Advanced Synthesis of Substituted Acrylic Acid Phenyl Esters for Industrial Scale-Up
Advanced Synthesis of Substituted Acrylic Acid Phenyl Esters for Industrial Scale-Up
The chemical industry continuously seeks robust manufacturing pathways that balance high purity with operational efficiency, particularly for functional monomers used in advanced materials. Patent CN101781205B introduces a significant technological advancement in the synthesis of substituted acrylic acid phenyl esters, a critical class of intermediates widely utilized in the formulation of high-performance coatings, adhesives, and printing inks. Unlike traditional methods that struggle with equilibrium limitations and complex purification requirements, this patented approach leverages an acyl chloride intermediate strategy to drive reaction completion. By converting substituted acrylic acids into their corresponding acid chlorides using agents like phosphorus trichloride or thionyl chloride, the process creates a highly reactive species that readily undergoes esterification with substituted phenols. This fundamental shift in synthetic strategy not only enhances the overall reaction yield but also simplifies the downstream processing, addressing key pain points for manufacturers seeking reliable supply chains for specialty chemical intermediates.
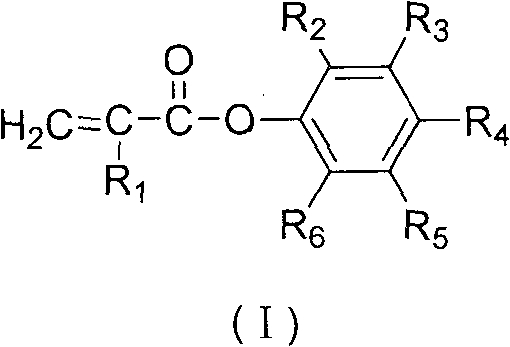
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of phenyl acrylates and methacrylates has relied heavily on transesterification reactions, typically involving the reaction of methyl methacrylate with phenols. This conventional pathway is fundamentally constrained by thermodynamic equilibrium, meaning the reaction is reversible and often fails to proceed to completion without aggressive intervention. To drive the reaction forward, manufacturers must employ complex reaction-distillation coupling operations to continuously remove by-product alcohols, such as methanol, from the system. This requirement necessitates sophisticated and energy-intensive rectification columns, significantly increasing capital expenditure and operational complexity. Furthermore, the reversible nature of the transesterification often results in limited transformation efficiency, leading to lower yields and a reaction mixture contaminated with unreacted starting materials that are difficult to separate due to similar boiling points. The reliance on strong acid catalysts in the initial esterification steps further complicates the process, introducing corrosion risks and necessitating neutralization steps that generate additional salt waste.
The Novel Approach
In stark contrast to the equilibrium-limited transesterification routes, the methodology disclosed in patent CN101781205B employs a two-step or one-step acyl chloride pathway that effectively bypasses these thermodynamic barriers. The core innovation lies in the activation of the carboxylic acid group through chlorination, generating an acryloyl chloride intermediate that is far more electrophilic than its ester counterpart. When this activated intermediate reacts with a substituted phenol, the formation of the ester bond is essentially irreversible under the specified conditions, driven by the evolution of hydrogen chloride gas which can be easily managed or scavenged. This mechanistic advantage allows for reaction conversions that are significantly higher than those achievable via transesterification, directly translating to improved material efficiency. Additionally, the process offers flexibility in the choice of chlorinating agents, including phosphorus trichloride, phosphorus oxychloride, thionyl chloride, or phosphorus pentachloride, allowing manufacturers to optimize based on local availability and waste treatment capabilities. The ability to operate without a catalyst in the preferred schemes further streamlines the workflow, eliminating the need for catalyst removal and reducing the potential for metal contamination in the final product.
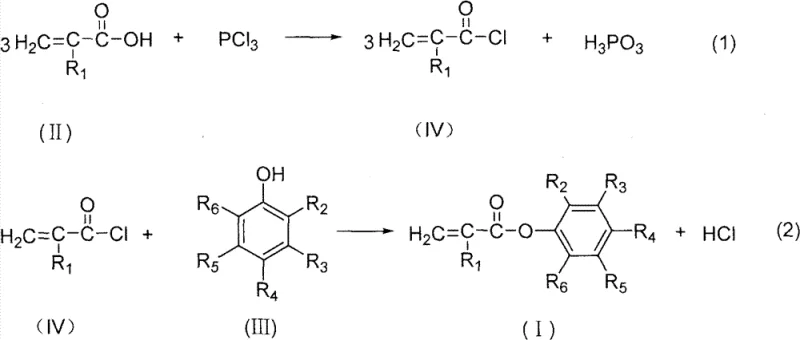
Mechanistic Insights into Acyl Chloride-Mediated Esterification
The chemical mechanism underpinning this synthesis involves a nucleophilic acyl substitution that is kinetically favorable and thermodynamically driven. In the first stage, the hydroxyl group of the substituted acrylic acid is replaced by a chlorine atom through reaction with a chlorinating agent. For instance, when using phosphorus trichloride, the reaction proceeds to form the acyl chloride along with phosphorous acid as a by-product. This step is critical because the resulting carbon-chlorine bond in the acyl chloride is weak and highly susceptible to nucleophilic attack. In the subsequent esterification stage, the oxygen atom of the phenolic hydroxyl group acts as a nucleophile, attacking the carbonyl carbon of the acyl chloride. This attack leads to the formation of a tetrahedral intermediate which rapidly collapses, expelling the chloride ion and forming the stable ester linkage. The presence of polymerization inhibitors, such as hydroquinone or cuprous chloride, is a crucial mechanistic detail; these additives function as radical scavengers that intercept any free radicals generated by thermal stress or light exposure, thereby preventing the premature polymerization of the reactive vinyl or allyl groups present in the acrylic backbone. This control is essential for maintaining the monomeric state of the product during synthesis and purification.
Impurity control is inherently built into this reaction design through the volatility differences between the intermediates and the final product. The acyl chloride intermediate, being relatively volatile, can be purified via distillation prior to the esterification step, ensuring that only high-purity reactant enters the second stage. This pre-purification step effectively removes any unreacted acrylic acid or phosphorus-containing by-products that could otherwise carry over into the final ester. Furthermore, the reaction conditions are optimized to minimize side reactions; for example, maintaining temperatures between 40°C and 75°C during the esterification phase provides sufficient energy for the reaction to proceed at a practical rate while avoiding the thermal degradation of the sensitive acrylic moiety. The use of solvents like pyridine or toluene not only facilitates heat transfer and mixing but can also act as an acid scavenger, neutralizing the evolved hydrogen chloride and preventing acid-catalyzed side reactions or equipment corrosion. The result is a product profile with exceptional purity, typically exceeding 99%, with single impurity levels maintained below 0.3%, which is a stringent specification required for high-end optical and electronic applications.
How to Synthesize Substituted Acrylic Acid Phenyl Ester Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing precise control over stoichiometry and temperature to maximize yield. The process begins with the careful selection of the chlorinating agent, where the molar ratio of acrylic acid to agent is tightly controlled, for example, using a 6:1 ratio for phosphorus trichloride or a 4:1 ratio for thionyl chloride. Following the formation of the acyl chloride, the reaction mixture is distilled to isolate the intermediate, which is then immediately reacted with the chosen substituted phenol.
- React substituted acrylic acid with a chlorinating agent such as phosphorus trichloride or thionyl chloride to generate the corresponding acryloyl chloride intermediate.
- Purify the acryloyl chloride intermediate via distillation to remove by-products and excess reagents before the next step.
- React the purified acryloyl chloride with a substituted phenol in the presence of a solvent or under melt conditions to form the final ester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this acyl chloride-based synthesis route offers substantial strategic benefits regarding cost structure and supply reliability. The elimination of equilibrium constraints means that raw material utilization is maximized, significantly reducing the volume of unreacted starting materials that must be recovered or disposed of. This high atom economy directly correlates to a reduction in the effective cost per kilogram of the final product, as less feedstock is wasted in the process. Moreover, the absence of heavy metal catalysts in the preferred embodiment removes the need for expensive and time-consuming metal scavenging steps, which are often required to meet stringent regulatory limits for pharmaceutical or food-contact applications. This simplification of the purification train not only lowers operational expenditures related to filtration media and adsorbents but also shortens the overall batch cycle time, enhancing the throughput capacity of existing manufacturing assets without the need for major capital investment in new equipment.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the dependency on complex reaction-distillation coupling systems that are energy-intensive and maintenance-heavy. By shifting to a simpler batch or semi-batch operation that relies on standard stirred reactors and basic distillation units, facilities can reduce their utility consumption, particularly steam and cooling water, which are major cost drivers in chemical processing. The high yield reported in the patent examples implies that less raw material is required to produce a fixed amount of product, providing a buffer against volatility in the prices of acrylic acid and phenol derivatives. Additionally, the ability to recover and reuse certain reagents, such as thionyl chloride, further contributes to a leaner cost structure, ensuring that the manufacturing process remains economically viable even when raw material markets fluctuate.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this synthetic route ensures consistent product quality and availability, which is critical for downstream customers in the coating and adhesive industries. The tolerance for various substituents on both the acrylic acid and the phenol ring allows for a flexible production platform capable of manufacturing a diverse portfolio of esters using the same core infrastructure. This versatility mitigates the risk of supply disruptions caused by the shortage of a specific niche intermediate, as the process can be adapted to utilize alternative feedstocks if necessary. Furthermore, the simplified purification requirements mean that production bottlenecks associated with complex separation columns are eliminated, leading to more predictable lead times and the ability to respond rapidly to changes in market demand without compromising on the stringent purity specifications required by high-value applications.
- Scalability and Environmental Compliance: The environmental profile of this method is markedly improved compared to traditional transesterification, primarily due to the reduction in waste generation and the avoidance of strong acid catalysts. The by-products formed, such as phosphorous acid or hydrogen chloride, are well-understood chemicals that can be efficiently neutralized or valorized, simplifying wastewater treatment and reducing the environmental footprint of the facility. The scalability of the process is demonstrated by the successful transition from laboratory flasks to larger reactor volumes without loss of yield or purity, indicating that the chemistry is robust enough for multi-ton commercial production. This ease of scale-up reduces the technical risk associated with technology transfer, allowing companies to bring new products to market faster while maintaining compliance with increasingly rigorous global environmental regulations regarding volatile organic compounds and hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the detailed specifications and examples provided in the patent documentation. These insights are intended to clarify the operational parameters and quality outcomes associated with the acyl chloride route, helping stakeholders make informed decisions about adopting this method for their specific manufacturing needs.
Q: What are the primary advantages of the acyl chloride route over transesterification?
A: The acyl chloride route avoids the equilibrium limitations inherent in transesterification reactions, leading to significantly higher conversion rates and eliminating the need for complex distillation setups to shift equilibrium.
Q: How is polymerization controlled during the synthesis of acrylic esters?
A: The process utilizes specific polymerization inhibitors such as hydroquinone or cuprous chloride added during the reaction phases to effectively suppress the polymerization of the reactive double bond.
Q: What purity levels can be achieved with this manufacturing method?
A: The described method consistently achieves product purity greater than 99% with single impurity concentrations below 0.3%, making it suitable for high-performance coating and adhesive applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Acrylic Acid Phenyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation coatings and adhesives. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN101781205B are fully realized in a commercial setting. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch meets the >99% purity benchmark with minimal impurity profiles. Our commitment to quality assurance means that we can consistently deliver materials that perform reliably in your final formulations, reducing the risk of batch-to-batch variability that can plague less controlled manufacturing processes.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific application requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how our optimized process can lower your total cost of ownership. Please contact us to request specific COA data for our current inventory or to discuss route feasibility assessments for custom derivatives, ensuring that your supply chain is built on a foundation of chemical excellence and operational reliability.
