Scalable One-Step Synthesis of Substituted Porphyrins for Commercial Applications
The landscape of macrocyclic compound synthesis is undergoing a significant transformation with the introduction of patent CN102775415A, which details a revolutionary synthetic method for porphyrin derivatives. This technology addresses long-standing inefficiencies in producing substituted porphyrins by enabling the simultaneous formation of three distinct structural variants—mono-, di-, and tri-substituted species—within a single reaction vessel. Unlike conventional approaches that often require parallel reactors or sequential steps to access different substitution patterns, this novel pathway leverages a unique fusion of reaction mechanisms to maximize output diversity. The strategic selection of fat-soluble substituents further enhances the practicality of the process, ensuring that the resulting complex macrocycles can be efficiently isolated using standard purification techniques. For industries relying on high-purity porphyrin intermediates for pharmaceutical or electronic applications, this represents a critical advancement in process chemistry. By consolidating multiple synthesis targets into one streamlined operation, the method not only reduces operational complexity but also significantly boosts the overall economic viability of producing these high-value fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methodologies for porphyrin synthesis, such as the Adler-Longo, MacDonald, and standard Lindsey methods, have historically presented substantial challenges for large-scale manufacturing and research flexibility. These established routes are typically designed to produce a single specific product, meaning that accessing a library of structurally related porphyrins requires running multiple independent reactions with different starting materials. This parallel processing approach drastically increases solvent consumption, catalyst usage, and labor hours, leading to inflated production costs and extended lead times. Furthermore, conventional methods often suffer from low yields and the formation of complex by-product mixtures that are notoriously difficult to separate, particularly when dealing with water-soluble variants that resist standard chromatographic techniques. The inability to efficiently generate a series of structurally regular porphyrins hinders structure-property relationship studies, which are essential for optimizing performance in applications like photodynamic therapy or organic electronics. Consequently, the industry has long sought a more versatile synthetic platform that can overcome these bottlenecks without compromising on product quality or purity.
The Novel Approach
The innovative strategy outlined in the patent data overcomes these historical barriers by employing a one-step condensation reaction between 2-pyrrole benzaldehyde and R-substituted benzaldehyde under controlled acidic conditions. This clever manipulation of reaction dynamics allows for the partial decomposition of the pyrrole aldehyde, facilitating the concurrent formation of mono-, di-, and tri-substituted porphyrins alongside a minor amount of tetra-substituted byproduct. 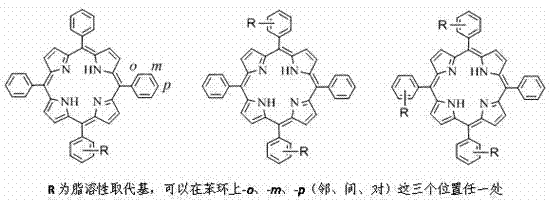 The use of fat-soluble substituting groups (R) on the benzaldehyde precursor is a critical design choice, as it imparts excellent solubility to the final products in organic solvents like chloroform and methylene chloride. This solubility profile ensures that the different porphyrin species exhibit distinct Rf values during chromatography, enabling their clean separation into discrete fractions. By generating a spectrum of substitution patterns in a single batch, this method provides researchers and manufacturers with a powerful tool for rapid material screening and cost-effective production of diverse porphyrin libraries.
The use of fat-soluble substituting groups (R) on the benzaldehyde precursor is a critical design choice, as it imparts excellent solubility to the final products in organic solvents like chloroform and methylene chloride. This solubility profile ensures that the different porphyrin species exhibit distinct Rf values during chromatography, enabling their clean separation into discrete fractions. By generating a spectrum of substitution patterns in a single batch, this method provides researchers and manufacturers with a powerful tool for rapid material screening and cost-effective production of diverse porphyrin libraries.
Mechanistic Insights into BF3-Catalyzed Condensation and Oxidation
The core of this synthetic breakthrough lies in its mechanistic hybridization, effectively merging elements of the MacDonald [2+2] condensation with the Lindsey acid-catalyzed methodology. In the initial phase, the Lewis acid catalyst, typically boron trifluoride etherate (BF3·Et2O) or trifluoroacetic acid, promotes the condensation of the aldehyde precursors to form porphyrinogen intermediates. The reaction conditions are carefully tuned to allow for the statistical distribution of the substituted and unsubstituted pyrrole units, which is the key to obtaining the mixture of substitution patterns. Following the condensation phase, the addition of a strong oxidant such as 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ) drives the aromatization of the macrocycle, locking in the conjugated 4n+2 electron system that defines porphyrin stability. 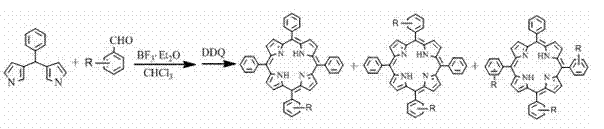 This oxidative step is crucial for establishing the rigid, planar structure required for the compound's optical and electronic properties. The mechanism ensures that while the tetraphenylporphyrin byproduct is formed, the primary yield is directed towards the mono-, di-, and tri-substituted targets, optimizing the utility of the raw materials.
This oxidative step is crucial for establishing the rigid, planar structure required for the compound's optical and electronic properties. The mechanism ensures that while the tetraphenylporphyrin byproduct is formed, the primary yield is directed towards the mono-, di-, and tri-substituted targets, optimizing the utility of the raw materials.
Impurity control in this process is managed primarily through the physical properties imparted by the fat-soluble R groups rather than complex chemical quenching. Because the substituent R (such as alkyl or alkoxy chains) renders the porphyrins highly soluble in organic developing agents, the separation of the target compounds from the reaction matrix becomes a function of polarity differences rather than solubility limits. The patent data indicates that the Rf values for the mono-, di-, and tri-substituted products are sufficiently spaced (e.g., 0.77, 0.68, 0.60) to allow for baseline resolution on silica gel columns. This predictable elution behavior minimizes the risk of cross-contamination between fractions, ensuring that the final isolated materials meet stringent purity specifications required for sensitive applications. The robustness of this purification strategy significantly reduces the need for repetitive recrystallization or specialized HPLC methods, streamlining the downstream processing workflow.
How to Synthesize Substituted Porphyrin Efficiently
The synthesis protocol described in the patent offers a straightforward yet highly effective route for producing these valuable macrocycles, suitable for both laboratory scale-up and industrial adaptation. The process begins with the dissolution of equimolar amounts of 2-pyrrole benzaldehyde and the chosen R-substituted benzaldehyde in a dry chloroform solvent under an inert nitrogen atmosphere to prevent premature oxidation.
- Mix 2-pyrrole benzaldehyde and R-substituted benzaldehyde in a chloroform solvent under nitrogen protection.
- Add a Lewis acid catalyst such as BF3·Et2O or trifluoroacetic acid and stir at room temperature for approximately 24 hours.
- Introduce an oxidant like DDQ or tetrachloro-p-benzoquinone, continue stirring for 30 hours, then purify the mixture via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible operational efficiencies and risk mitigation strategies. The ability to generate multiple product variants from a single reaction batch fundamentally alters the cost structure of porphyrin manufacturing by maximizing the utility of every kilogram of raw material processed. Instead of procuring separate batches of reagents for different substitution patterns, facilities can consolidate their purchasing and production schedules, leading to substantial reductions in inventory holding costs and logistical overhead. The simplified purification process, driven by the enhanced solubility of the fat-soluble derivatives, further decreases the consumption of expensive chromatography media and solvents, contributing to a leaner and more sustainable manufacturing footprint. These factors combined create a resilient supply chain capable of responding rapidly to fluctuating market demands for specialized porphyrin intermediates.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the reported increase in total yield, which surpasses traditional Lindsey method outputs by a margin of 15-30 percent for comparable substitution patterns. This efficiency gain means that less raw material is wasted on unreacted starting materials or intractable by-products, directly lowering the cost of goods sold (COGS). Furthermore, by eliminating the need for multiple parallel reactors to synthesize different porphyrin analogs, the method reduces energy consumption and labor allocation per unit of product. The removal of complex metal catalysts in favor of accessible Lewis acids like BF3 also avoids the costly and time-consuming heavy metal scavenging steps often required in pharmaceutical grade synthesis, resulting in additional downstream savings.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as chloroform, DDQ, and simple benzaldehyde derivatives ensures that the supply chain is not vulnerable to the shortages of exotic or highly regulated reagents. The robustness of the fat-soluble purification strategy means that production timelines are less likely to be delayed by difficult separations or failed crystallizations, which are common bottlenecks in macrocycle synthesis. This predictability allows for more accurate forecasting and tighter delivery windows, enabling manufacturers to maintain consistent stock levels of critical intermediates. Additionally, the versatility of the R group allows for the use of widely available alkyl or alkoxy precursors, further insulating the production process from raw material volatility.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram quantities is facilitated by the use of standard reaction conditions and ambient temperature stirring phases, which do not require extreme pressure or cryogenic cooling. The solvent system, primarily based on chlorinated hydrocarbons, is well-understood in industrial settings, allowing for efficient recovery and recycling systems to be implemented to minimize environmental impact. The high selectivity of the reaction reduces the volume of hazardous waste generated per kilogram of product, aligning with increasingly strict global environmental regulations. This scalability ensures that the technology can support the transition from R&D prototyping to full commercial production without the need for fundamental process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this porphyrin synthesis technology, derived directly from the patent specifications and experimental data. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production lines or research pipelines.
Q: What is the yield advantage of this new porphyrin synthesis method?
A: According to patent CN102775415A, the total yield of the porphyrins obtained by this synthetic method is higher than the yield of mono-substituted or tri-substituted porphyrin by 15-30 percent compared to using the traditional Lindsey method.
Q: How are the different porphyrin products separated?
A: The method utilizes fat-soluble substituting groups (R) on the benzaldehyde raw materials. This ensures the final porphyrins are soluble in organic developing agents, allowing for efficient separation and purification of the mono-, di-, and tri-substituted products through standard column chromatography.
Q: What catalysts and oxidants are required for this reaction?
A: The process employs common Lewis acids such as BF3·Et2O or trifluoroacetic acid as catalysts. For the oxidation step to aromatize the porphyrin ring, 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ) or tetrachloro-p-benzoquinone is used.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Porphyrin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced porphyrin synthesis method for the fine chemical and pharmaceutical sectors. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market-ready product is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of porphyrin intermediate meets the exacting standards required for high-performance applications. We understand that consistency and quality are paramount, and our technical team is prepared to validate the reproducibility of this one-step synthesis at any scale required by your operations.
We invite you to engage with our technical procurement team to discuss how this innovative route can optimize your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits tailored to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating these high-value porphyrin derivatives into your product portfolio.
