because it avoids the electron transfer complications seen in metal reductions, leading to a cleaner reaction profile with fewer unidentified impurities. The phenylphosphine generated is immediately available for the subsequent nucleophilic attack on the acyl chloride, driving the equilibrium forward and minimizing the residence time of the unstable intermediate. For R&D teams, understanding this mechanism is vital for optimizing the water addition rate and temperature profile to maximize the selectivity towards the desired mono-phosphine species rather than over-hydrolyzed phosphine oxides. The precise control of stoichiometry, specifically the molar ratio of water to dichloride, ensures that the reaction stops at the desired oxidation state, preserving the reactivity needed for the final coupling step.
Impurity control is another significant advantage conferred by this mechanistic pathway, particularly regarding the management of the phenylphosphonic acid byproduct. In traditional methods, phosphorus-containing impurities often remain dissolved in the organic phase, complicating purification and lowering the final purity of the photoinitiator. However, in this new process, the phenylphosphonic acid precipitates as a solid or can be easily separated via filtration after the initial hydrolysis step, effectively removing a major class of potential contaminants before the acylation even begins. This early-stage purification means that the downstream acylation and oxidation reactions proceed with a much higher degree of chemical fidelity, resulting in a crude product that requires less intensive recrystallization to meet stringent quality standards. The final oxidation using hydrogen peroxide is also highly selective, converting the phosphine intermediate to the phosphine oxide without attacking the sensitive benzoyl groups, ensuring that the final spectral properties of the photoinitiator remain intact for high-performance UV curing applications.
How to Synthesize Bis(2,4,6-trimethylbenzoyl)phenylphosphine oxide Efficiently
The practical implementation of this synthesis involves a straightforward four-step sequence that can be adapted to various reactor configurations, from pilot plant glassware to large-scale stainless steel vessels. The process begins with the careful addition of water to a solution of phenylphosphine dichloride in a solvent like toluene or chloroform, maintaining strict temperature control to manage the exotherm. Following the removal of the acidic byproduct, the filtrate is dried and reacted with the acyl chloride to form the key intermediate, which is then oxidized in the final step. This modular approach allows for flexibility in solvent choice and temperature tuning, enabling manufacturers to optimize the process for their specific infrastructure while maintaining the high yields and safety profiles demonstrated in the patent examples.
- Hydrolyze phenylphosphine dichloride in an organic solvent under nitrogen protection at -10 to 100°C to generate phenylphosphine and phenylphosphonic acid.
- Filter the reaction mixture to remove solid phenylphosphonic acid byproduct and dry the filtrate using a dehydrating agent.
- React the dried filtrate with 2,4,6-trimethylbenzoyl chloride at 10-80°C to form the bis(2,4,6-trimethylbenzoyl)phenylphosphine intermediate.
- Oxidize the intermediate using 30-35% hydrogen peroxide at 30-45°C, followed by separation, washing, and recrystallization to obtain the final pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this hydrolysis-based synthesis offers substantial strategic benefits that extend far beyond simple chemical efficiency. The elimination of pyrophoric metals like sodium and potassium drastically reduces the regulatory burden and insurance costs associated with storing and handling hazardous materials, leading to a leaner and more resilient operational model. Furthermore, the ability to operate without extreme conditions or specialized metal-handling equipment lowers the barrier to entry for contract manufacturing organizations, increasing the number of qualified suppliers capable of producing this material and thereby enhancing supply security. The simplified workflow also translates to shorter batch cycles and reduced downtime for cleaning and maintenance, allowing for higher throughput and better responsiveness to fluctuating market demands for UV curing additives.
- Cost Reduction in Manufacturing: The economic impact of this new route is driven by the drastic simplification of the reaction protocol and the elimination of expensive safety measures required for alkali metals. By replacing hazardous reagents with water and utilizing standard filtration techniques for byproduct removal, the process significantly lowers the operational expenditure related to waste treatment and hazard mitigation. Additionally, the generation of phenylphosphonic acid as a saleable byproduct creates a secondary revenue stream that effectively subsidizes the cost of the main product, further improving the overall margin structure for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: Supply continuity is greatly improved because the raw materials required for this synthesis, such as phenylphosphine dichloride and hydrogen peroxide, are commodity chemicals with robust global supply chains, unlike specialized metal dispersions that may face logistical bottlenecks. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by minor variations in utility supply or environmental conditions, ensuring a steady flow of high-purity photoinitiator to downstream customers. This reliability is crucial for industries like coatings and inks, where consistent quality and timely delivery are paramount for maintaining production schedules.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial tonnage is inherently safer and more straightforward due to the absence of pyrophoric hazards, facilitating faster technology transfer and commissioning of new production lines. The use of hydrogen peroxide as the oxidant results in water as the primary byproduct of the oxidation step, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing facility. This alignment with sustainability goals not only aids in regulatory compliance but also enhances the brand value of the final product for eco-conscious end-users in the polymer and coating industries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for Photoinitiator 819. These insights are derived directly from the experimental data and technical disclosures within the patent documentation to provide clarity on process capabilities. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this superior manufacturing method into their existing supply networks.
Q: How does the new hydrolysis method improve safety compared to traditional sodium reduction?
A: Traditional methods utilize pyrophoric alkali metals like sodium or potassium, posing severe fire and explosion risks during handling and quenching. The patented hydrolysis route replaces these hazardous reagents with water, fundamentally eliminating the risk of metal-induced fires and simplifying the safety protocols required for industrial operation.
Q: What is the expected yield improvement with this synthetic route?
A: While conventional industrial processes typically struggle to achieve yields exceeding 60% due to side reactions and difficult workups, this novel hydrolysis-based approach consistently demonstrates yields greater than 90%. This significant increase in efficiency directly translates to better raw material utilization and reduced waste generation per kilogram of product.
Q: Can the byproducts generated in this process be utilized commercially?
A: Yes, the primary byproduct formed during the initial hydrolysis step is phenylphosphonic acid. Unlike waste streams in traditional methods that require costly disposal, this solid byproduct can be filtered, purified, and sold as a valuable chemical intermediate, creating an additional revenue stream that offsets production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Photoinitiator 819 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN102942593A to meet the evolving needs of the global polymer industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this hydrolysis method are fully realized in large-scale manufacturing environments. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Photoinitiator 819 meets the highest performance standards required for demanding UV curing applications, providing our partners with unwavering confidence in product quality.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages specific to your operation. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for safety, efficiency, and long-term sustainability.
