Revolutionizing Photoinitiator 819 Production: A Green CO2 Fixation Strategy for Commercial Scale-Up
Introduction to Novel Photoinitiator Manufacturing Technologies
The global demand for high-efficiency photoinitiators, particularly bis(2,4,6-trimethylbenzoyl)phenylphosphine oxide (commonly known as Photoinitiator 819 or BAPO), continues to surge driven by the expansion of UV-curing applications in coatings, inks, and advanced composites. A pivotal breakthrough in the sustainable manufacturing of this critical additive is detailed in Chinese patent CN110343134B, which outlines a groundbreaking synthetic pathway that diverges from traditional acyl chloride-dependent methodologies. This patent introduces a green chemistry approach centered on the direct carboxylation of phenylphosphine metalates using carbon dioxide, followed by a robust Weinreb amide strategy to construct the molecular skeleton. For R&D directors and technical decision-makers, this represents a significant paradigm shift towards atom-economical processes that leverage inexpensive C1 building blocks rather than pre-functionalized aromatic acids. The technology not only addresses environmental concerns regarding halogenated waste but also offers a distinct economic advantage by utilizing commodity gases and readily available organometallic reagents.
The strategic importance of this innovation lies in its ability to decouple the production of high-purity photoinitiators from the volatile supply chains of specialized acyl chlorides. By fixing CO2 directly onto the phosphorus center, the process creates a versatile dicarboxylate intermediate that can be selectively transformed into the target ketone structure with exceptional precision. This method effectively bypasses the harsh conditions and safety hazards often associated with handling large volumes of acid chlorides and strong Lewis acids in conventional routes. Furthermore, the patent emphasizes the visual controllability of the reaction, where the precipitation of intermediate salts serves as an in-process control indicator, thereby enhancing operational safety and reproducibility in a commercial plant setting. As the industry moves towards greener manufacturing standards, adopting such CO2-utilization technologies positions suppliers as leaders in sustainable chemical innovation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of bis(2,4,6-trimethylbenzoyl)phenylphosphine oxide has relied heavily on the nucleophilic substitution reactions between phenylphosphine derivatives and 2,4,6-trimethylbenzoyl chloride. While effective, this traditional pathway suffers from several intrinsic drawbacks that impact both cost efficiency and environmental compliance. The primary limitation is the dependence on acyl chlorides, which are not only expensive to produce due to the requirement for thionyl chloride or oxalyl chloride but also generate stoichiometric amounts of corrosive hydrochloric acid as a byproduct. Managing this acidic waste requires substantial investment in neutralization and treatment infrastructure, driving up the overall operational expenditure for manufacturers. Additionally, the direct reaction of phosphines with acyl chlorides can sometimes lead to over-acylation or oxidation side reactions if not meticulously controlled, complicating the purification process and potentially lowering the final yield of the active photoinitiator.
Another significant challenge in conventional methods involves the handling of sensitive phosphine intermediates which are prone to spontaneous oxidation upon exposure to air, necessitating rigorous inert atmosphere protocols throughout the entire synthesis. Alternative routes involving the reduction of phosphine oxides or the use of red phosphorus often require high temperatures, high pressures, or the use of hazardous reducing agents like red aluminum or sodium metal in large excess. These conditions pose severe safety risks during scale-up and limit the flexibility of the production facility. Moreover, the reliance on specific halogenated precursors ties the supply chain to upstream chlorination capacities, creating vulnerabilities in procurement when market dynamics shift. Consequently, there is a pressing need for a synthetic route that mitigates these risks while maintaining the high purity standards required for premium UV-curing applications.
The Novel Approach
The methodology disclosed in patent CN110343134B presents a sophisticated alternative that fundamentally reimagines the construction of the P-C(O) bond. Instead of attacking a carbonyl carbon with a phosphine nucleophile, this novel approach reverses the polarity by generating a nucleophilic phosphine metalate that attacks electrophilic carbon dioxide. This carboxylation step is remarkably efficient, precipitating the phenylphosphine diformate salt directly from the reaction mixture, which allows for easy isolation and purification of the intermediate before proceeding to the next step. This "stop-and-go" capability is a major advantage for quality control, as it prevents the carryover of unreacted starting materials into subsequent stages. The subsequent conversion of this carboxylate salt into a Weinreb amide using N,O-dimethylhydroxylamine hydrochloride (DMHA) creates a stable "masked" ketone equivalent that is resistant to over-reaction.
This strategic use of Weinreb amide chemistry ensures that when the Grignard reagent or organolithium species is introduced, the reaction stops cleanly at the ketone stage without forming tertiary alcohol byproducts, a common issue in direct ester or acid chloride additions. The final oxidation step to convert the phosphine ketone to the phosphine oxide is straightforward and can be achieved using common oxidants like hydrogen peroxide. By integrating CO2 fixation with modern amide coupling techniques, this route achieves a level of selectivity and safety that is difficult to match with older technologies. The process operates under relatively mild conditions, often near room temperature or with moderate cooling, reducing energy consumption and thermal stress on the equipment. This combination of mild conditions, high selectivity, and the use of benign gaseous reagents defines the new standard for next-generation photoinitiator manufacturing.
Mechanistic Insights into CO2 Fixation and Weinreb Amide Formation
The core mechanistic brilliance of this synthesis lies in the initial activation of the phosphorus center and its subsequent reaction with carbon dioxide. In the first stage, phenylphosphine or phenylphosphine dichloride is treated with active metals such as lithium, sodium, or potassium, or alkyl metal reagents like n-butyllithium, to generate a highly nucleophilic phenylphosphine metalate species. This anionic phosphorus center possesses significant electron density, making it an ideal candidate for attacking the electrophilic carbon atom of CO2. Upon introduction of dry carbon dioxide gas, the metalate undergoes carboxylation to form a phenylphosphine dicarboxylate salt. A unique feature of this reaction, as highlighted in the patent data, is the low solubility of this dicarboxylate salt in non-polar solvents like pentane, benzene, or heptane. This property drives the equilibrium forward by precipitating the product as a colorless solid, effectively removing it from the solution phase and preventing reverse reactions or decomposition. This precipitation phenomenon serves as a built-in visual indicator of reaction completion, allowing operators to monitor the process without the need for complex analytical instrumentation.
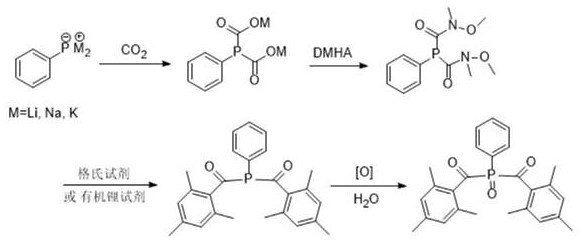
Following the isolation or in-situ utilization of the dicarboxylate salt, the mechanism proceeds through an amide coupling reaction to form the Weinreb amide. This transformation involves the activation of the carboxylate group, either through direct condensation with DMHA in the presence of a base and a coupling activator like PCl3 or HATU, or via an activated ester intermediate. The resulting N-methoxy-N-methylamide functionality is crucial because it forms a stable tetrahedral intermediate with the incoming organometallic reagent (Grignard or organolithium). Unlike simple esters or acid chlorides which collapse rapidly to release the leaving group and allow a second addition, the Weinreb amide chelates the metal cation of the Grignard reagent, stabilizing the tetrahedral intermediate and preventing further nucleophilic attack until the workup phase. During the acidic quench, this intermediate collapses to release the desired ketone product exclusively. This mechanistic safeguard is vital for ensuring the structural integrity of the bis-acyl phosphine oxide framework, preventing the formation of difficult-to-remove tertiary alcohol impurities that would otherwise compromise the photoinitiator's performance in UV curing applications.
How to Synthesize Bis(2,4,6-trimethylbenzoyl)phenylphosphine Oxide Efficiently
The implementation of this novel synthetic route requires careful attention to moisture exclusion and temperature control, particularly during the metalation and carboxylation phases. The process begins with the generation of the phosphine metalate under an inert atmosphere, followed by the controlled introduction of CO2 to precipitate the dicarboxylate intermediate. Once this key building block is secured, it is converted into the Weinreb amide, which then serves as the substrate for the carbon-carbon bond-forming step with mesityl magnesium bromide or mesityl lithium. The final oxidation step transforms the phosphine moiety into the active phosphine oxide. For detailed operational parameters, stoichiometry, and specific workup procedures required to replicate this high-yielding process in a pilot or production environment, please refer to the standardized technical guide below.
- Generate phenylphosphine metalate using active metals (Li, Na, K) and react with dry CO2 to precipitate phenylphosphine diformate salts.
- Activate the diformate salt in situ with N,O-dimethylhydroxylamine hydrochloride (DMHA) and organic base to form the stable Weinreb amide intermediate.
- Perform nucleophilic addition using 2,4,6-trimethylphenyl Grignard or organolithium reagents, followed by final oxidation to yield the target photoinitiator.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the CO2-fixation route described in patent CN110343134B offers compelling strategic benefits that extend beyond mere technical novelty. The most immediate impact is observed in the raw material cost structure, where the substitution of expensive 2,4,6-trimethylbenzoyl chloride with commodity carbon dioxide gas results in a drastic reduction in direct material costs. Carbon dioxide is not only inexpensive but also widely available, insulating the manufacturing process from the price volatility associated with specialized acylating agents. Furthermore, the elimination of stoichiometric amounts of acid chloride means that the facility no longer needs to procure, store, and handle large quantities of corrosive and hazardous chlorinating reagents. This simplification of the raw material portfolio reduces inventory holding costs and minimizes the regulatory burden associated with the transport and storage of dangerous goods, leading to substantial indirect cost savings across the logistics network.
From a supply chain reliability perspective, this process enhances resilience by diversifying the input stream. Traditional routes are often bottlenecked by the availability of high-purity acyl chlorides, which may be subject to production shutdowns or environmental restrictions at supplier sites. By shifting the synthesis to a pathway that relies on basic petrochemical feedstocks (benzene, phosphorus) and universal gases (CO2), manufacturers can secure a more stable and continuous supply of Photoinitiator 819. The robustness of the Weinreb amide intermediate also contributes to supply security; because it is a stable solid that can be isolated and stored, it acts as a buffer stock that decouples the upstream metalation steps from the downstream Grignard addition. This modularity allows for flexible production scheduling and ensures that disruptions in one part of the line do not necessarily halt the entire operation. Additionally, the visual nature of the reaction endpoints reduces the risk of batch failures due to operator error, ensuring consistent throughput and on-time delivery performance for customers.
Scalability and environmental compliance are further strengthened by the inherent safety and cleanliness of this new method. The avoidance of exothermic acyl chloride reactions reduces the cooling load on reactors, allowing for larger batch sizes without the need for expensive retrofitting of heat exchange systems. The reduction in halogenated waste streams significantly lowers the cost of wastewater treatment and hazardous waste disposal, aligning the production process with increasingly stringent global environmental regulations. This "green" credential is not just a compliance checkbox but a market differentiator that appeals to downstream formulators in the coatings and electronics industries who are under pressure to reduce the carbon footprint of their supply chains. By minimizing the E-factor (mass of waste per mass of product) and utilizing a greenhouse gas as a feedstock, this technology positions the manufacturer as a leader in sustainable chemistry, potentially unlocking premium market segments and long-term contracts with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this CO2-based synthesis route for Photoinitiator 819. These answers are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for evaluating the technology's feasibility. Understanding these details is essential for R&D teams assessing the transferability of the lab-scale protocol to commercial reactors and for procurement specialists evaluating the long-term viability of the supply source.
Q: What are the primary advantages of the CO2 fixation route over traditional acyl chloride methods?
A: The CO2 fixation route eliminates the need for expensive and corrosive 2,4,6-trimethylbenzoyl chloride in the early stages, utilizing inexpensive carbon dioxide gas instead. This significantly reduces raw material costs and simplifies waste treatment by avoiding large quantities of acidic byproducts associated with acyl chloride reactions.
Q: How does the Weinreb amide strategy improve impurity control in Photoinitiator 819 synthesis?
A: The use of Weinreb amide intermediates prevents over-addition of the Grignard reagent, which is a common side reaction in direct ketone synthesis. This ensures high selectivity for the mono-addition product, resulting in a cleaner crude profile and reducing the burden on downstream purification processes.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process features visual reaction monitoring where the formation and dissolution of precipitates indicate reaction progress. This inherent controllability, combined with the use of common solvents like THF and toluene, makes the route highly adaptable for commercial scale-up from pilot plants to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Photoinitiator 819 Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing technologies like the CO2-fixation route requires a partner with deep technical expertise and proven scale-up capabilities. As a premier CDMO and supplier in the fine chemical sector, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are translated into robust industrial realities. Our engineering teams are adept at managing the specific challenges of organometallic chemistry and gas-liquid reactions, guaranteeing that the stringent purity specifications required for high-performance photoinitiators are consistently met. With our rigorous QC labs and state-of-the-art production facilities, we are fully equipped to deliver high-purity Photoinitiator 819 that meets the exacting standards of the global UV-curing industry.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Our technical sales team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener synthesis route for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample quantities for your validation testing. By partnering with NINGBO INNO PHARMCHEM, you secure not just a product, but a strategic alliance focused on continuous improvement, cost optimization, and sustainable growth in the competitive landscape of polymer additives.
