Advanced Total Synthesis of D-Penicillamine: Scalable Chiral Technology for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical active pharmaceutical ingredients that ensure patient safety while optimizing production efficiency. Patent CN111909067A introduces a groundbreaking organic total synthesis method for D-penicillamine, a vital chelating agent used extensively in treating rheumatoid arthritis and heavy metal poisoning. Unlike traditional methods that rely on the hydrolysis of penicillin, this novel approach utilizes L-serine ester derivatives as the chiral starting material, fundamentally altering the risk profile and economic feasibility of production. By leveraging the inherent chirality of abundant amino acids, this process bypasses the need for complex resolution steps or expensive chiral catalysts, offering a streamlined route to high-purity intermediates. For R&D directors and procurement specialists, understanding this shift from semi-synthesis to total synthesis is crucial for securing a reliable D-penicillamine supplier capable of meeting stringent regulatory standards without allergen contamination. This report analyzes the technical merits and commercial implications of this patented technology for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the commercial production of penicillamine has relied heavily on the semi-synthetic hydrolysis of penicillin G or V, a process that carries inherent biological risks and supply chain vulnerabilities. As illustrated in the reaction pathway below, the cleavage of the beta-lactam ring in penicillin to yield D-penicillamine often results in trace residues of the parent antibiotic remaining in the final product. For patients with severe penicillin allergies, even minute quantities of residual penicillin can trigger life-threatening anaphylactic reactions, rendering such batches unsuitable for specific clinical populations. Furthermore, the fermentation-based supply of penicillin is subject to biological variability, seasonal fluctuations, and complex purification requirements to remove fermentation byproducts. The structural complexity of the penicillin molecule also necessitates harsh hydrolysis conditions that can degrade the sensitive thiol group, leading to lower overall yields and increased formation of disulfide impurities that are difficult to separate. These factors collectively increase the cost of goods sold and introduce significant quality control challenges for manufacturers aiming to produce high-purity D-penicillamine.
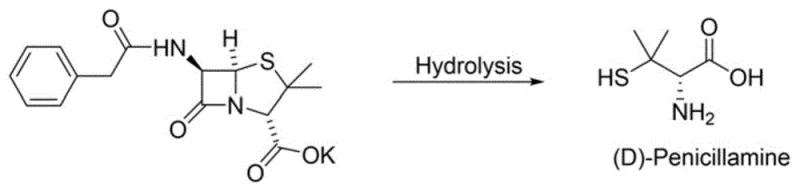
The Novel Approach
In stark contrast to the biological limitations of penicillin hydrolysis, the novel total synthesis route described in the patent utilizes a chiral pool strategy starting from L-serine ester derivatives, which are cheap, stable, and readily available in high optical purity. This method completely eliminates the risk of penicillin allergen contamination, thereby expanding the potential patient demographic and simplifying the safety validation process for regulatory filings. The synthetic pathway is designed to be linear and convergent, avoiding the formation of racemic mixtures that typically plague non-chiral synthetic routes and require costly chromatographic separation. By constructing the carbon skeleton through controlled Grignard addition and functional group interconversion, the process ensures that the stereochemical integrity of the starting L-serine is preserved throughout the sequence. This approach not only enhances the safety profile of the final API but also offers a more predictable and controllable manufacturing process that is less dependent on biological fermentation cycles. Consequently, this represents a significant advancement in cost reduction in pharmaceutical intermediates manufacturing by stabilizing raw material costs and improving process reliability.
Mechanistic Insights into L-Serine Based Chiral Synthesis
The core of this innovative synthesis lies in the precise manipulation of the L-serine backbone through a five-step sequence that meticulously builds the gem-dimethyl and thiol functionalities characteristic of D-penicillamine. The process initiates with a Grignard reaction where a methyl Grignard reagent attacks the ester functionality of the protected L-serine derivative at low temperatures, typically around -30°C, to form a tertiary alcohol intermediate. This step is critical as it introduces the two methyl groups at the beta-position while maintaining the chiral center at the alpha-carbon, a transformation that requires careful temperature control to prevent epimerization. Following this, an oxidation step converts the primary alcohol moiety into a carboxylic acid using mild oxidants like bleach water, ensuring that the sensitive sulfur-containing groups introduced later are not prematurely oxidized to sulfones or sulfoxides. The subsequent sulfonylation activates the tertiary hydroxyl group, making it a suitable leaving group for nucleophilic substitution, which is then displaced by a sulfur nucleophile in the thioesterification step. Finally, a rigorous hydrolysis under acidic conditions removes all protecting groups, such as the Boc or Cbz groups, to liberate the free amine and thiol, yielding the target D-penicillamine hydrochloride which can be further neutralized to the free base.
Impurity control is inherently built into this mechanistic pathway through the use of high-purity starting materials and the avoidance of racemization-prone conditions. Since the chirality is derived directly from the L-serine starting material, which is commercially available with ee values exceeding 99%, the final product inherits this high optical purity without the need for chiral resolution. The patent data indicates that the final recrystallization steps effectively remove any minor diastereomeric impurities or unreacted intermediates, resulting in a final product with purity levels reaching 99.4% and ee values of 99%. This high level of stereochemical control is paramount for R&D directors focusing on the impurity profile of the API, as enantiomeric impurities can have distinct toxicological profiles and must be strictly controlled according to ICH guidelines. The mechanism avoids the use of transition metal catalysts that could leave toxic metal residues, further simplifying the purification train and reducing the burden on analytical quality control teams. This clean reaction profile ensures that the commercial scale-up of complex amino acid derivatives remains feasible and compliant with global pharmacopeia standards.
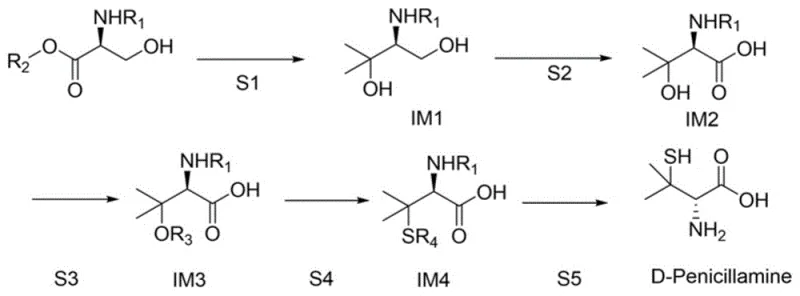
How to Synthesize D-Penicillamine Efficiently
The operational execution of this synthesis requires precise adherence to the reaction parameters outlined in the patent to ensure consistent quality and yield across different batch sizes. The process begins with the preparation of the Grignard adduct, followed by oxidation, sulfonylation, thioesterification, and final hydrolysis, each step requiring specific workup procedures to isolate the intermediates in high purity. Detailed standard operating procedures for each transformation, including reagent stoichiometry, solvent choices, and temperature profiles, are essential for technology transfer from the laboratory to the pilot plant. Manufacturers must pay particular attention to the exothermic nature of the Grignard addition and the acidic conditions required for the final deprotection to ensure safety and product integrity. The following guide outlines the critical operational milestones necessary to replicate this high-efficiency pathway.
- Perform Grignard reaction on L-serine ester derivative with methyl Grignard reagent at -30°C to form the first intermediate.
- Execute oxidation reaction on the first intermediate using bleach water to obtain the corresponding carboxylic acid intermediate.
- Conduct sulfonylation followed by thioesterification and final hydrolysis to yield D-penicillamine with high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this total synthesis route offers substantial advantages over traditional methods by decoupling production from the volatile fermentation markets associated with penicillin. The reliance on L-serine derivatives, which are produced via large-scale fermentation of carbohydrates, provides a more stable and predictable raw material supply chain that is less susceptible to the biological contaminants and supply shocks often seen in antibiotic production. This shift allows supply chain heads to negotiate long-term contracts with greater confidence, knowing that the raw material base is robust and widely available from multiple global suppliers. Furthermore, the synthetic route's simplicity and use of common organic reagents reduce the dependency on specialized catalysts or reagents that might have single-source supply risks. This diversification of the supply base is a critical factor in reducing lead time for high-purity chiral intermediates, ensuring that manufacturing schedules can be maintained even during periods of global raw material scarcity.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive chiral resolution steps and the use of low-cost, commodity-grade starting materials. By inheriting chirality from L-serine, the process avoids the significant material loss associated with racemic synthesis and subsequent separation, effectively doubling the theoretical yield compared to non-chiral routes. Additionally, the absence of heavy metal catalysts removes the need for costly scavenging steps and extensive testing for metal residues, which significantly lowers the operational expenditure per kilogram of product. The mild reaction conditions also translate to lower energy consumption for heating and cooling, contributing to overall utility savings in a large-scale plant. These factors combine to create a manufacturing cost structure that is significantly more competitive than semi-synthetic alternatives, allowing for better margin management in a price-sensitive generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The robustness of this synthetic pathway enhances supply chain continuity by minimizing the number of critical process parameters that can cause batch failures. The use of stable intermediates and straightforward workup procedures reduces the risk of production delays caused by complex purification bottlenecks or equipment compatibility issues. Since the raw materials are non-hazardous and widely traded chemicals, procurement teams can easily source alternatives in case of vendor disruptions, ensuring a continuous flow of materials to the production line. This reliability is crucial for maintaining the inventory levels required by just-in-time manufacturing models prevalent in the pharmaceutical industry. Consequently, partners can expect a more consistent delivery schedule and a reduced risk of stockouts, which is vital for maintaining patient access to essential medications.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from liter-scale reactors to multi-ton production vessels without significant re-optimization. The absence of toxic reagents and the generation of benign byproducts simplify waste treatment protocols, aligning with increasingly stringent environmental regulations and sustainability goals. This green chemistry profile reduces the environmental compliance burden and associated disposal costs, making the facility more sustainable in the long term. The high atom economy of the route ensures that raw materials are efficiently converted into the final product, minimizing waste generation and maximizing resource utilization. These environmental and operational efficiencies make the process highly attractive for manufacturers looking to expand capacity while adhering to corporate social responsibility mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived from the specific benefits and limitations identified in the patent literature. Understanding these aspects is essential for stakeholders evaluating the feasibility of adopting this route for their specific product portfolios. The answers provided are based on the technical data and comparative analysis presented in the preceding sections of this report.
Q: Why is total synthesis preferred over penicillin hydrolysis for D-penicillamine?
A: Traditional semi-synthesis via penicillin hydrolysis often leaves penicillin residues, posing severe allergy risks to patients. Total synthesis from L-serine eliminates this biological hazard entirely.
Q: How does the L-serine route ensure high optical purity?
A: The process utilizes the chiral pool strategy, inheriting the stereochemistry from commercially available high-purity L-serine esters, avoiding complex chiral induction steps that often lower ee values.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild, reagents are inexpensive and readily available, and the process avoids toxic residues, making it highly adaptable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-Penicillamine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing, leveraging advanced synthetic technologies like the one described in CN111909067A to deliver superior value to our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of D-penicillamine meets the highest international standards for safety and efficacy. Our infrastructure is designed to handle complex chiral syntheses with precision, providing a secure and reliable source for your critical pharmaceutical intermediates.
We invite you to collaborate with us to optimize your supply chain and achieve significant operational efficiencies through the adoption of this advanced synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate how our capabilities align with your project goals. By partnering with us, you gain access to a wealth of technical expertise and a commitment to quality that drives success in the competitive pharmaceutical landscape.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
