Advanced Synthesis of Substituted Acrylic Acid Phenyl Esters for Industrial Scale Production
Advanced Synthesis of Substituted Acrylic Acid Phenyl Esters for Industrial Scale Production
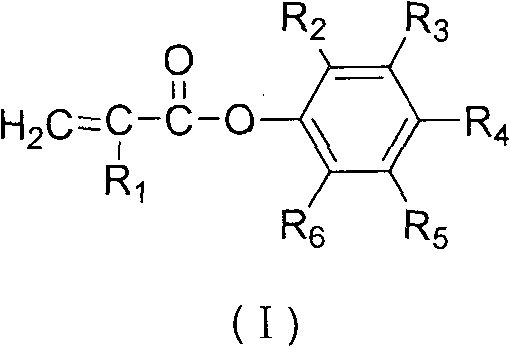
The chemical industry constantly seeks more efficient pathways for producing high-performance monomers, particularly those used in radiation-curable coatings and adhesives. Patent CN101781205A introduces a groundbreaking method for synthesizing substituted acrylic acid phenyl esters, addressing critical bottlenecks in yield and purity that have plagued conventional manufacturing. This technology leverages a robust acylation strategy to produce compounds with active double bonds, essential for UV curing applications, while achieving product purities exceeding 99%. By shifting away from equilibrium-limited transesterification, this process offers a reliable substituted acrylic acid phenyl ester supplier pathway that ensures consistent quality and operational efficiency for large-scale chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of phenyl acrylates and methacrylates has relied heavily on transesterification reactions, a process fraught with thermodynamic limitations. As detailed in prior art such as CN1321102C, traditional methods often involve reacting methyl methacrylate with phenols, a reversible reaction that inherently restricts the transformation efficiency of the reactants. To drive the equilibrium forward, manufacturers must employ complex reaction-distillation coupling operations to continuously remove by-product alcohols, which significantly increases energy consumption and equipment complexity. Furthermore, these reversible systems often suffer from lower conversion ratios, necessitating multiple rectification steps to isolate the target ester, thereby driving up separation costs and reducing the overall economic viability of the manufacturing process.
The Novel Approach
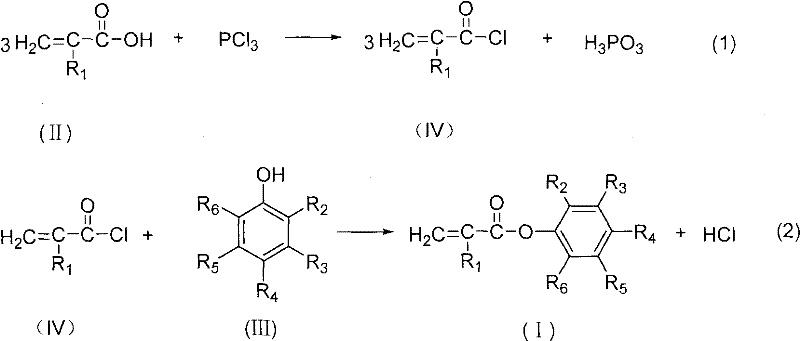
In stark contrast, the methodology disclosed in CN101781205A circumvents these thermodynamic barriers by utilizing an irreversible acylation route via acid chlorides. This novel approach involves first converting substituted acrylic acid into the corresponding acryloyl chloride using chlorinating agents like phosphorus trichloride or thionyl chloride, followed by esterification with substituted phenols. Because the formation of the ester from an acid chloride is not an equilibrium-limited process in the same way transesterification is, the reaction proceeds with much higher conversion rates and yields. This fundamental shift in reaction chemistry eliminates the need for continuous by-product removal to drive equilibrium, simplifying the reactor design and allowing for cost reduction in coating additive manufacturing through streamlined processing and reduced energy inputs.
Mechanistic Insights into Acid Chloride-Mediated Esterification
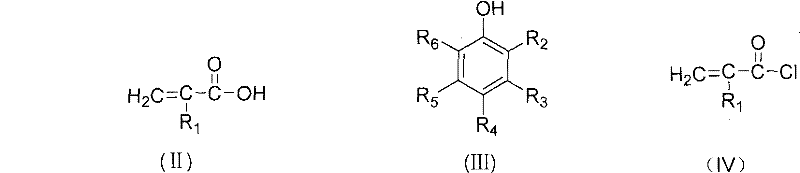
The core of this synthesis lies in the high reactivity of the acid chloride intermediate, which acts as a potent electrophile towards the nucleophilic oxygen of the phenol. In the first stage, the carboxylic acid group of the substituted acrylic acid reacts with the chlorinating agent to replace the hydroxyl group with a chlorine atom, generating the highly reactive acyl chloride species shown in Formula IV. This activation step is crucial as it lowers the activation energy for the subsequent nucleophilic attack by the phenol. The reaction conditions are carefully controlled, typically heating the mixture to boiling and then cooling to moderate temperatures (30-70°C) to manage the exothermic nature of the chlorination while minimizing side reactions such as the polymerization of the acrylic double bond.
Following the formation of the acid chloride, the second stage involves the nucleophilic substitution where the phenolic hydroxyl group attacks the carbonyl carbon of the acid chloride. This step releases hydrogen chloride as a by-product, which can be neutralized or managed within the system. A critical aspect of this mechanism is the inclusion of polymerization inhibitors, such as cuprous chloride or hydroquinone, which are added to stabilize the unsaturated vinyl or allyl groups present in the molecule. These inhibitors scavenge free radicals that might initiate premature polymerization, ensuring that the final product remains a monomeric ester with high purity (>99%) and low impurity content (<0.3%), which is vital for its performance in sensitive optical or coating applications.
How to Synthesize Substituted Acrylic Acid Phenyl Ester Efficiently
The synthesis protocol outlined in the patent provides a clear, scalable roadmap for producing these valuable intermediates with high fidelity. The process begins with the precise stoichiometric mixing of the substituted acrylic acid and the chosen chlorinating agent, followed by a controlled thermal cycle to generate the intermediate acid chloride. Once the intermediate is distilled to purity, it is reacted with the substituted phenol, either in a solvent like pyridine or toluene, or under solvent-free melt conditions depending on the melting point of the phenol. For a comprehensive understanding of the specific operational parameters, temperature gradients, and workup procedures required to replicate this high-yield process, the detailed standardized synthesis steps are provided in the guide below.
- React substituted acrylic acid with phosphorus trichloride, phosphorus oxychloride, thionyl chloride, or phosphorus pentachloride to generate the corresponding acryloyl chloride intermediate.
- Purify the acryloyl chloride intermediate via distillation to remove excess reagents and by-products.
- React the purified acryloyl chloride with substituted phenol in the presence of a solvent or under melt conditions at 40-75°C to form the final ester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis technology translates directly into enhanced operational resilience and financial efficiency. By eliminating the need for complex equilibrium-shifting equipment and reducing the number of distillation columns required for purification, the capital expenditure for setting up production lines is significantly lowered. The high reaction yield means that less raw material is wasted as unreacted starting material or by-products, leading to substantial cost savings in raw material procurement. Furthermore, the simplicity of the workup procedure, often requiring only a single recrystallization or vacuum distillation to achieve >99% purity, drastically reduces the utility costs associated with prolonged heating and pumping, making the overall manufacturing process leaner and more sustainable.
- Cost Reduction in Manufacturing: The elimination of strong acid catalysts typically required in traditional esterification methods removes the need for expensive neutralization and washing steps, which generates large volumes of wastewater. Additionally, the ability to recover and reuse excess chlorinating agents, such as thionyl chloride, further drives down the variable cost per kilogram of the final product. This streamlined chemical pathway ensures that the production of high-purity acrylic monomers becomes economically viable even at smaller batch sizes, offering flexibility in meeting diverse market demands without compromising on margin.
- Enhanced Supply Chain Reliability: The robustness of this two-step or one-step synthesis ensures a consistent supply of critical intermediates for the coatings and adhesives industries. Since the reaction does not rely on sensitive biological catalysts or rare earth metals that might be subject to geopolitical supply constraints, the raw material sourcing is stable and predictable. The high conversion rates minimize the accumulation of difficult-to-separate impurities, reducing the risk of batch failures and ensuring that delivery schedules for high-purity intermediates are met with greater reliability and consistency.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory glassware to multi-ton industrial reactors without significant re-engineering of the reaction kinetics. The reduction in solvent usage, particularly in the solvent-free melt variation of the process, aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. By generating fewer by-products and utilizing recyclable reagents, this method supports the commercial scale-up of complex fine chemicals while maintaining a smaller environmental footprint, a key metric for modern sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. Understanding these nuances is essential for R&D teams looking to integrate this technology into their existing production portfolios. The answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for process evaluation.
Q: What are the primary advantages of this synthesis method over traditional transesterification?
A: Unlike traditional transesterification which is a reversible reaction with limited conversion rates, this method utilizes an irreversible acylation route. This results in significantly higher reaction yields (>85% for two-step) and simplifies the purification process, eliminating the need for complex reaction-distillation coupling operations.
Q: How does the process control polymerization during the reaction?
A: The process effectively controls polymerization by incorporating specific polymerization inhibitors such as cuprous chloride, hydroquinone, or phenothiazine. These inhibitors are added during the formation of the acid chloride or the final esterification step, ensuring the stability of the unsaturated double bond and preventing the formation of polymeric by-products.
Q: Can this synthesis be performed in a single step?
A: Yes, the patent describes a viable one-step variation where the substituted acrylic acid, chlorinating agent, inhibitor, and substituted phenol are mixed together. While the two-step method offers slightly higher yields (>85%), the one-step method provides a streamlined alternative with yields exceeding 75%, offering flexibility depending on production priorities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Acrylic Acid Phenyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance monomers play in the advancement of next-generation coatings and electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that utilize advanced chromatographic techniques to verify every batch. Our capability to handle complex esterification chemistries allows us to offer a reliable supply of specialized intermediates that empower your downstream applications.
We invite you to collaborate with us to optimize your supply chain for these essential chemical building blocks. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to determine how our advanced synthesis capabilities can enhance your product portfolio and reduce your overall manufacturing costs.
