Industrial Scale Synthesis of Beta-NMN: A Technical Breakthrough for Global Supply Chains
The global demand for Beta-Nicotinamide Mononucleotide (NMN), a critical precursor in NAD+ biosynthesis, has surged due to its potential in anti-aging and metabolic health applications. However, the transition from laboratory curiosity to commercial viability has been hindered by complex purification challenges and safety concerns regarding impurities. Patent CN111548383B presents a transformative approach to this bottleneck, detailing a robust chemical synthesis route that replaces inefficient chromatographic separations with scalable crystallization techniques. This innovation is particularly significant for a reliable pharmaceutical intermediate supplier aiming to secure the supply chain for high-value nutraceuticals. By optimizing the condensation, ammonolysis, and specifically the chiral resolution steps, this methodology ensures that the final product meets stringent purity specifications required for medical and therapeutic use, effectively bridging the gap between academic synthesis and industrial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of NMN has been plagued by significant technical barriers that prevent large-scale production. Prior art methods, such as those described in early medicinal chemistry literature, often relied heavily on activated carbon column chromatography to separate the desired beta-anomer from the alpha-anomer and other racemic impurities. This reliance on chromatography is a major liability for industrial operations because it is inherently batch-limited, consumes vast quantities of solvents, and suffers from inconsistent recovery rates. Furthermore, earlier routes frequently utilized hazardous Class 1 solvents like 1,2-dichloroethane or genotoxic reagents without adequate control strategies, rendering the final material unsuitable for human consumption. The inability to effectively resolve the chiral center without expensive preparative HPLC or chromatography meant that yields were low, costs were prohibitive, and the risk of genotoxic impurities remaining in the final active pharmaceutical ingredient was unacceptably high for regulatory bodies.
The Novel Approach
The methodology outlined in CN111548383B fundamentally reengineers the synthesis pathway to prioritize scalability and safety. Instead of relying on physical separation via chromatography, this novel approach introduces a dedicated chemical resolution step using (S)-(-)-alpha-methylbenzyl isocyanate. This allows for the separation of enantiomers through crystallization, a unit operation that is far more amenable to ton-scale manufacturing. Additionally, the process meticulously optimizes the choice of catalysts and solvents, substituting high-risk reagents with safer alternatives like acetonitrile and tetrahydrofuran. By integrating a specific quenching protocol for the triflate catalyst and employing ion exchange resins for final desalting, the process ensures that genotoxic impurities are reduced to trace levels. This shift from chromatographic dependency to chemical resolution represents a paradigm shift in cost reduction in nutraceutical manufacturing, enabling the production of high-purity NMN that is both economically viable and regulatory compliant.
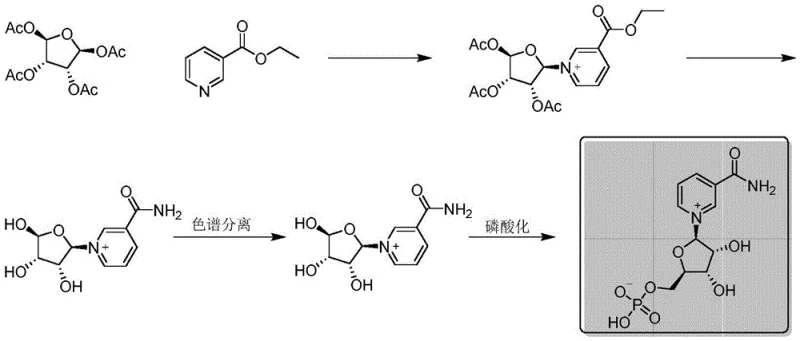
Mechanistic Insights into Chemical Resolution and Phosphorylation
The core of this technological advancement lies in the precise control of stereochemistry during the intermediate stages. The process begins with the condensation of tetraacetyl ribose and nicotinamide (or ethyl nicotinate) catalyzed by trimethylsilyl trifluoromethanesulfonate (TMsOTf). This Lewis acid catalyst activates the anomeric center of the sugar, facilitating the formation of the N-glycosidic bond. Crucially, the reaction conditions are tightly controlled at low temperatures (0°C to 5°C) to minimize side reactions, followed by a specific quenching step with triethylamine to neutralize the acidic catalyst and prevent degradation of the sensitive nucleoside intermediate. Following deprotection of the acetyl groups using sodium ethoxide, the resulting nicotinamide ribose exists as a racemic mixture. The patent's key innovation is the treatment of this racemate with a chiral resolving agent, (S)-(-)-alpha-methylbenzyl isocyanate, which forms diastereomeric salts or derivatives that exhibit different solubilities, allowing the desired beta-isomer to be isolated through repeated recrystallization.
Following the successful isolation of the chiral beta-nicotinamide ribose, the final phosphorylation step converts the primary hydroxyl group at the 5'-position into a phosphate ester using phosphorus oxychloride (POCl3). This reaction is highly exothermic and requires careful temperature management to prevent over-phosphorylation or degradation of the pyridine ring. The mechanistic precision extends to the workup procedure, where the reaction mixture is quenched in water and subjected to organic extraction to remove non-polar byproducts. The aqueous layer, containing the polar NMN product, is then passed through an ion exchange resin. This step is critical for removing inorganic salts and residual acidic species without introducing new organic contaminants. The final lyophilization yields a white solid with a confirmed structure, as evidenced by comprehensive NMR analysis including HSQC and HMBC correlations, ensuring that the commercial scale-up of complex nucleoside analogs does not compromise structural integrity or biological activity.
How to Synthesize Beta-Nicotinamide Mononucleotide Efficiently
The synthesis protocol described in the patent provides a clear roadmap for transitioning from gram-scale experiments to kilogram-level production. It emphasizes the importance of maintaining strict temperature controls during the addition of reactive reagents like TMsOTf and POCl3 to manage exotherms effectively. The procedure also highlights the necessity of monitoring reaction progress via TLC or HPLC to determine exact endpoints, preventing over-reaction which could lead to difficult-to-remove impurities. For detailed operational parameters regarding molar ratios, solvent volumes, and specific crystallization conditions, please refer to the standardized guide below.
- Condensation of tetraacetyl ribose with nicotinamide or ethyl nicotinate using a silyl catalyst like TMsOTf.
- Ammonolysis and deprotection using ammonia gas and organic bases like sodium ethoxide to remove acetyl groups.
- Chemical resolution of the racemic nicotinamide ribose intermediate using (S)-(-)-alpha-methylbenzyl isocyanate.
- Phosphorylation of the resolved beta-nicotinamide ribose using phosphorus oxychloride followed by ion exchange purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process offers substantial strategic benefits beyond mere technical feasibility. The elimination of activated carbon chromatography removes a significant bottleneck in production throughput, directly translating to improved lead times and consistent batch availability. By shifting to crystallization-based purification, the process becomes significantly more robust against variations in raw material quality, ensuring a stable supply of intermediates even during market fluctuations. Furthermore, the avoidance of restricted solvents simplifies the environmental compliance burden, reducing the costs associated with waste disposal and solvent recovery. This streamlined workflow allows manufacturers to respond more agilely to market demand without the lengthy validation cycles typically associated with chromatographic processes.
- Cost Reduction in Manufacturing: The most significant economic driver of this process is the replacement of chromatographic separation with chemical resolution and crystallization. Chromatography columns have limited lifespans, require expensive stationary phases, and consume large volumes of mobile phase solvents, all of which drive up the cost of goods sold (COGS). By utilizing standard crystallization techniques, the process leverages existing infrastructure found in most fine chemical plants, drastically lowering capital expenditure requirements. Additionally, the optimized catalyst loading (limited to 0.1 equivalents) and efficient quenching protocols minimize the consumption of expensive silyl reagents. The cumulative effect of these optimizations results in a leaner manufacturing process with significantly reduced operational expenses, making the final NMN product more price-competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability of raw materials and the simplicity of the manufacturing process. This synthesis route utilizes readily available starting materials such as tetraacetyl ribose and nicotinamide, which are produced at scale by multiple suppliers globally, mitigating the risk of single-source dependency. The robustness of the chemical resolution step ensures that even if minor variations occur in the upstream condensation reaction, the final purity can still be achieved through recrystallization. This tolerance for variance reduces the rate of batch failures and reprocessing, ensuring a steady flow of finished goods. Consequently, partners can rely on a more predictable delivery schedule, reducing lead time for high-purity nucleoside analogs and securing their own downstream production lines against interruptions.
- Scalability and Environmental Compliance: Scaling a chemical process often introduces new safety and environmental challenges, but this methodology is designed with green chemistry principles in mind. By avoiding Class 1 solvents like 1,2-dichloroethane and genotoxic agents like 1,4-dioxane, the process aligns with strict international regulatory standards such as ICH Q3C. This compliance reduces the regulatory friction when filing Drug Master Files (DMFs) or submitting dossiers for nutraceutical ingredients. From an environmental perspective, the reduced solvent load and the ability to recycle mother liquors from crystallization steps lower the overall E-factor (environmental factor) of the synthesis. The use of ion exchange resins for desalting further minimizes the generation of saline wastewater, facilitating easier treatment and discharge. These factors collectively make the process highly scalable from pilot plant to multi-ton commercial production without requiring extensive retrofitting of environmental control systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific advantages and procedural details outlined in the patent documentation, providing clarity on how this method overcomes historical limitations in NMN production. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this technology for their own manufacturing portfolios.
Q: How does this patent address the scalability issues of previous NMN synthesis methods?
A: Previous methods relied on activated carbon chromatography for chiral separation, which is difficult to scale and has low throughput. This patent introduces a chemical resolution step using crystallization, a standard unit operation that allows for industrial batch production.
Q: What measures are taken to control genotoxic impurities in this process?
A: The process strictly controls the usage of triflate catalysts (limiting to 0.1 equivalents) and employs a specific quenching step with triethylamine. Subsequent recrystallization and ion exchange steps further ensure residual levels are negligible.
Q: Why is the avoidance of Class 1 solvents significant for pharmaceutical applications?
A: Avoiding solvents like 1,2-dichloroethane and 1,4-dioxane eliminates the risk of genotoxic solvent residues in the final API. This simplifies regulatory compliance and ensures the product is safe for human consumption in nutraceutical and medical fields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Nicotinamide Mononucleotide Supplier
The technical innovations described in CN111548383B represent a significant leap forward in the industrial synthesis of Beta-Nicotinamide Mononucleotide, yet realizing this potential requires a partner with deep expertise in process development and scale-up. NINGBO INNO PHARMCHEM stands at the forefront of this capability, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art reactors capable of handling the precise temperature controls and anhydrous conditions required for the condensation and phosphorylation steps. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the absence of genotoxic impurities and ensure the correct beta-anomeric configuration in every batch we produce.
We invite global pharmaceutical and nutraceutical companies to collaborate with us to leverage this advanced synthesis technology for your supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can improve your margins. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of quality, reliability, and scientific excellence.
