Advanced Synthesis of Isochroman Derivatives for Scalable Pharmaceutical Intermediate Manufacturing
Advanced Synthesis of Isochroman Derivatives for Scalable Pharmaceutical Intermediate Manufacturing
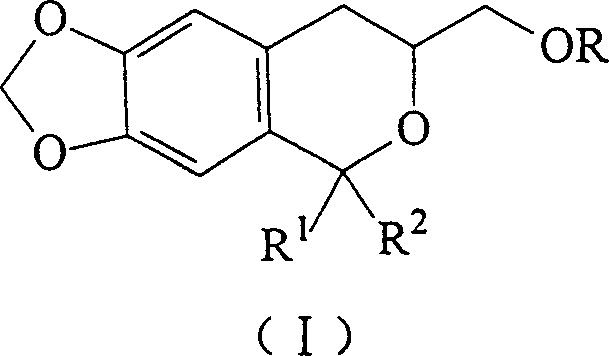
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for heterocyclic compounds that exhibit potent biological activities, such as anti-inflammatory and antibacterial properties. A significant breakthrough in this domain is documented in Chinese Patent CN1202110C, which discloses a versatile class of isochroman derivatives, specifically 3,4-dihydro-1,1-disubstituted-3-alkoxy(aryloxy)methyl-6,7-methylenedioxy-1H-benzo[c]furans. These compounds represent a critical scaffold for drug discovery, offering unique structural features that enhance pharmacological profiles. The patent outlines a novel preparation method that overcomes the severe limitations of prior art, utilizing a mild acid-catalyzed cyclization strategy that is highly amenable to industrial scale-up. By leveraging 2-alkoxy-piperonyl ethanol as a key building block, this technology enables the efficient construction of complex heterocyclic systems under relatively benign conditions. This report provides a deep technical analysis of this synthesis platform, highlighting its mechanistic elegance and commercial viability for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isochroman structural units has been plagued by harsh reaction conditions and limited substrate scope, creating significant bottlenecks for process chemists. Prior art, such as U.S. Patent 4523023, describes reactions conducted in pressure vessels at elevated temperatures around 135°C in the presence of aggressive reagents like acid anhydrides and phosphoric acid. These extreme conditions not only pose serious safety risks regarding pressure containment but also necessitate specialized, expensive reactor equipment that increases capital expenditure. Furthermore, other conventional methods, like those cited in British Patent GB1524076, rely on azeotropic water removal with tosic acid and phosphoric acid, which often leads to difficult downstream processing due to the corrosive nature of the catalysts. Crucially, existing literature has failed to provide efficient access to isochroman derivatives connected with alkoxy methylene or aryloxy methylene radicals at the 3-position, limiting the chemical space available for medicinal chemists to explore structure-activity relationships. The inability to easily introduce these functional groups restricts the potential for optimizing solubility and metabolic stability in lead compounds.
The Novel Approach
The methodology presented in CN1202110C represents a paradigm shift by introducing a mild, flexible, and operationally simple cyclization protocol. Instead of high-pressure environments, this novel approach utilizes a reflux reaction in common organic solvents such as methanol, ethanol, or toluene, driven by catalytic amounts of accessible acids like p-toluenesulfonic acid, hydrogen chloride, or boron trifluoride diethyl etherate. This shift allows the reaction to proceed at atmospheric pressure, drastically reducing energy consumption and eliminating the need for autoclaves. The process demonstrates exceptional versatility, accommodating a wide range of aldehyde and ketone compounds to generate 1,1-disubstituted variants with high efficiency. By reacting 2-alkoxy-piperonyl ethanol with these carbonyl partners, the method successfully installs the elusive 3-alkoxy(aryloxy)methyl moiety, expanding the library of accessible heterocycles. The workup procedure is equally streamlined, involving standard extraction and silica gel chromatography, which facilitates the isolation of high-purity products suitable for biological screening without the need for complex purification technologies.
Mechanistic Insights into Acid-Catalyzed Cyclization
The core of this synthetic innovation lies in the acid-catalyzed condensation and subsequent intramolecular cyclization mechanism. The reaction initiates with the protonation of the carbonyl oxygen of the aldehyde or ketone by the acid catalyst, increasing the electrophilicity of the carbonyl carbon. The hydroxyl group of the 2-alkoxy-piperonyl ethanol then acts as a nucleophile, attacking the activated carbonyl to form a hemiacetal intermediate. Under the reflux conditions, this intermediate undergoes dehydration to generate an oxocarbenium ion species. The electron-rich aromatic ring, activated by the methylenedioxy group at the 6,7-positions, then participates in an intramolecular Friedel-Crafts-type alkylation. This cyclization step closes the pyran ring, forming the stable isochroman core. The presence of the methylenedioxy group is critical, as it provides the necessary electron density to facilitate the electrophilic aromatic substitution at the ortho-position relative to the ether linkage. This mechanistic pathway is highly favorable thermodynamically, driving the equilibrium towards the cyclic product and minimizing the formation of linear oligomers or polymeric byproducts that often plague acid-catalyzed condensations.
Impurity control in this process is managed through careful optimization of the catalyst loading and reaction time, typically ranging from 10 to 60 hours depending on the steric bulk of the substrates. The use of mild organic acids like p-toluenesulfonic acid ensures that side reactions such as polymerization of the aldehyde or degradation of the sensitive methylenedioxy acetal are minimized. Following the reaction, the removal of unreacted starting materials is achieved via reduced pressure distillation, which is effective due to the volatility difference between the solvents/reactants and the higher molecular weight product. Final purification via silica gel column chromatography using a petroleum ether and ethyl acetate gradient effectively separates the target isochroman from any remaining regioisomers or trace acidic residues. This rigorous purification protocol ensures that the final API intermediate meets stringent purity specifications required for pharmaceutical applications, with the patent reporting yields consistently between 60% and 88% across various embodiments.
How to Synthesize 3,4-Dihydro-1,1-disubstituted-3-alkoxymethyl-6,7-methylenedioxy-1H-benzo[c]furan Efficiently
The synthesis of these valuable heterocyclic compounds follows a straightforward one-pot procedure that integrates condensation and cyclization steps seamlessly. The process begins by dissolving the 2-alkoxy-piperonyl ethanol precursor in a suitable solvent, followed by the addition of the chosen aldehyde or ketone in a molar excess to drive the equilibrium forward. An acid catalyst is then introduced, and the mixture is heated to reflux, allowing the reaction to proceed to completion as monitored by TLC or HPLC. Upon cooling, the reaction mixture is subjected to an aqueous workup to neutralize the acid and extract the organic product, which is then dried and concentrated. The crude material is finally purified to afford the target isochroman derivative as a white solid or oil, ready for further functionalization or biological testing. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Mix 2-alkoxy-piperonyl ethanol and aldehyde or ketone compounds in a molar ratio of 1: (1.0-20) in a polar or non-polar solvent.
- Add an acid catalyst (such as p-toluenesulfonic acid) in an amount of 1-30% molar ratio and reflux the mixture for 10-60 hours.
- Remove solvent under reduced pressure, extract with ethyl acetate, dry, and purify the residue via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented synthesis route offers substantial strategic benefits that directly impact the bottom line and operational resilience. The transition from high-pressure, high-temperature batch processes to atmospheric reflux conditions significantly lowers the barrier to entry for manufacturing, allowing production to be executed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. This compatibility with existing infrastructure eliminates the need for costly capital investments in specialized high-pressure equipment, thereby accelerating the timeline from process development to commercial production. Furthermore, the use of commodity chemicals such as methanol, acetone, and p-toluenesulfonic acid ensures a stable and cost-effective supply of raw materials, mitigating the risk of supply chain disruptions associated with exotic or hazardous reagents. The robustness of the reaction conditions also implies a wider operating window, reducing the likelihood of batch failures and ensuring consistent output quality.
- Cost Reduction in Manufacturing: The elimination of energy-intensive high-pressure operations and the use of inexpensive, recyclable solvents contribute to a drastic reduction in overall manufacturing costs. By avoiding the need for complex pressure-rated vessels and the associated safety monitoring systems, operational expenditures are significantly lowered. Additionally, the mild acid catalysts used are far cheaper and easier to handle than the corrosive phosphoric acid or acid anhydrides required in legacy methods, further reducing raw material costs and waste disposal fees. The simplified workup procedure, which relies on standard extraction and chromatography rather than complex distillation or crystallization sequences, minimizes labor hours and solvent consumption, leading to substantial cost savings in the final production budget.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials like 2-alkoxy-piperonyl ethanol and common aldehydes ensures a secure and continuous supply chain. These precursors are commercially available from multiple global suppliers, reducing dependency on single-source vendors and mitigating geopolitical risks. The simplicity of the synthesis also means that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without significant re-engineering, providing flexibility in sourcing strategies. This adaptability is crucial for maintaining supply continuity in the face of market fluctuations or unexpected production halts at specific facilities, ensuring that downstream pharmaceutical customers receive their intermediates on schedule.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently from gram-scale laboratory synthesis to multi-kilogram production without loss of yield or purity. The use of less hazardous reagents and the absence of high-pressure hazards simplify environmental, health, and safety (EHS) compliance, reducing the regulatory burden on manufacturing sites. Waste generation is minimized through the use of catalytic amounts of acid and the potential for solvent recovery, aligning with green chemistry principles and corporate sustainability goals. This environmental friendliness not only reduces disposal costs but also enhances the brand reputation of the manufacturer as a responsible supplier of fine chemicals, which is increasingly important for partnerships with major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these isochroman derivatives. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this technology into your existing R&D pipelines or supply networks. We encourage stakeholders to review these insights to fully appreciate the versatility and efficiency of this synthetic platform.
Q: What are the key advantages of this novel isochroman synthesis method over conventional high-pressure techniques?
A: Unlike conventional methods requiring high pressure (135°C) and corrosive acid anhydrides, this novel process operates at atmospheric reflux temperatures using mild acid catalysts like p-toluenesulfonic acid, significantly improving operational safety and equipment longevity.
Q: What is the typical yield range for these isochroman derivatives using this protocol?
A: Based on the patent embodiments, the process consistently achieves yields ranging from 60% to 88%, depending on the specific aldehyde or ketone substrate and the reaction duration, demonstrating robust efficiency across various structural analogs.
Q: How is the purity of the final isochroman product ensured during the workup phase?
A: High purity is achieved through a rigorous purification sequence involving aqueous extraction to remove acid catalysts, followed by drying with anhydrous magnesium sulfate and final separation using silica gel column chromatography with a petroleum ether/ethyl acetate gradient.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isochroman Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both clinical trial material and full-scale commercial manufacturing. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and potency. Our expertise in heterocyclic chemistry allows us to optimize the synthesis of complex scaffolds like the isochroman derivatives described in CN1202110C, guaranteeing a reliable supply of high-purity pharmaceutical intermediates for your drug discovery programs.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality standards. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can accelerate your path to market while optimizing your supply chain efficiency.
